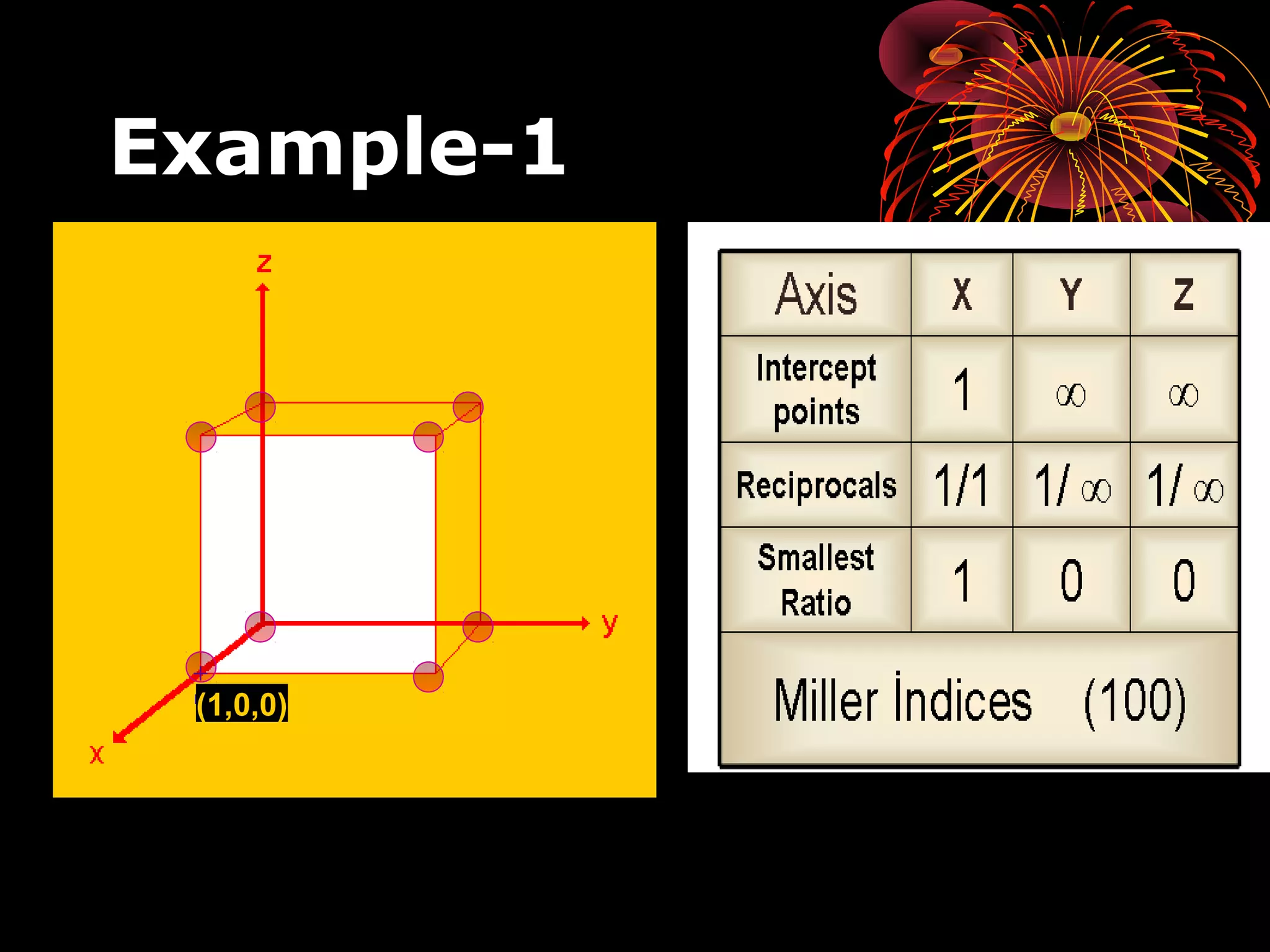
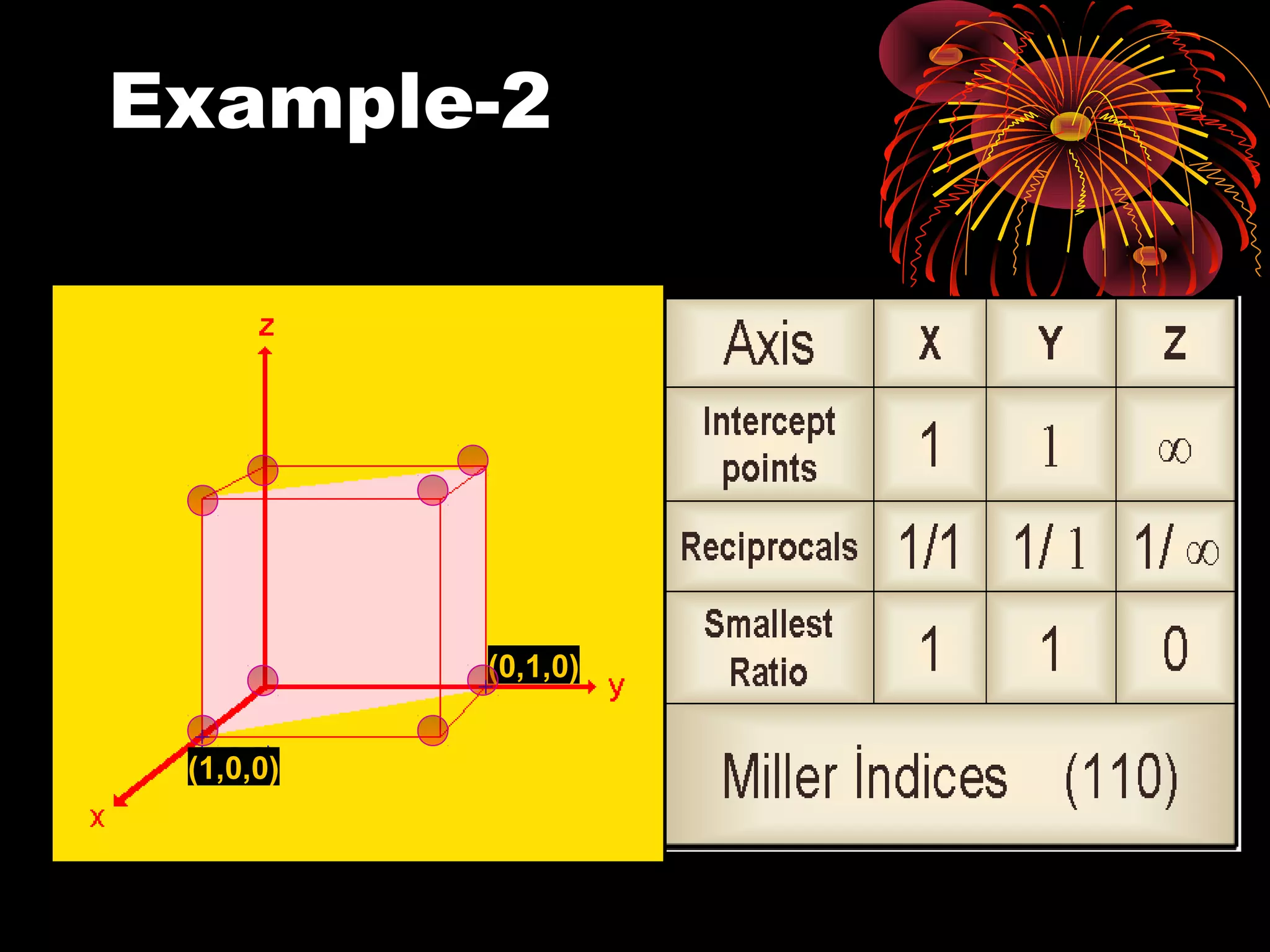
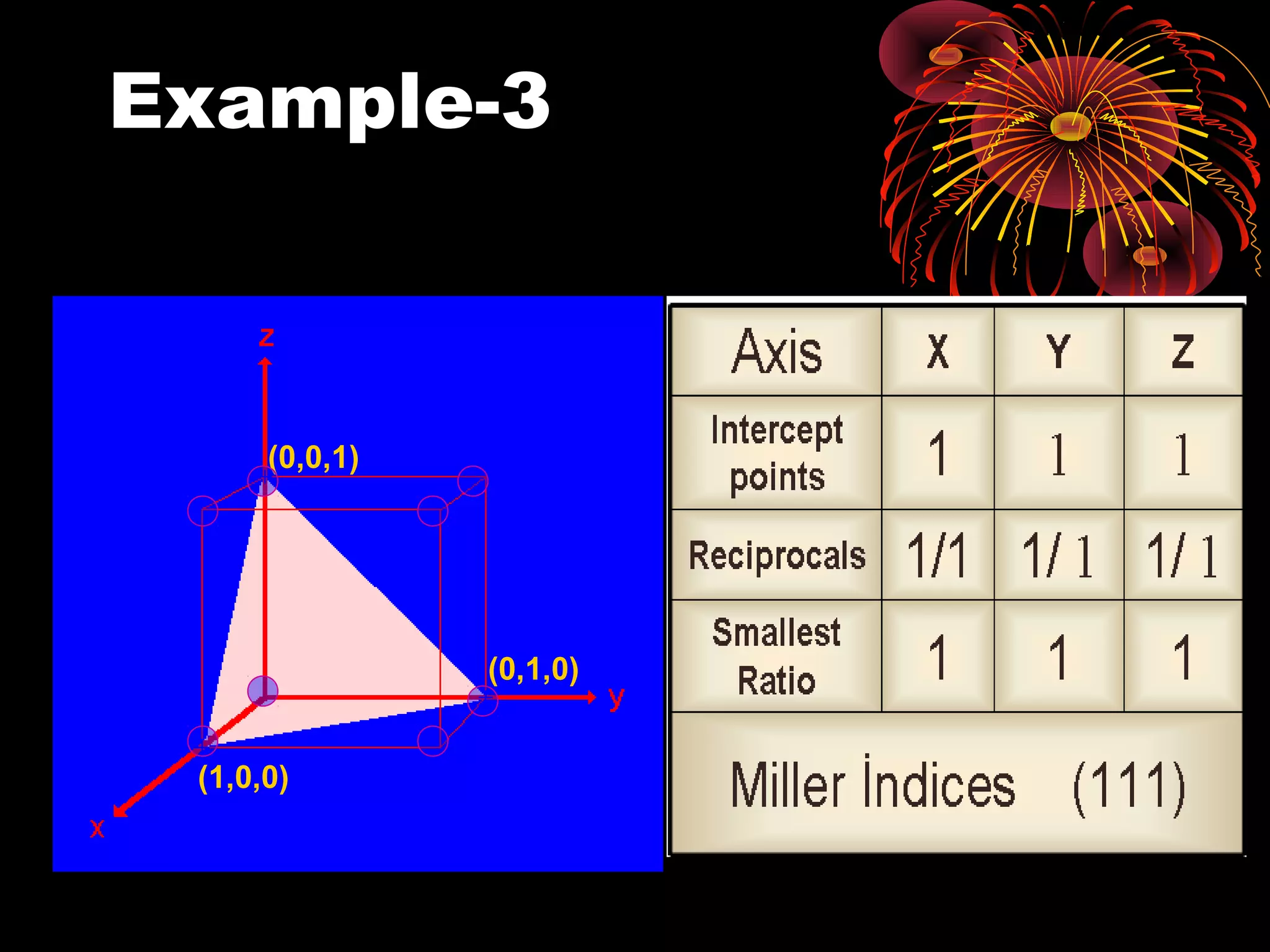
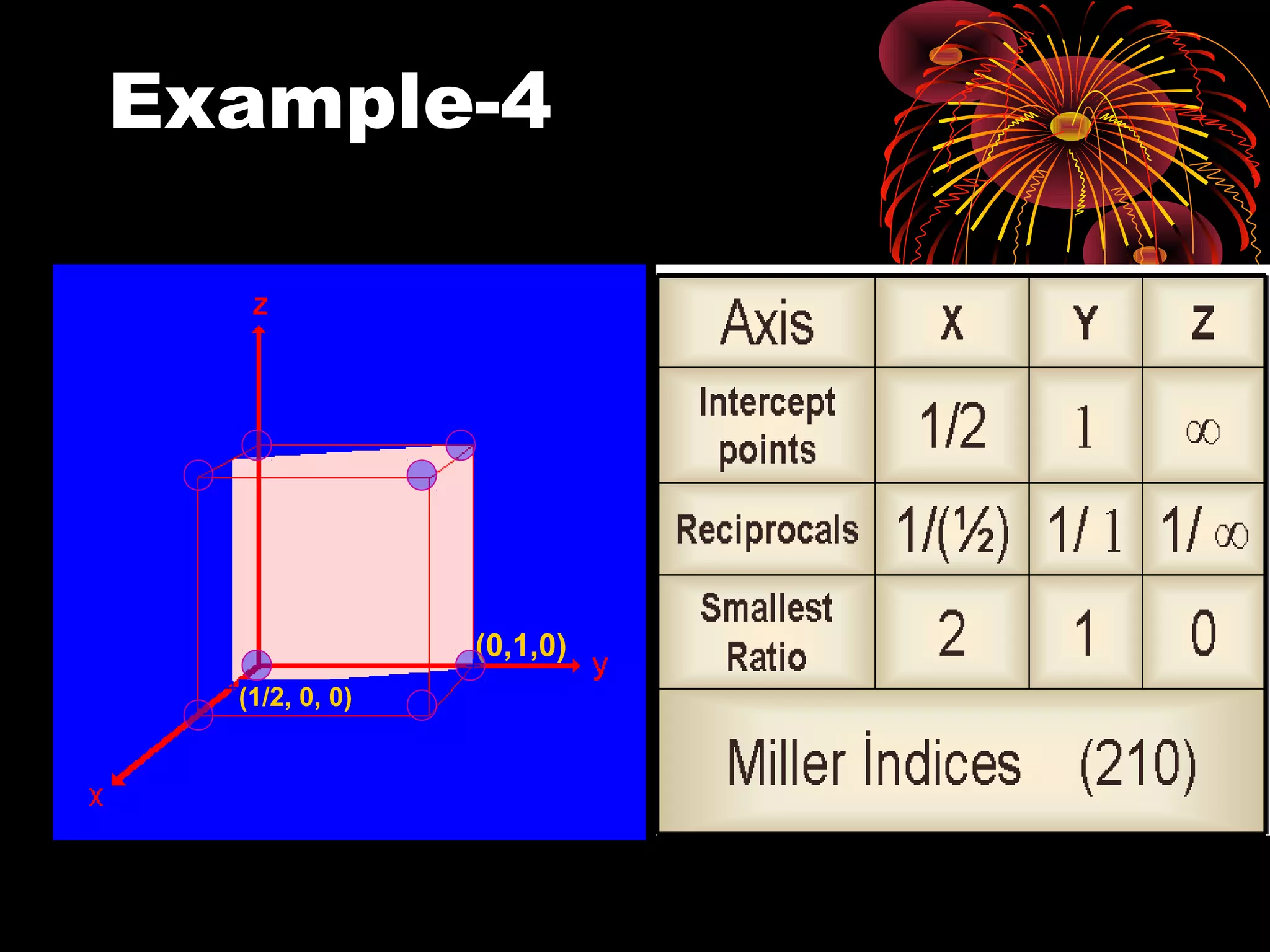
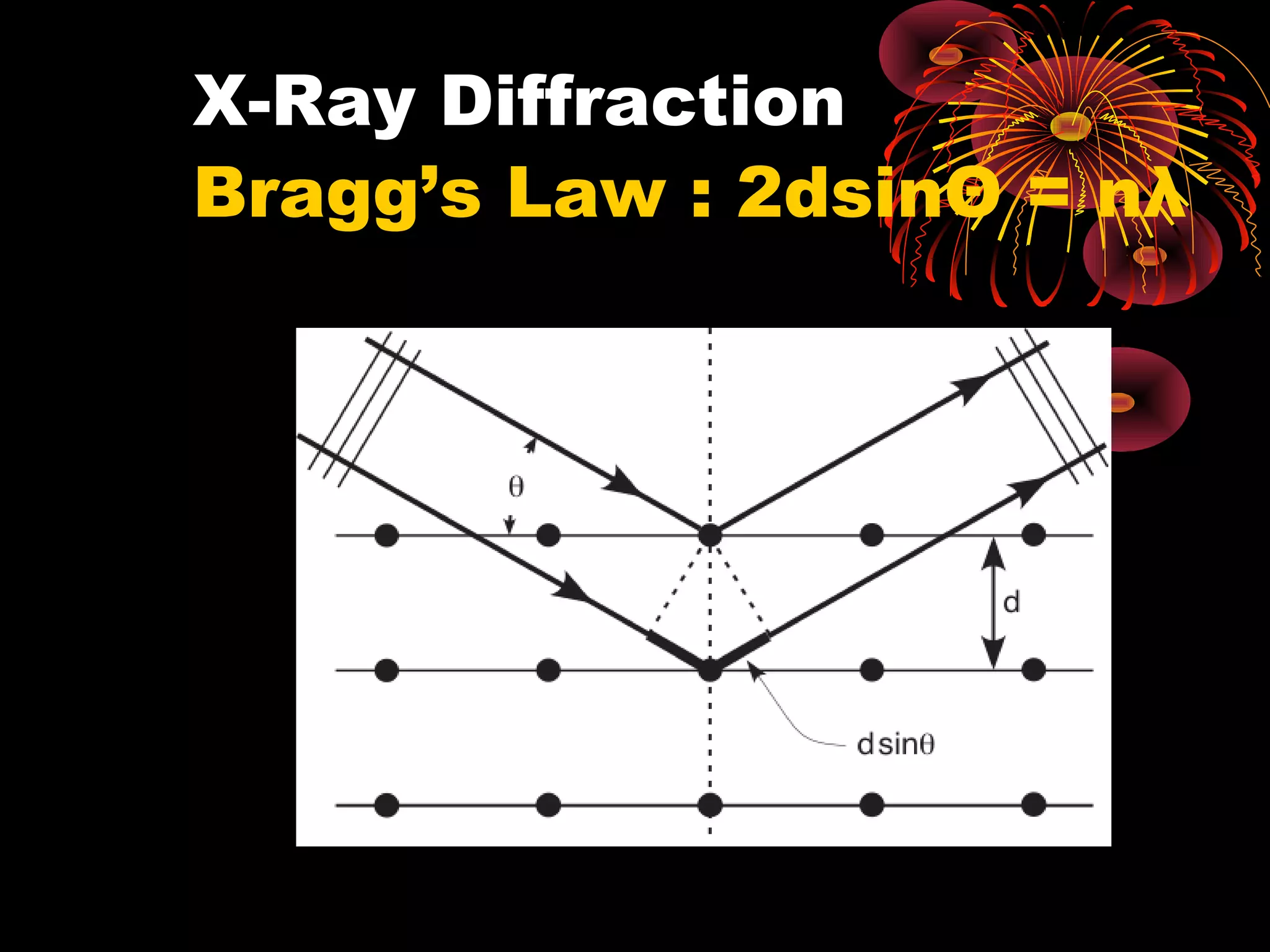
