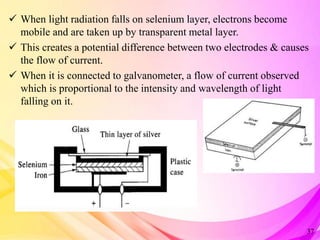
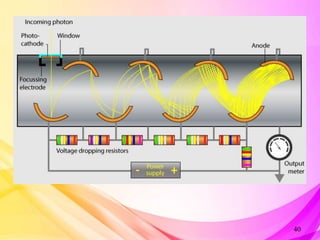
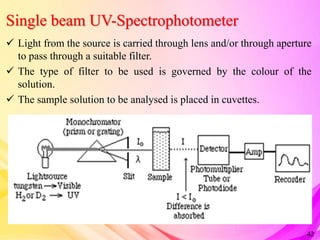
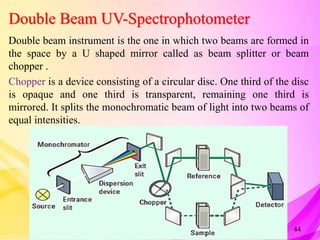
This document provides an overview of UV-Visible Spectroscopy. It discusses the basic principles including electromagnetic radiation, interaction of radiation with matter, and electronic transitions. It describes Beer-Lambert's law and how absorbance is directly proportional to concentration and path length. Different types of electronic transitions like σ→σ*, n→σ*, π→π*, and n→π* are explained. Instrumentation components like radiation sources, monochromators, sample holders and detectors are briefly outlined. Key terms like chromophore, auxochrome, bathochromic shift, hypsochromic shift, hyperchromic effect and hypochromic effect are also defined.