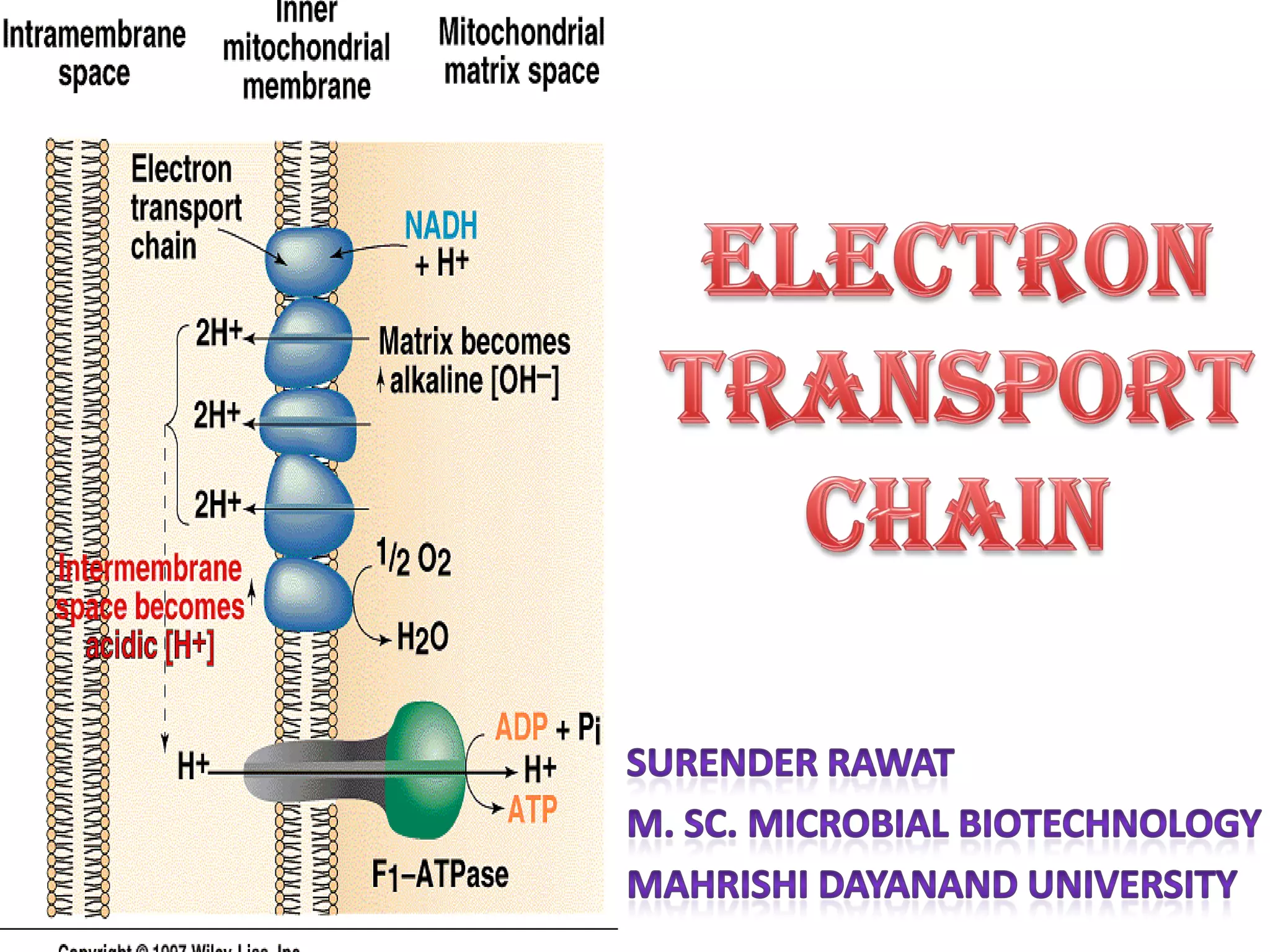
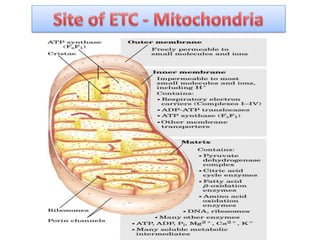
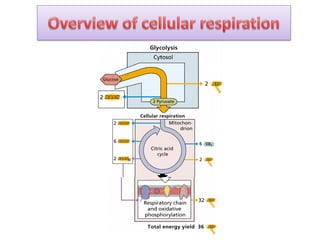
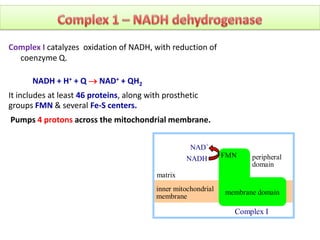
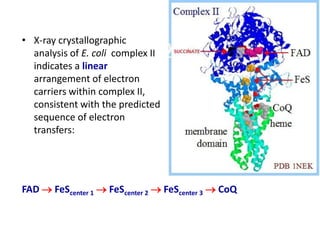
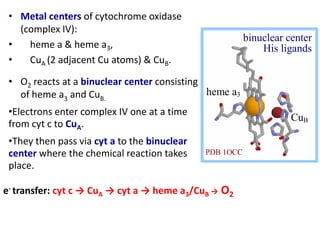
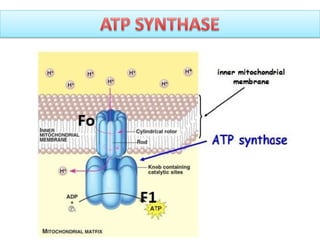
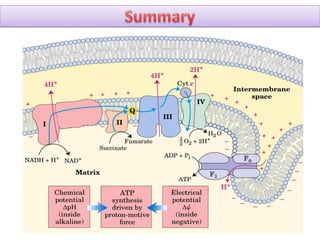
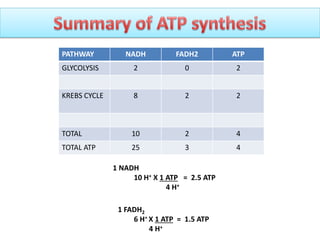
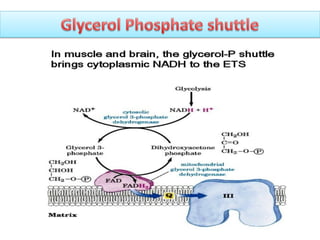
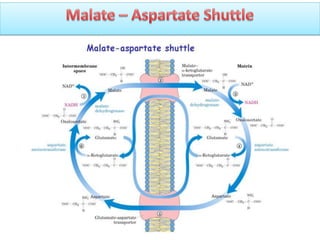
ETC is the transfer of electrons from NADH and FADH2 to oxygen via electron carriers. This releases energy to drive ATP synthesis from ADP and Pi. Multiple protein complexes make up the electron transport chain, passing electrons from one complex to the next until reaching oxygen. As electrons are passed, protons are pumped from the mitochondrial matrix to the intermembrane space, building up a proton gradient used for ATP production.