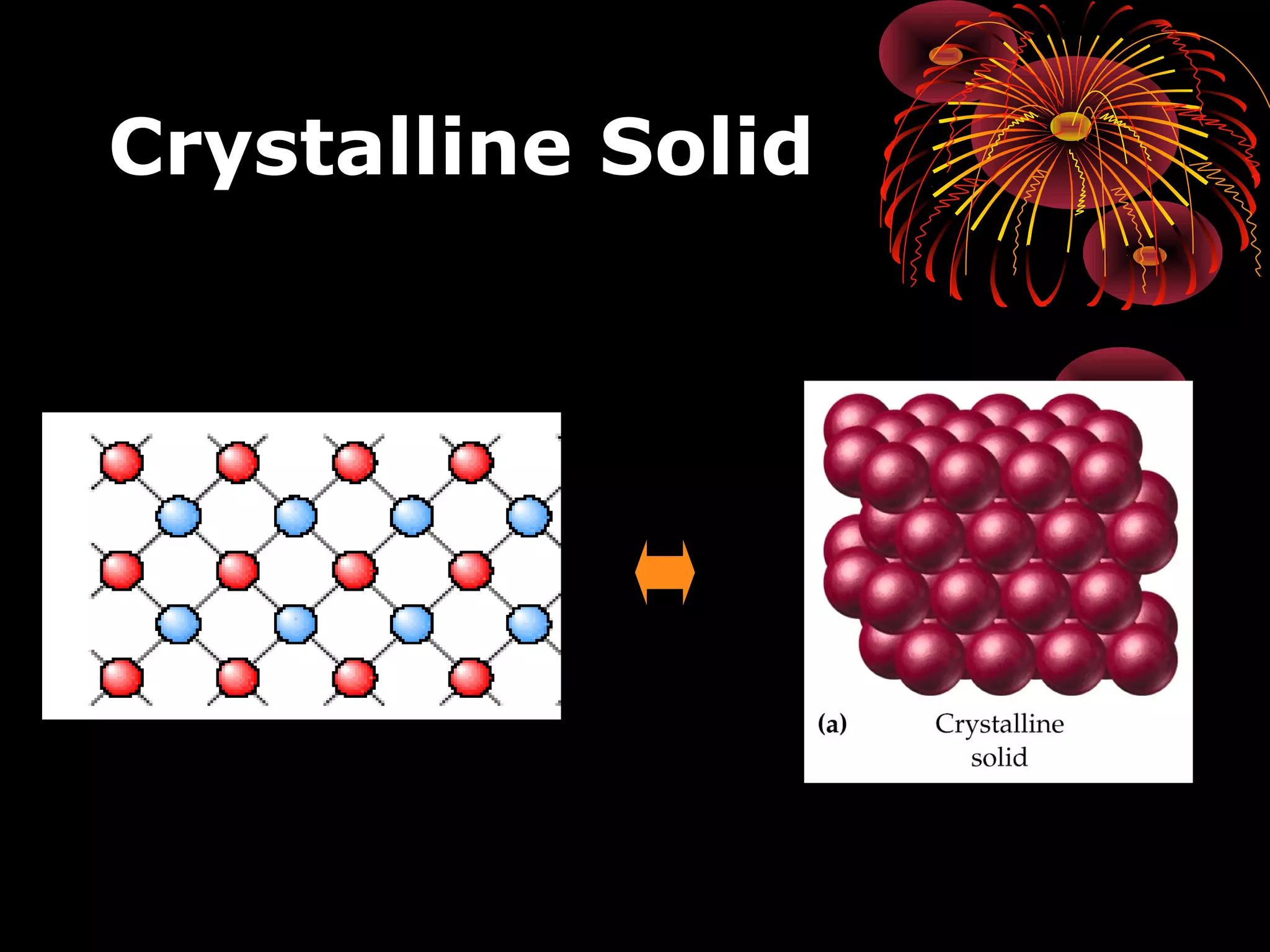
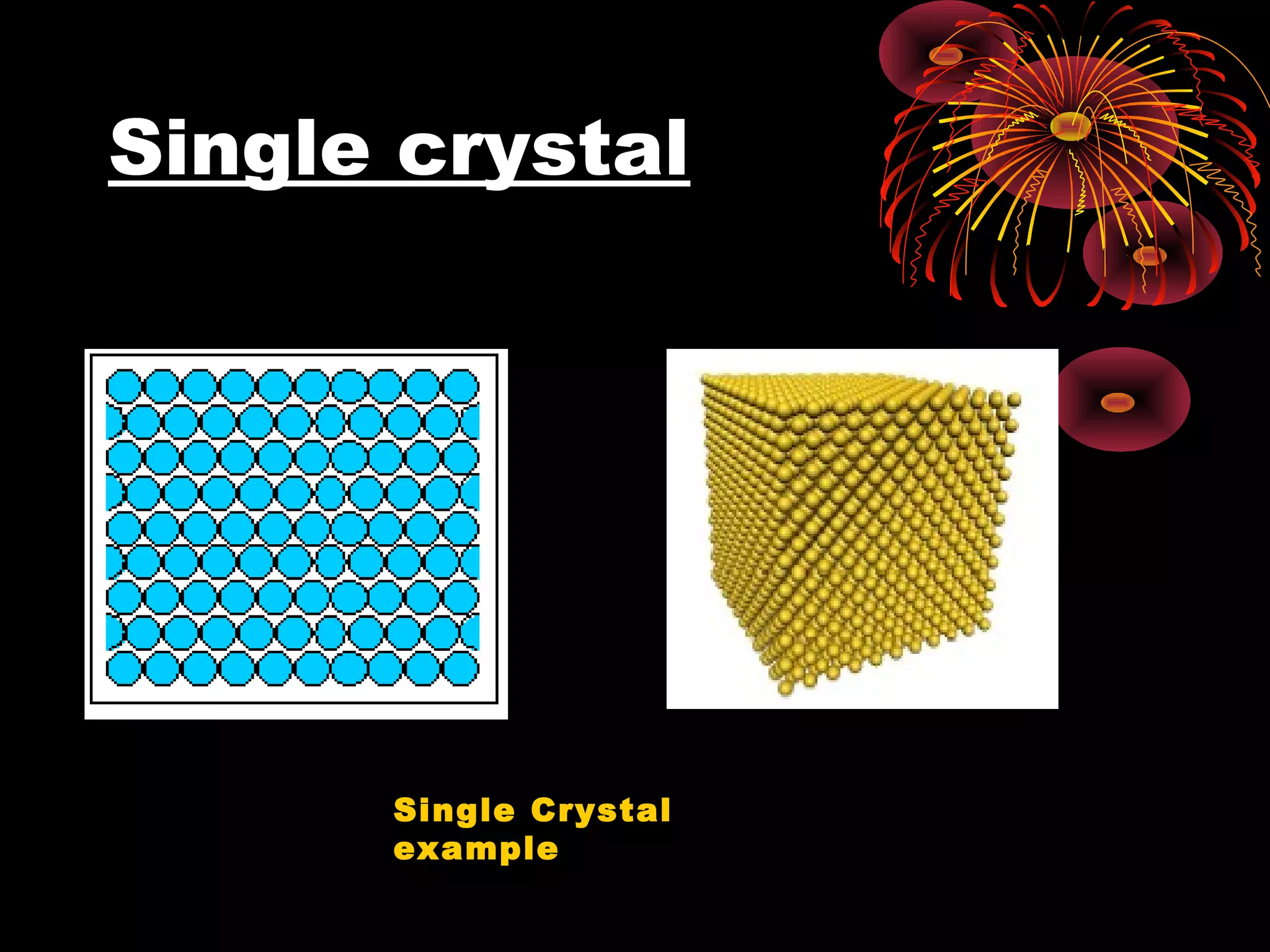
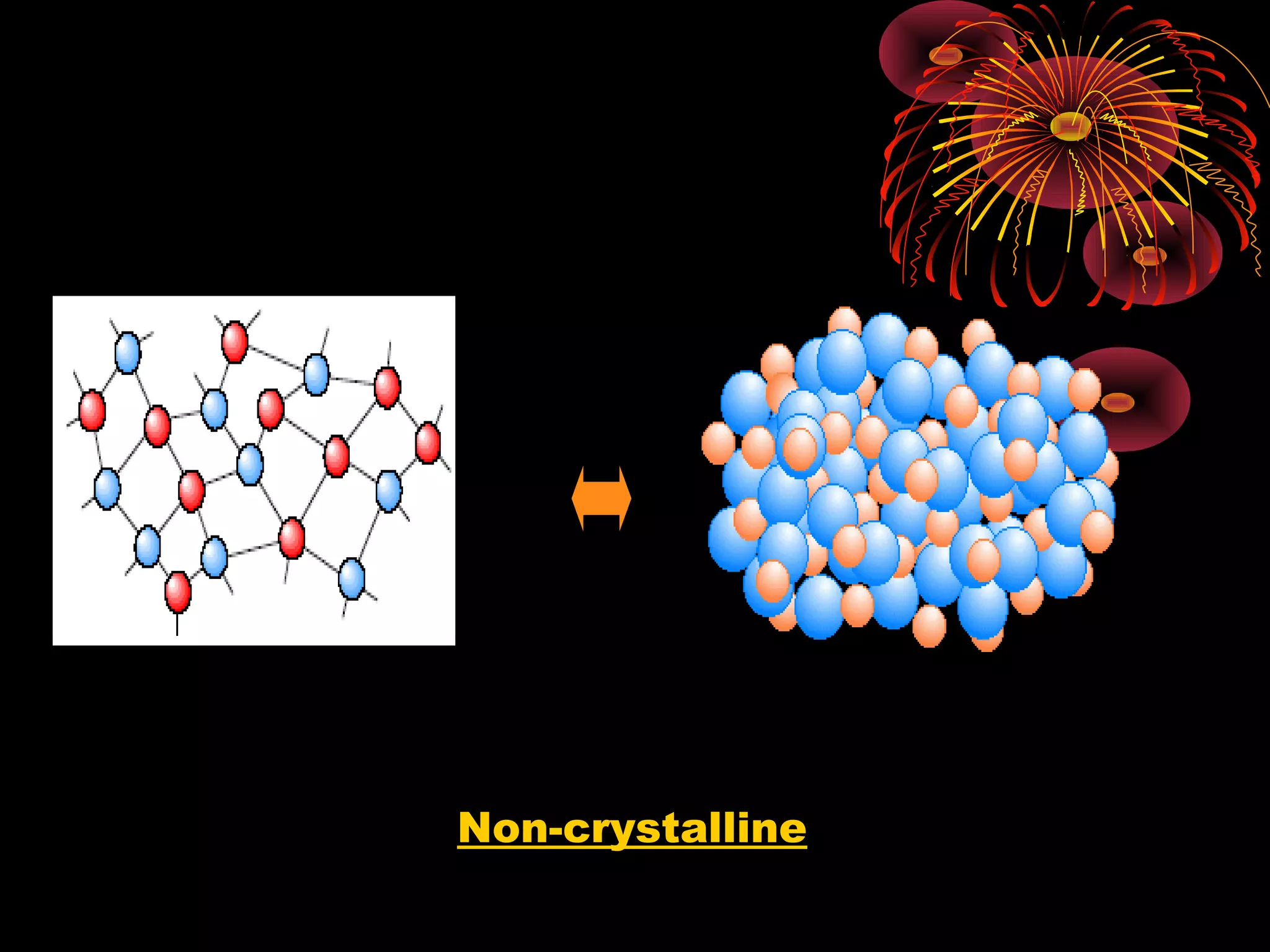
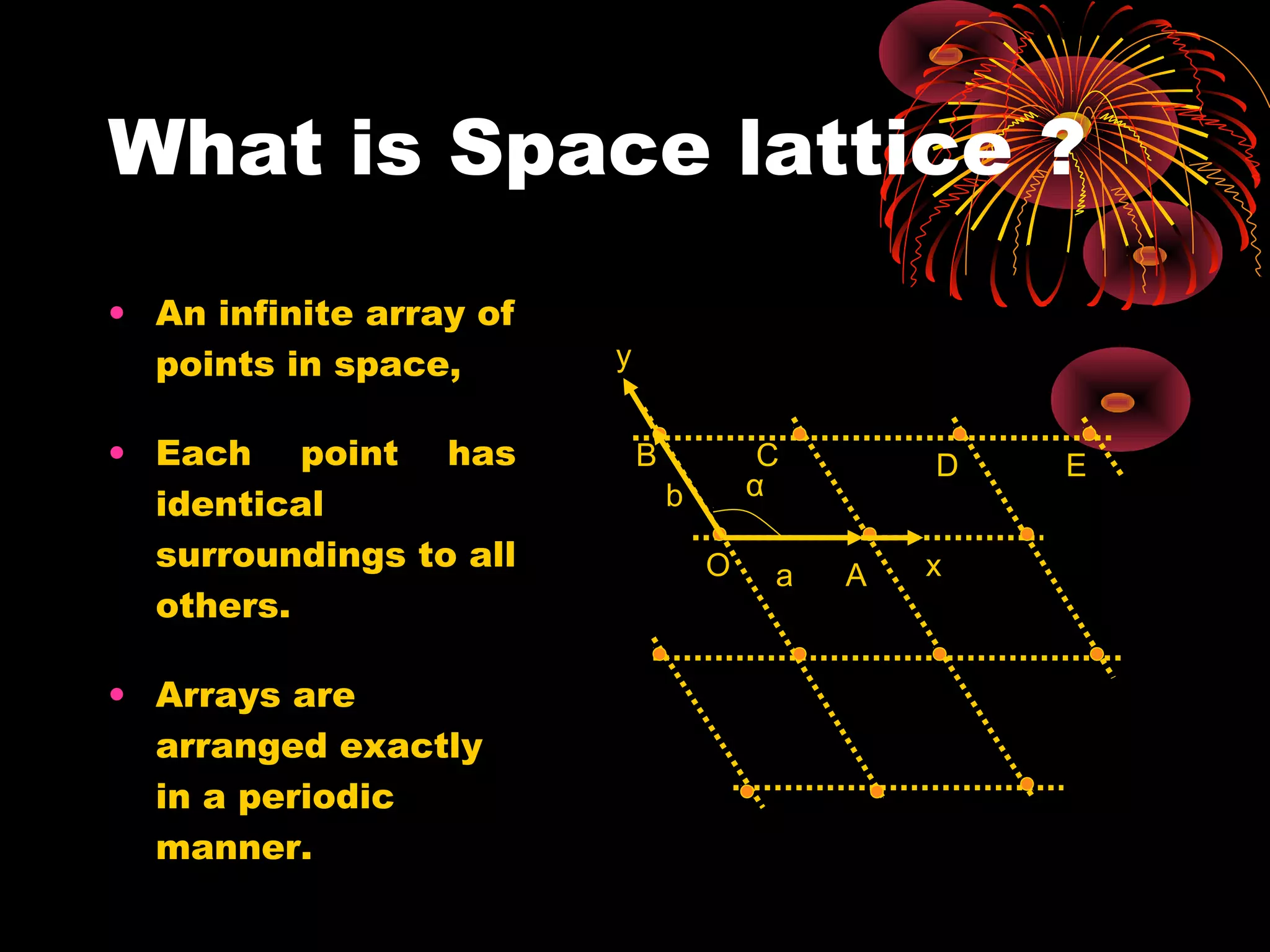
