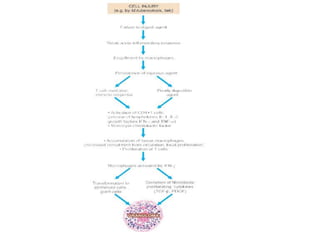
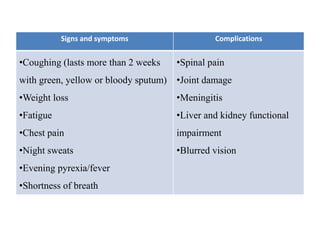
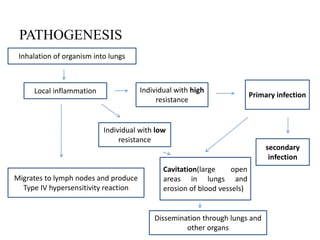
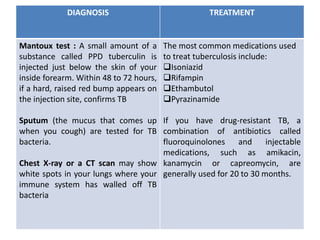
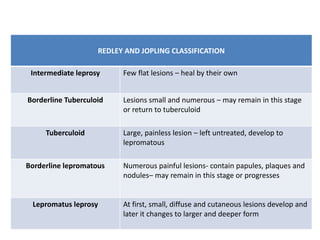
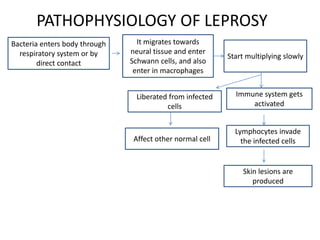
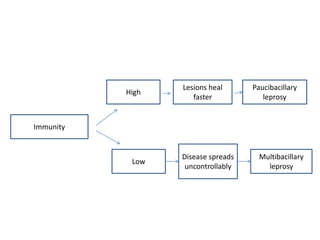
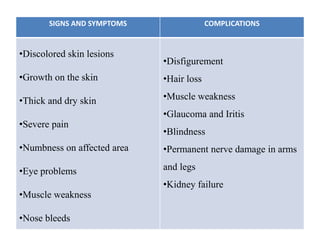
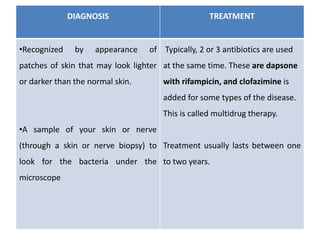
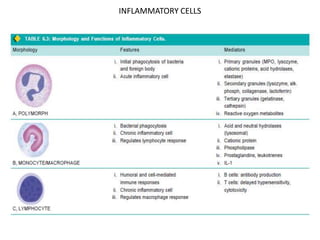
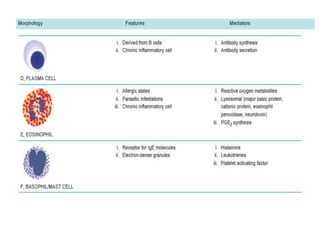
Chronic inflammation is defined as prolonged inflammation and tissue destruction occurring simultaneously. It can result from acute inflammation persisting, recurrent acute attacks, or starting de novo from low pathogenicity organisms. Features include mononuclear cell infiltration, tissue destruction, and proliferative changes like granulation tissue formation and fibrosis. Examples given are tuberculosis, leprosy, and actinomycosis. Granulomas are characteristic of chronic granulomatous inflammation and are composed of epithelioid cells, giant cells, lymphocytes, and may show caseation necrosis.