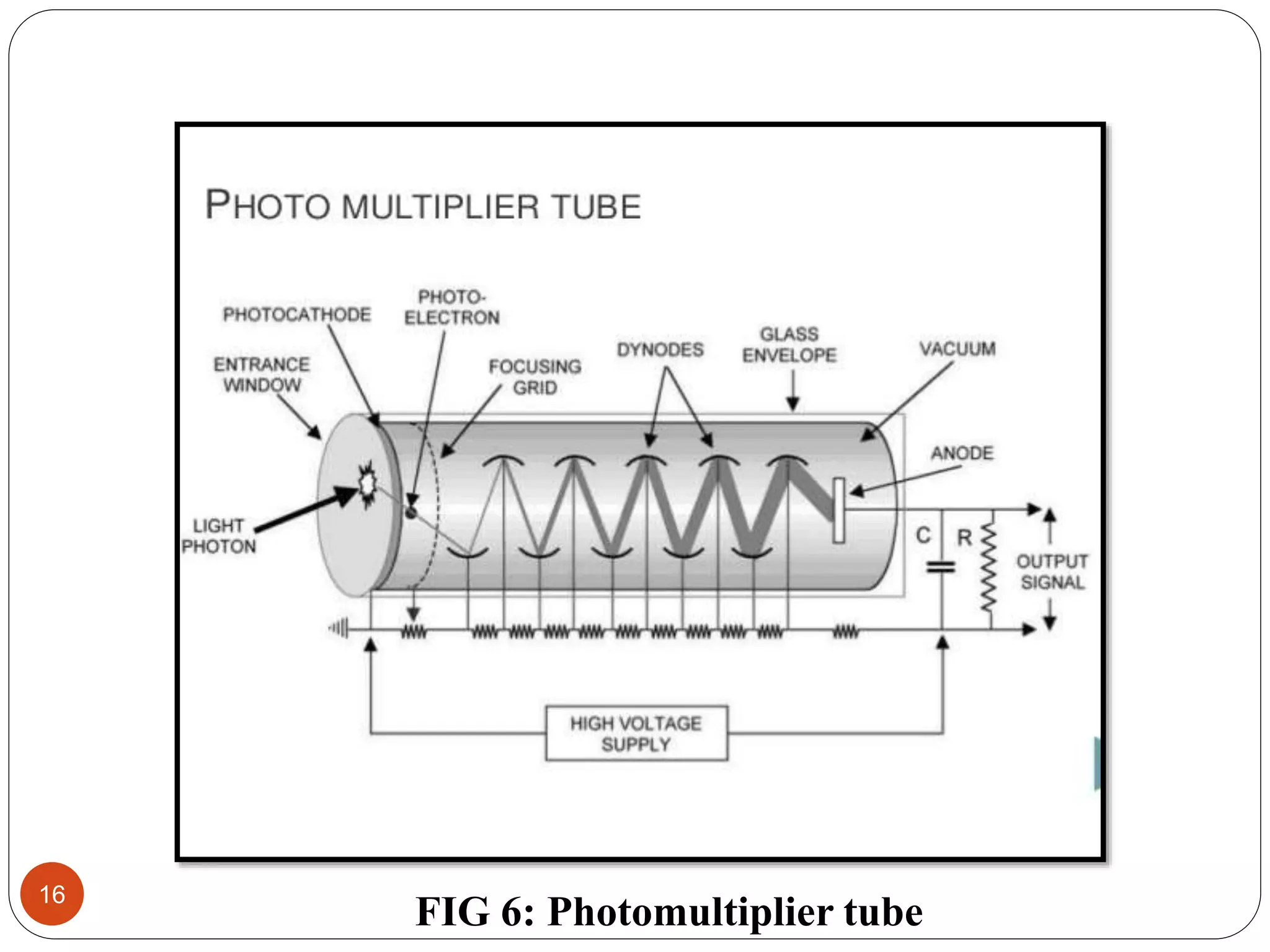
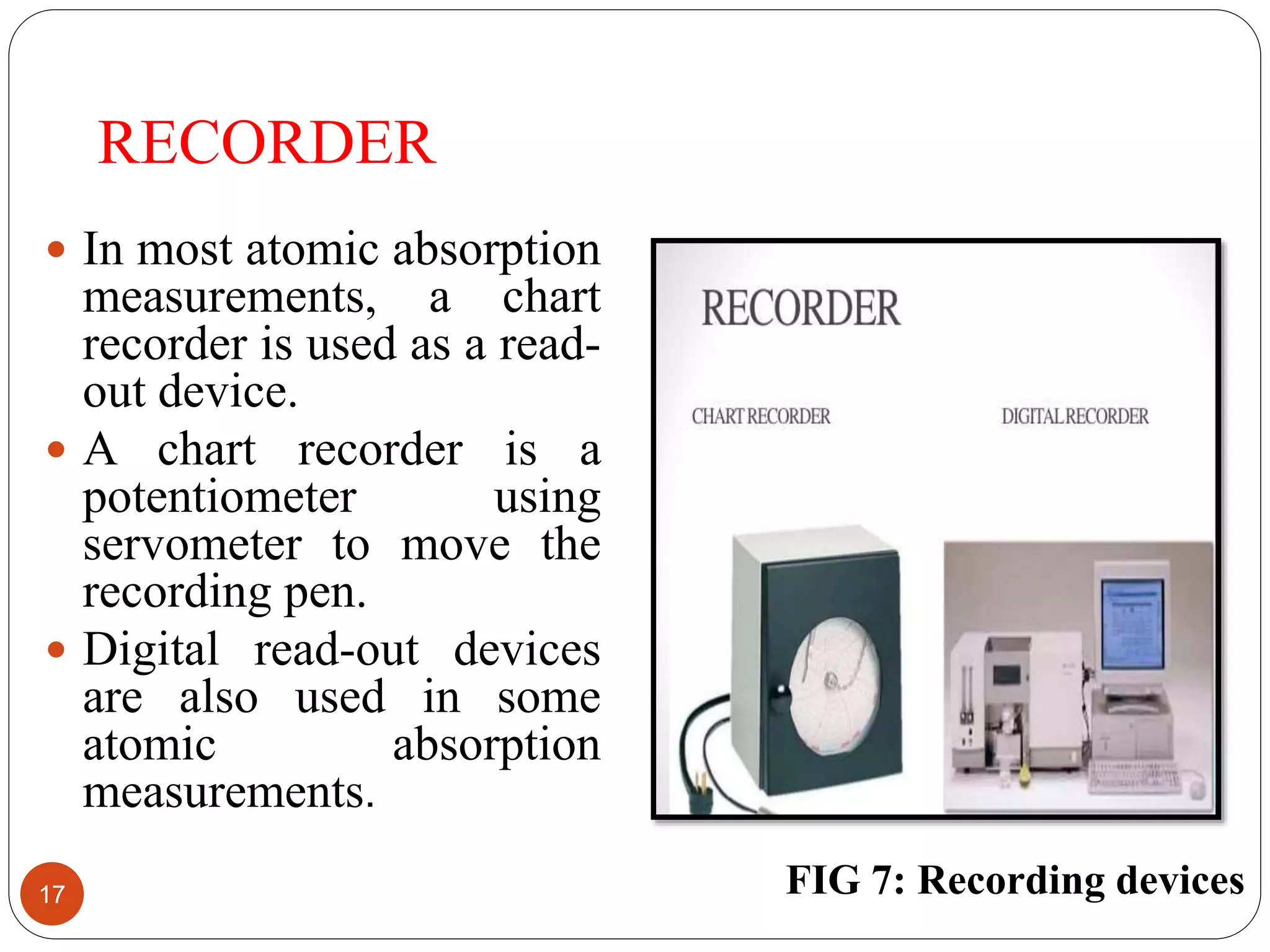
This document discusses atomic absorption spectroscopy, including its principle, instrumentation, and applications. Atomic absorption spectroscopy works by vaporizing a sample into neutral atoms that can absorb radiation from a hollow cathode lamp at specific wavelengths. The instrumentation includes a lamp, atomizer to vaporize the sample, monochromator to select wavelengths, detector such as a photomultiplier tube, and recorder. Applications include determining small amounts of metals in environmental, food, pharmaceutical, and other samples.