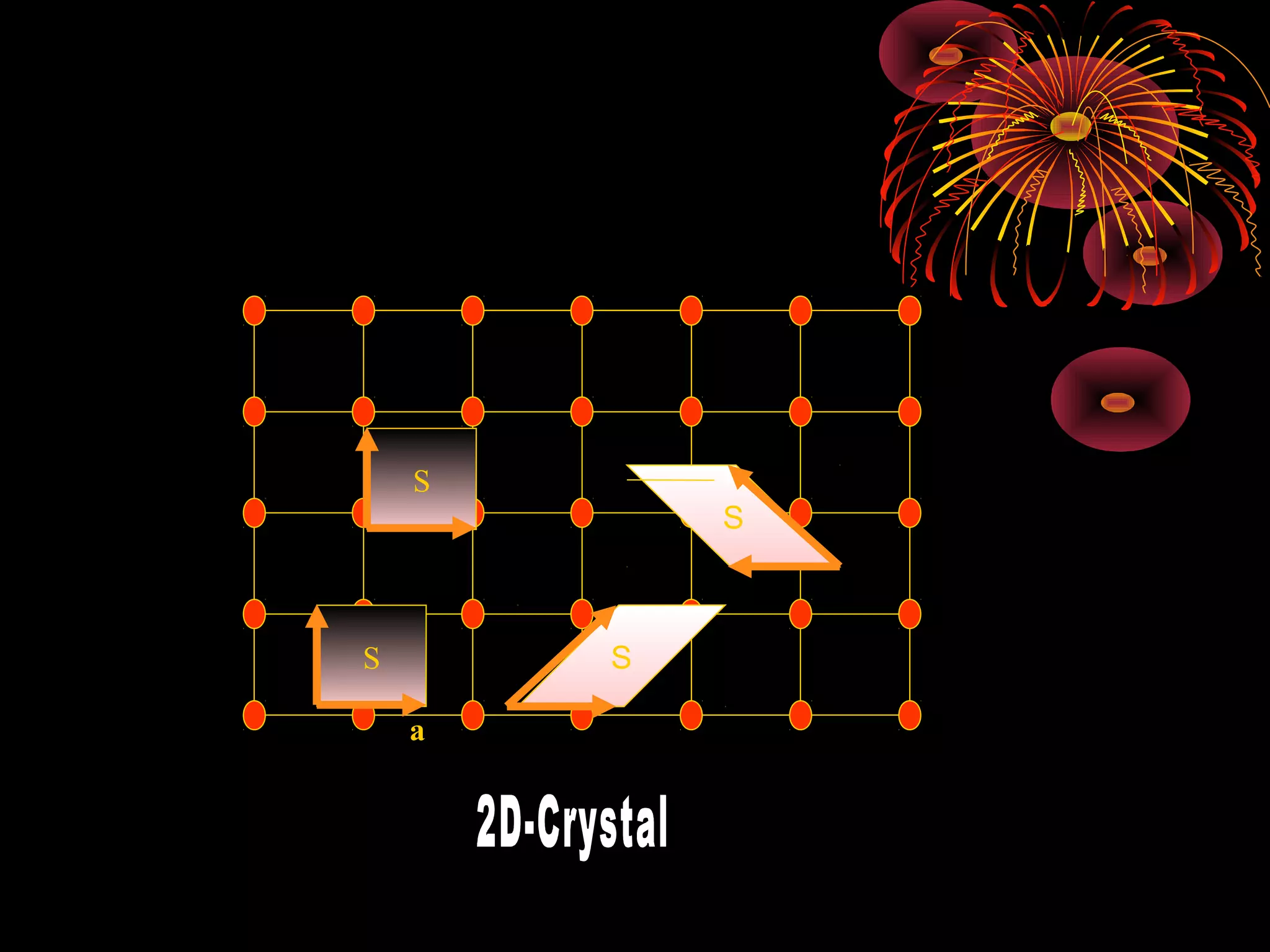
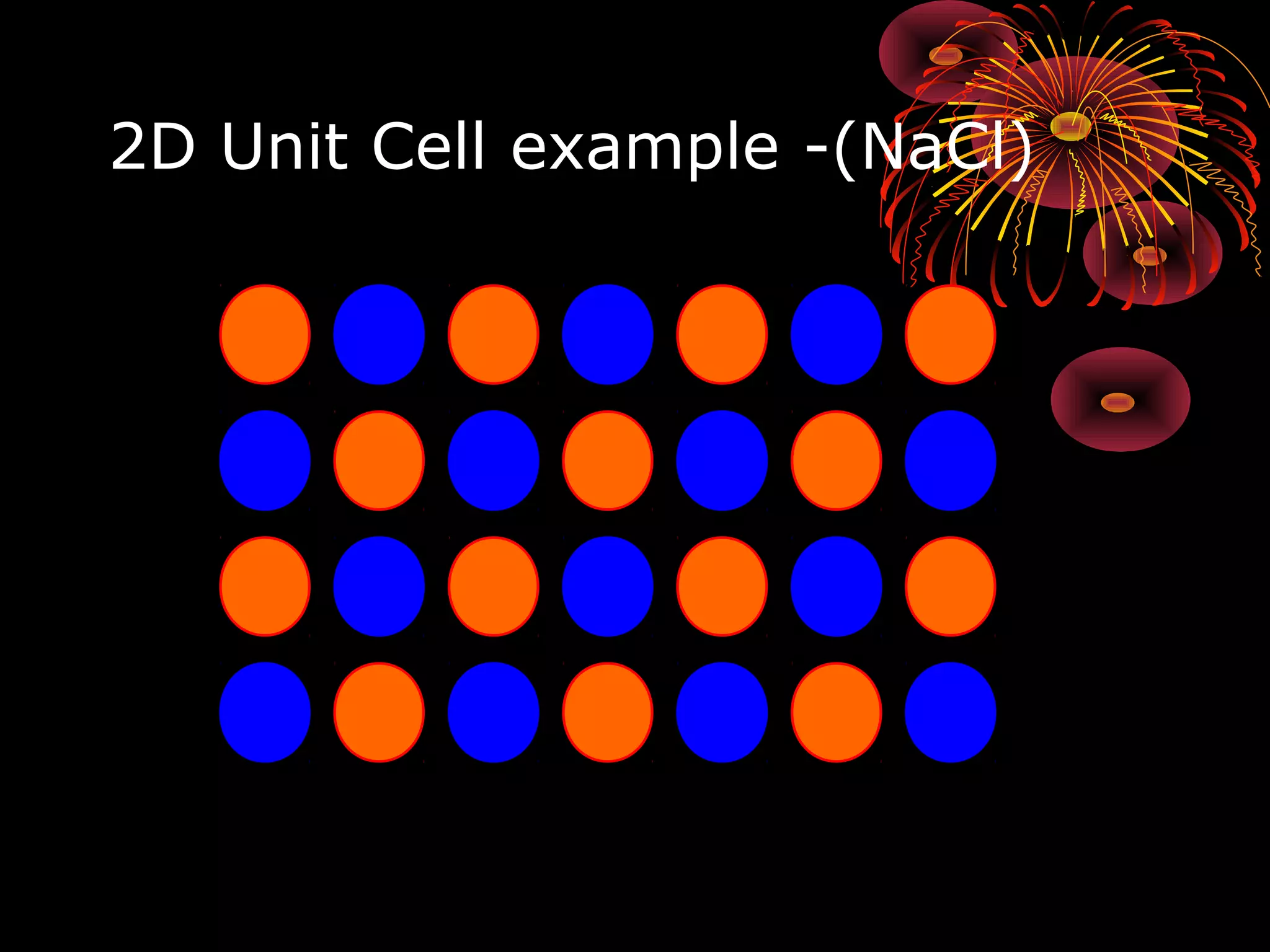
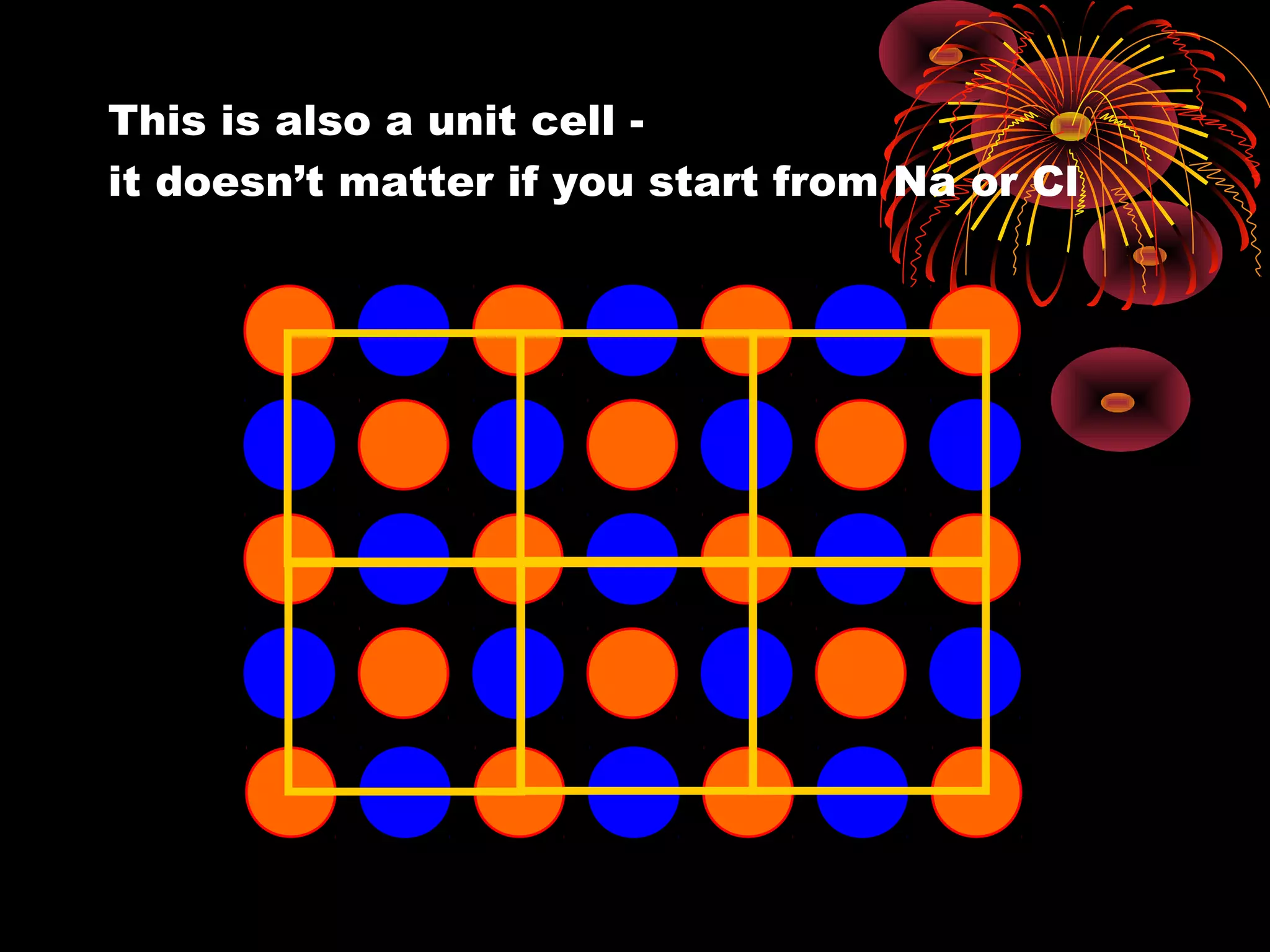
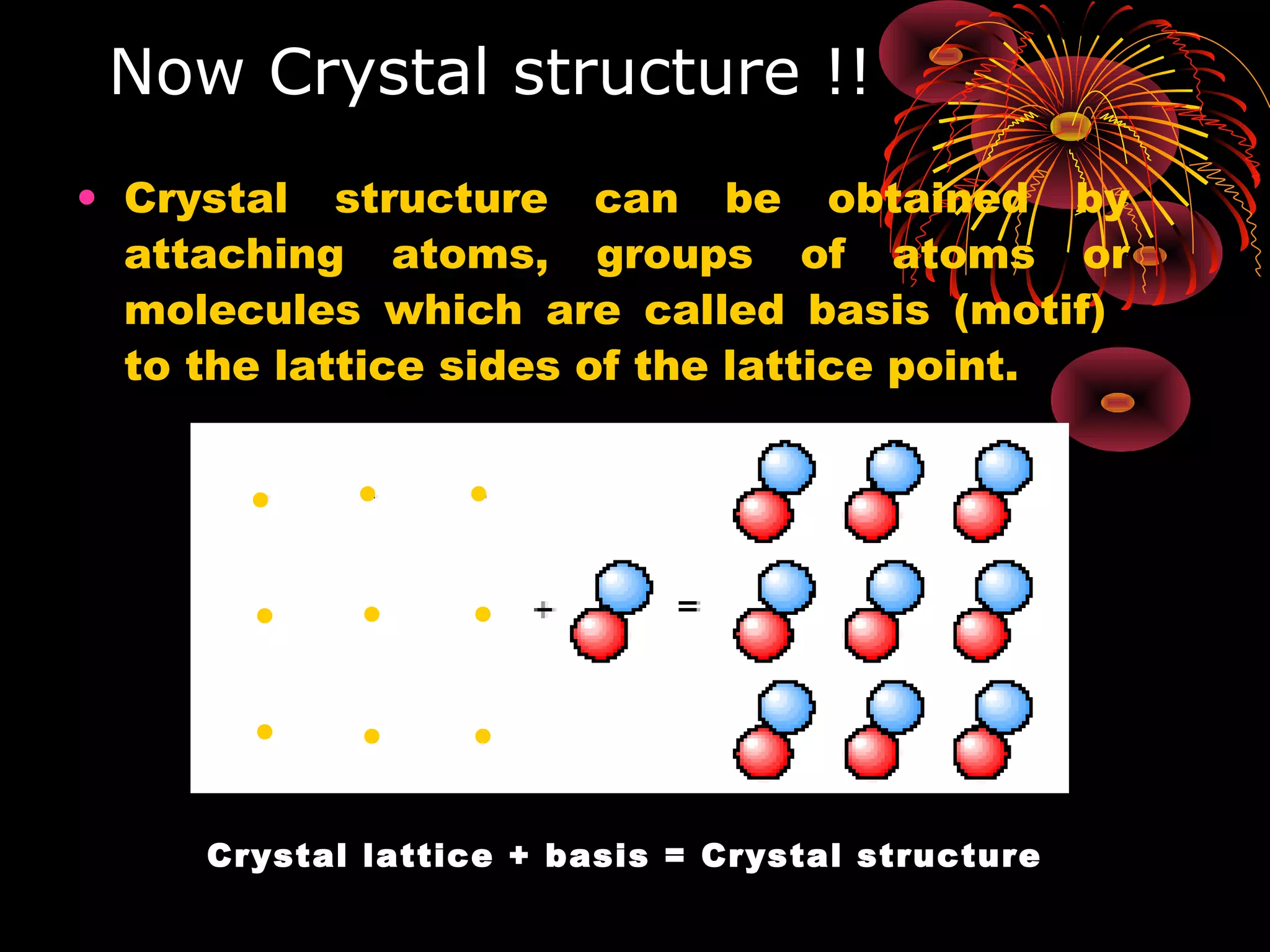
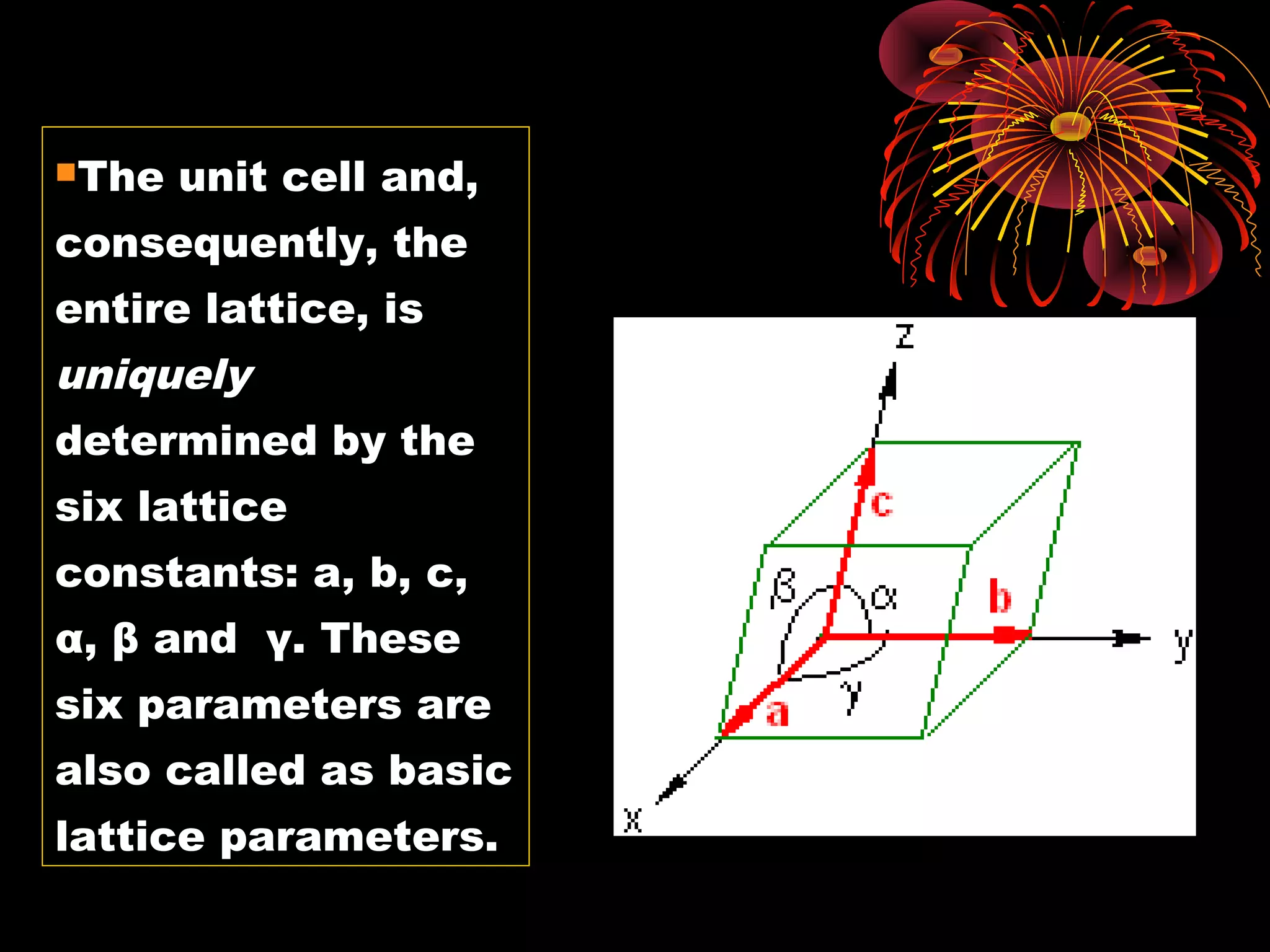
