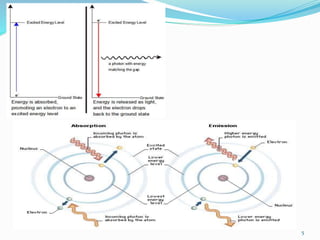
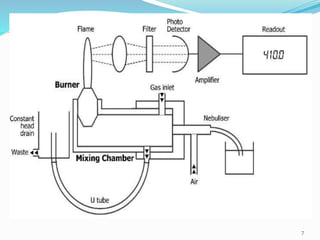
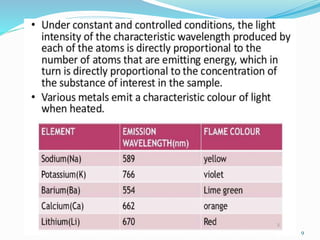
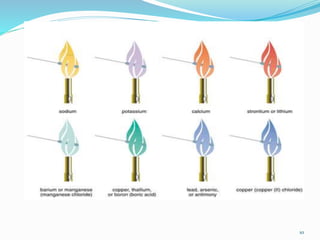
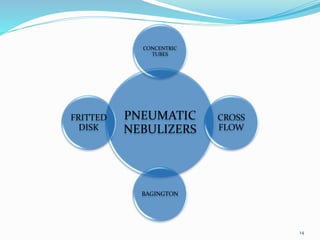
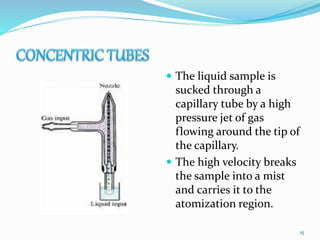
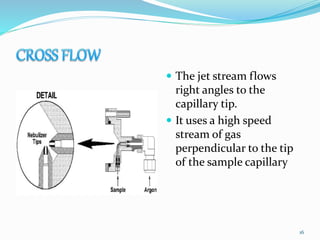
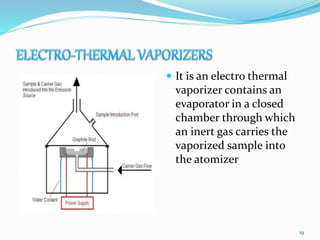
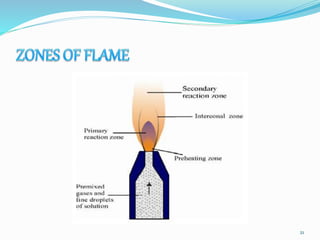
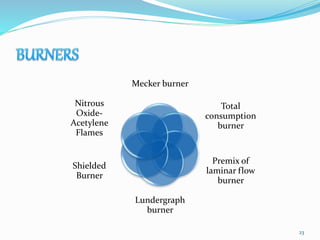
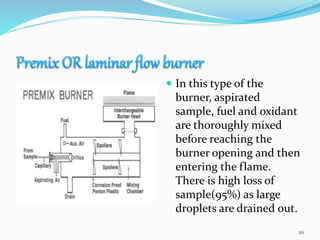
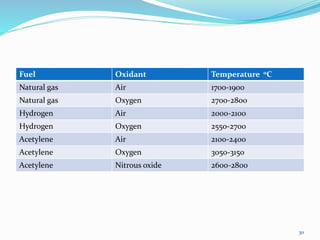
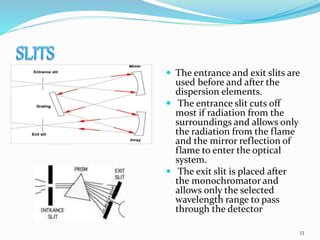
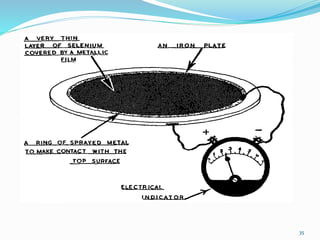
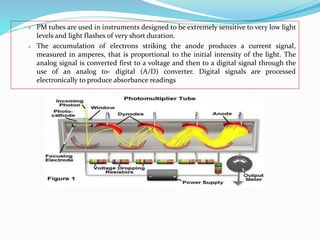
This document provides an overview of flame photometry, which uses the intensity of light emitted from atoms in a flame to determine the concentration of certain metal ions in solutions. It describes the basic principles, where a sample is nebulized into a flame and the heat excites the metal atoms to emit light at specific wavelengths. It then discusses the key components of a flame photometer in detail, including the nebulizer, burners, mirrors, slits, and detectors used to measure the light intensities and determine concentrations. Limitations of the technique are that only certain elements can be analyzed as solutions that are volatile in flames.