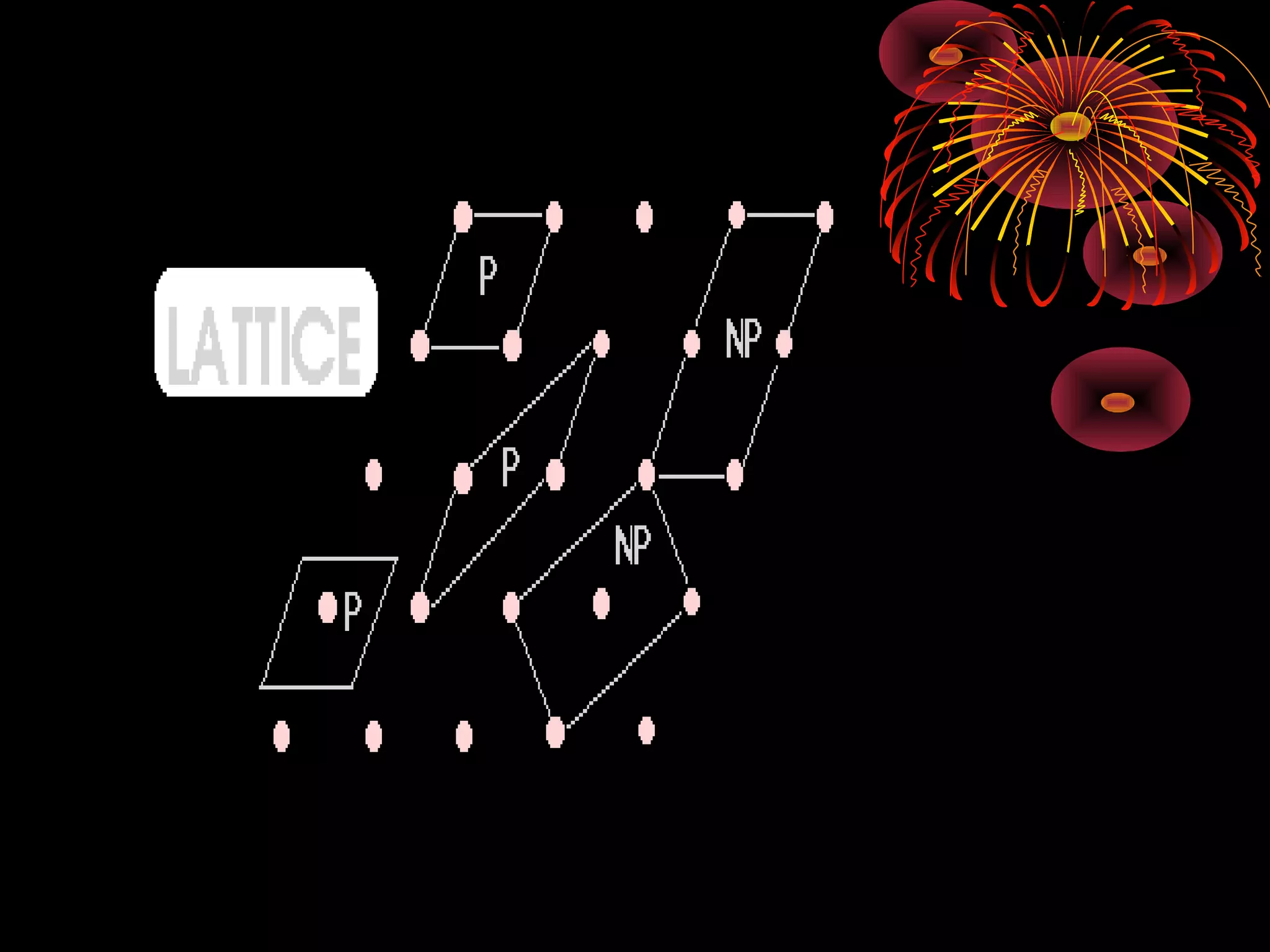
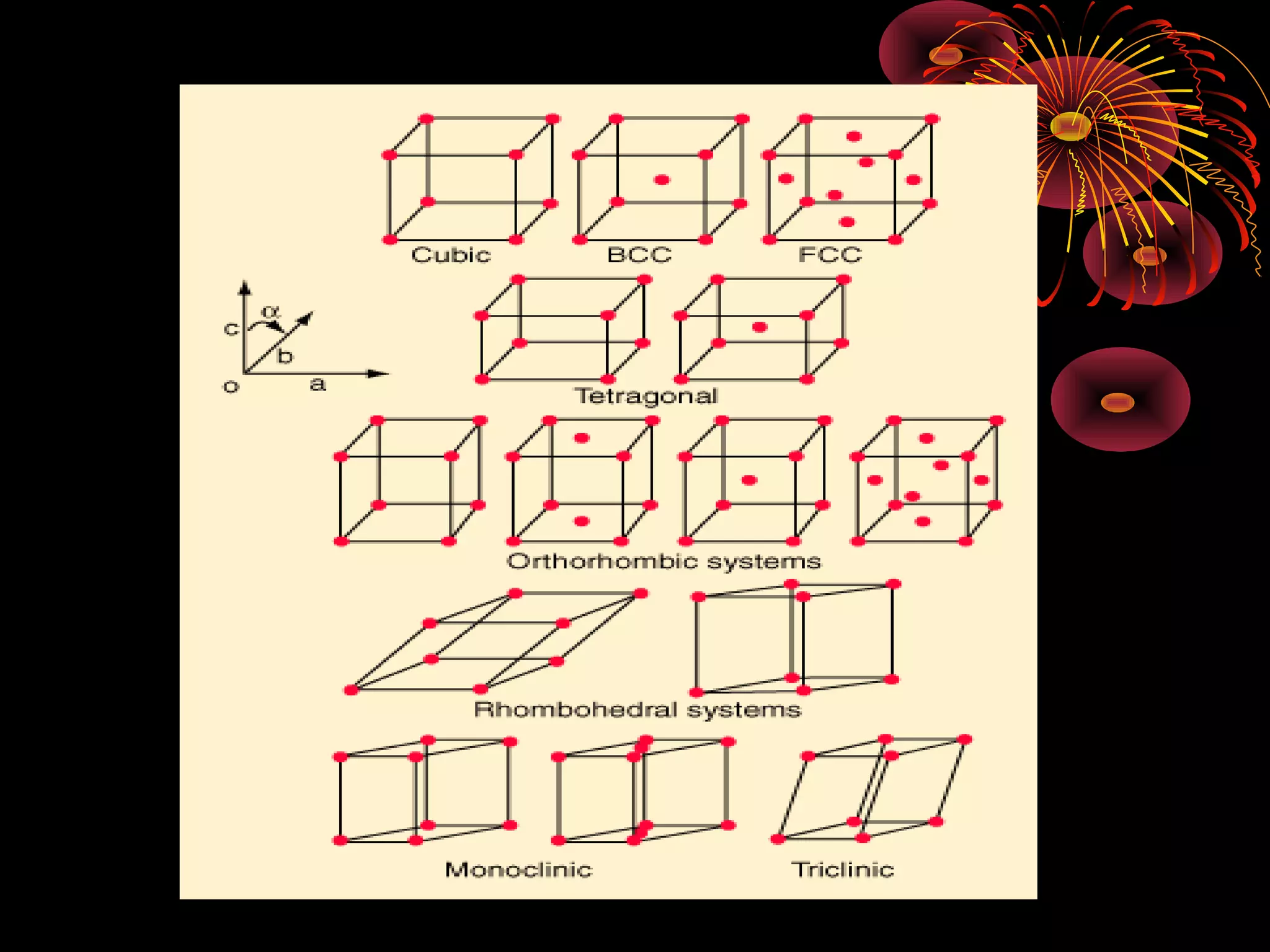
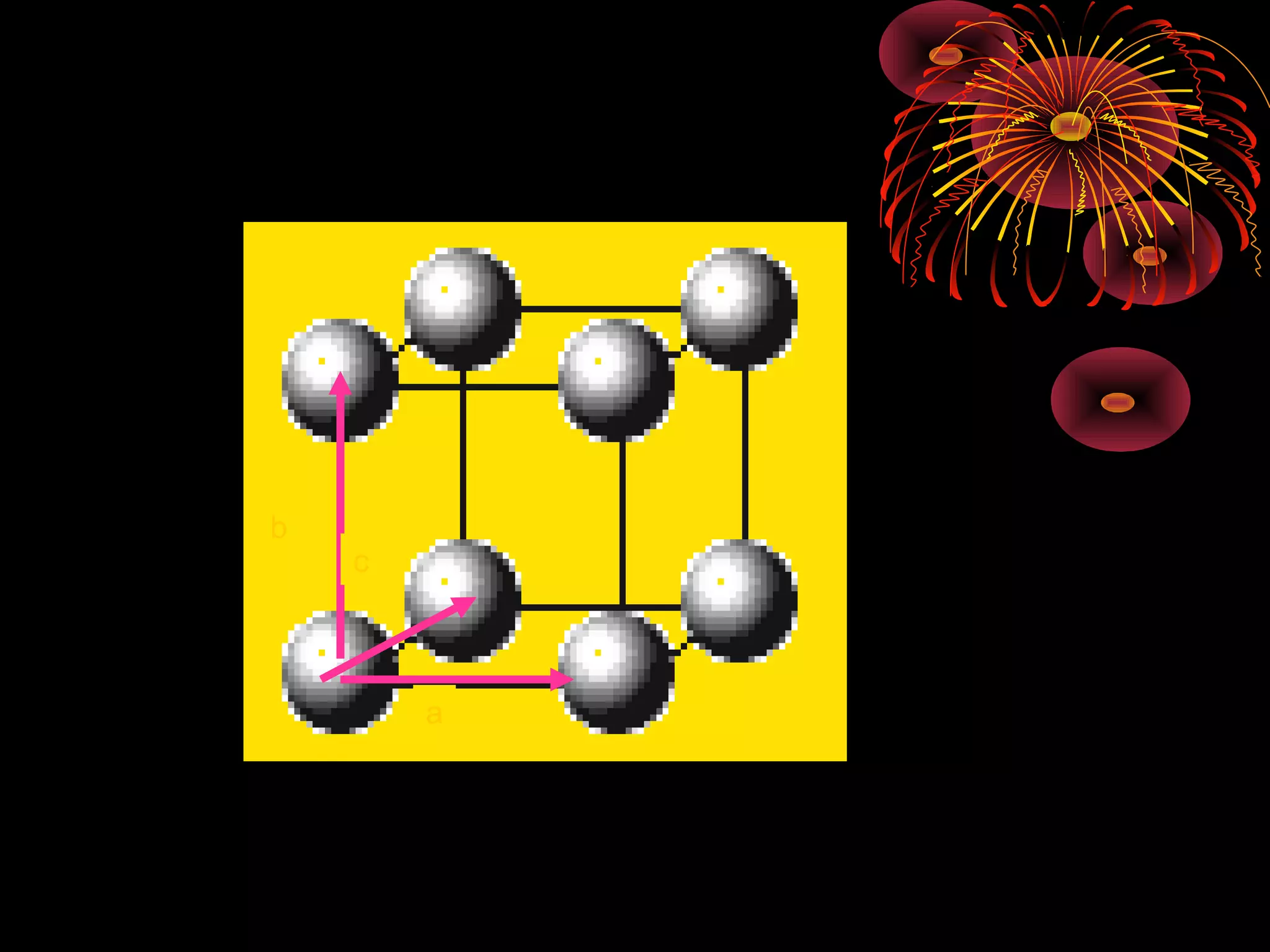
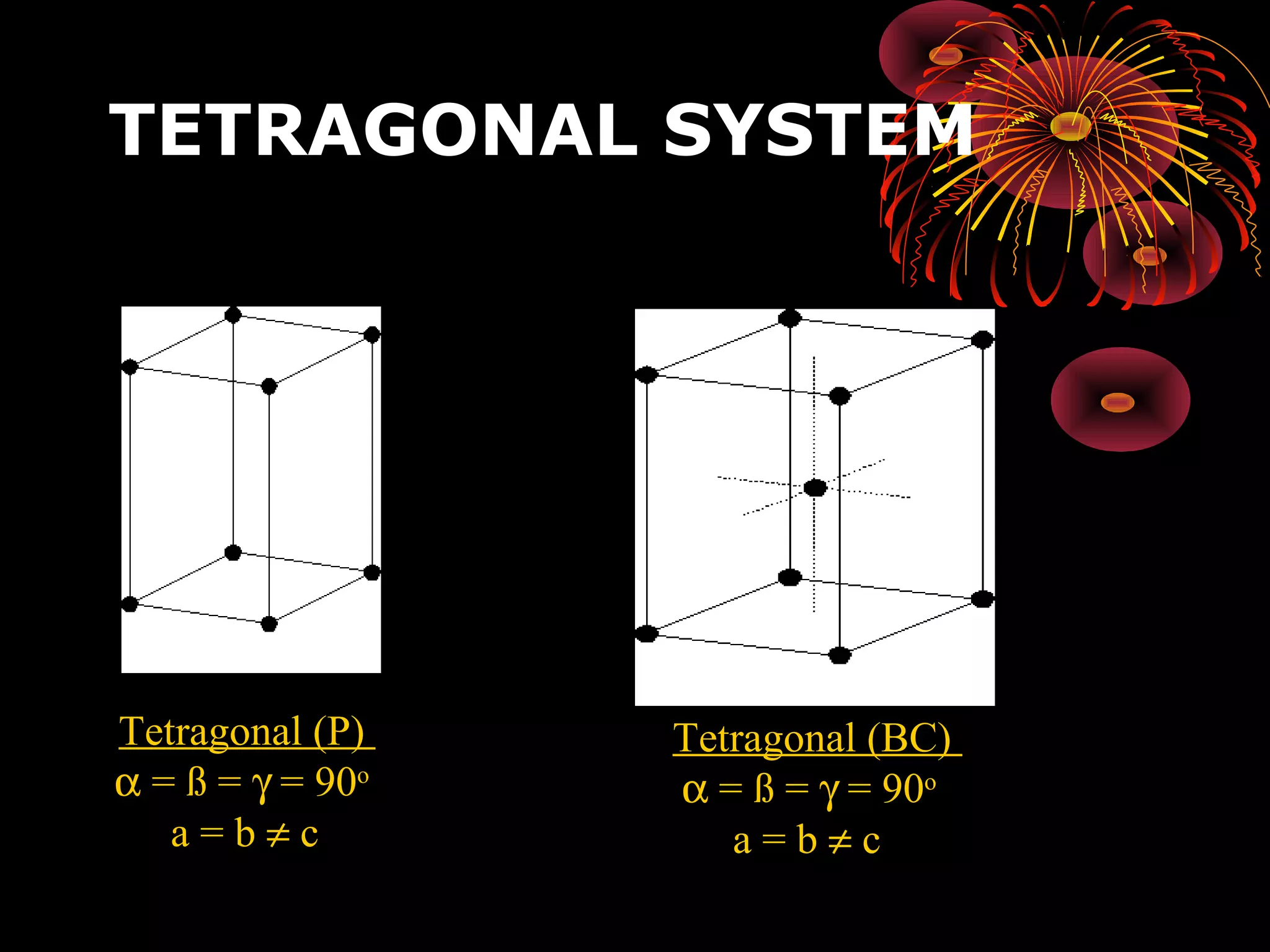
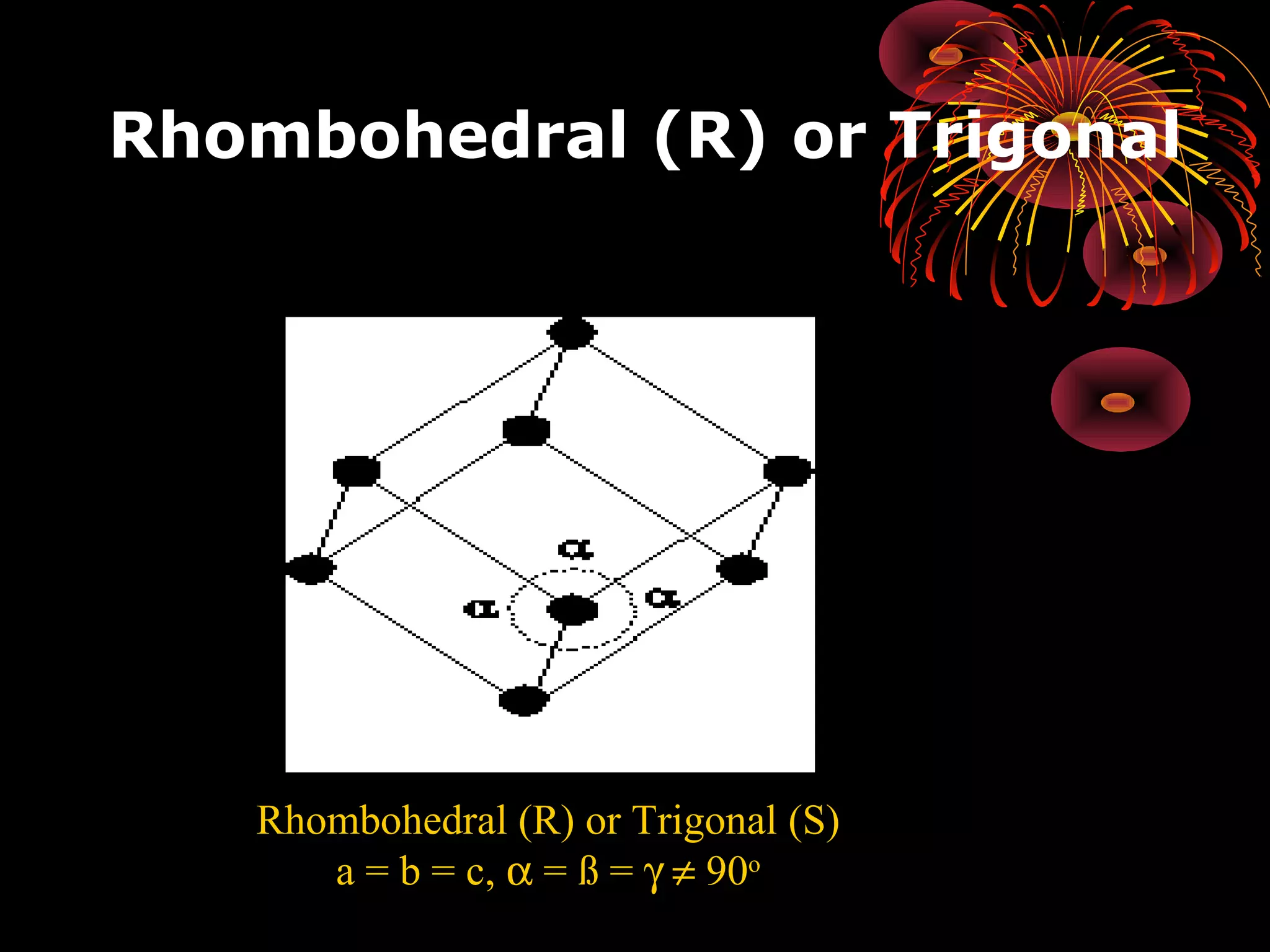
This document discusses crystal structures and x-ray diffraction. It defines crystalline and amorphous solids, unit cells, space lattices, and the seven crystal systems. It also explains Miller indices for identifying crystal planes and Bragg's law for x-ray diffraction, which relates the scattering angle θ, interplanar spacing d, wavelength λ, and order of reflection n. Crystals are characterized by their long-range ordered atomic arrangements, which can be analyzed using techniques like x-ray crystallography.























![Crystal Directions
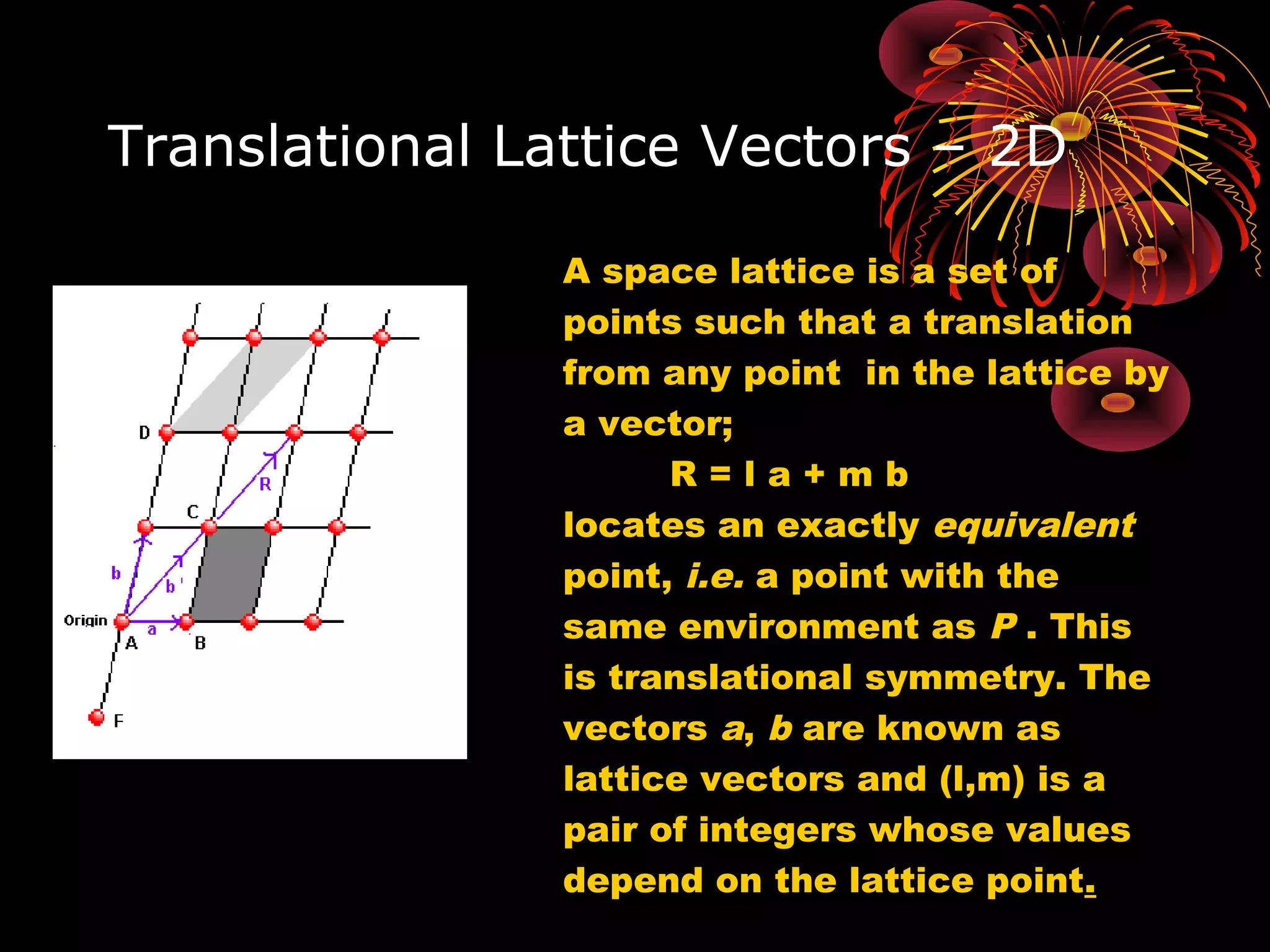
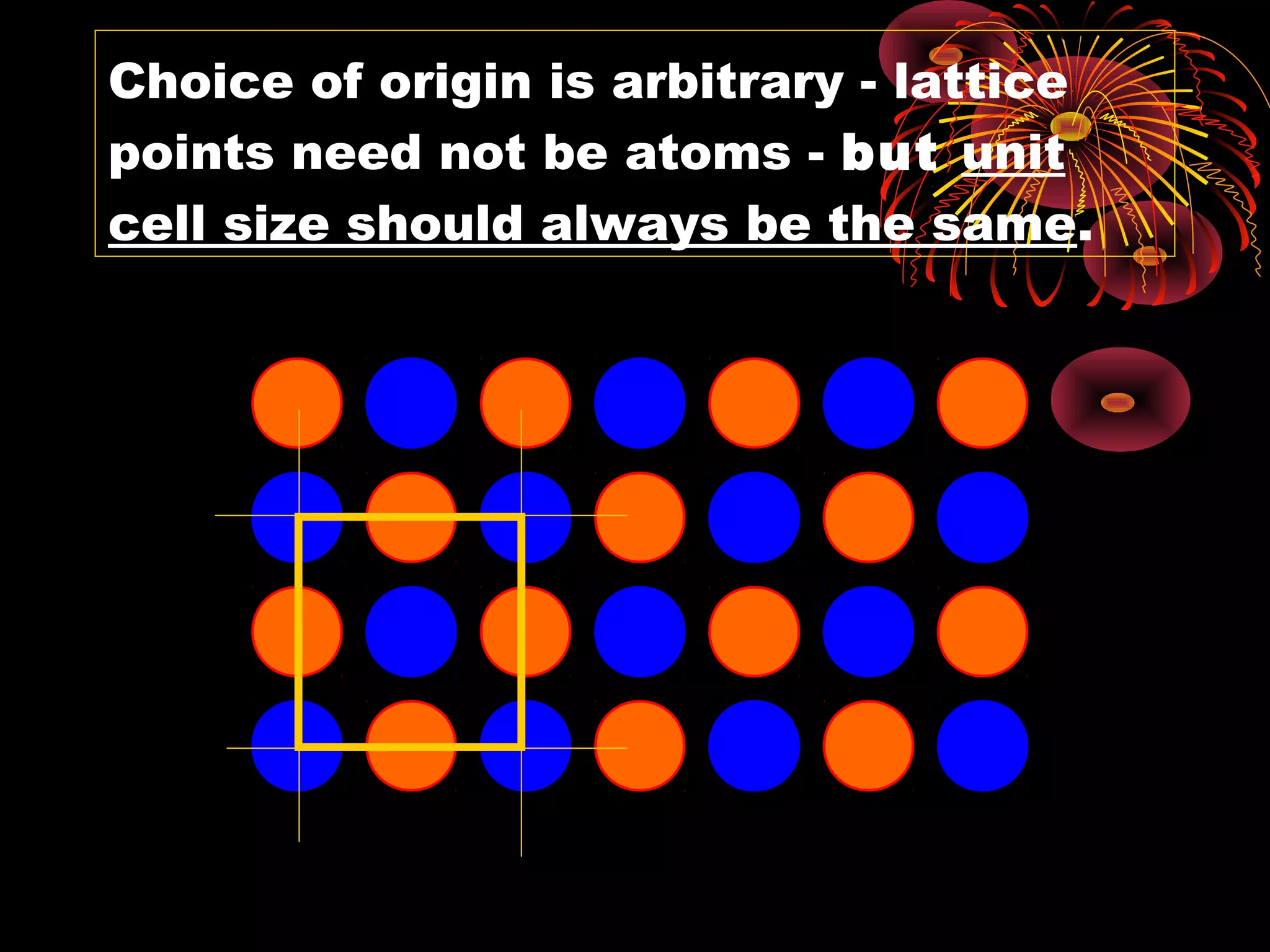
• We choose one lattice point on the line as an origin, say
the point O. Choice of origin is completely arbitrary, since
every lattice point is identical.
• Then we choose the lattice vector joining O to any point on
the line, say point T. This vector can be written as;
R = la + mb + nc
To distinguish a lattice direction from a lattice point, the
triple is enclosed in square brackets [ ... ] is used. [l, m, n]
• [l, m, n] is the smallest integer of the same relative ratios.](https://image.slidesharecdn.com/crystalstructure-130621042556-phpapp02/75/Crystal-structure-49-2048.jpg)
![210
X = 1 , Y = ½ , Z = 0
[1 ½ 0] [2 1 0]](https://image.slidesharecdn.com/crystalstructure-130621042556-phpapp02/75/Crystal-structure-50-2048.jpg)
![Negative directions
• When we write the
direction [n1n2n3]
depend on the origin,
negative directions can
be written as
• R = l a + m b + n c
• Direction must be
smallest integers.](https://image.slidesharecdn.com/crystalstructure-130621042556-phpapp02/75/Crystal-structure-51-2048.jpg)
![Examples of crystal directions
X = 1 , Y = 0 , Z = 0 ► [1 0 0]](https://image.slidesharecdn.com/crystalstructure-130621042556-phpapp02/75/Crystal-structure-52-2048.jpg)



![IMPORTANT HINTS:
• When a plane is parallel to any
axis,the intercept of the plane
on that axis is infinity.So,the
Miller index for that axis is Zero
• A bar is put on the Miller index
when the intercept of a plane on
any axis is negative
• The normal drawn to a plane
(h,k,l) gives the direction [h,k,l]](https://image.slidesharecdn.com/crystalstructure-130621042556-phpapp02/75/Crystal-structure-56-2048.jpg)