Systems Approach to Training
•Download as PPTX, PDF•
10 likes•17,655 views
The document outlines the six phases of training system design: analyze, design, develop, implement, evaluate. It provides details on the inputs, processes, and outputs of each phase. Analysis involves collecting data to understand needs and requirements. Design translates analysis into an organizational structure and training blueprint. Development creates training materials and programs. Implementation conducts training and activities outlined in previous phases. Evaluation determines if objectives are met and identifies revisions needed to continuously improve the system.
Report
Share
Report
Share
Recommended
Training Needs Assessment & Analysis

Training Needs Assessment & AnalysisWe Learn - A Continuous Learning Forum from Welingkar's Distance Learning Program.
Management Training requires Assessment and Analysis which is explained in Effective HR. This presentation explains the significance of ‘needs analyses’ in training. Understand various types of training needs and the processes involved in Training Analysis, know the components of a training Needs Assessment and the methods for collecting data.
For more such innovative content on management studies, join WeSchool PGDM-DLP Program: http://bit.ly/SlideShareEffectHR
Join us on Facebook: http://www.facebook.com/welearnindia
Follow us on Twitter: https://twitter.com/WeLearnIndia
Read our latest blog at: http://welearnindia.wordpress.com
Subscribe to our Slideshare Channel: http://www.slideshare.net/welingkarDLPTraining Functions, Training Needs Assessment, Action Research, Organizationa...

Training Functions, Training Needs Assessment, Action
Research, Organizational Objectives and Training
Designing Training Programs

Designing Training ProgramsWe Learn - A Continuous Learning Forum from Welingkar's Distance Learning Program.
Designing Training Programs is important. The fourth chapter of Effective HR discusses the meaning and significance of training design. In this chapter, Training design models and the factors involved for designing a training program is discussed. Understand the motivation dynamics of trainees and the process of training design from this presentation by Welingkar’s Distance Learning Division.
For more such innovative content on management studies, join WeSchool PGDM-DLP Program: http://bit.ly/SlideShareEffectHR
Join us on Facebook: http://www.facebook.com/welearnindia
Follow us on Twitter: https://twitter.com/WeLearnIndia
Read our latest blog at: http://welearnindia.wordpress.com
Subscribe to our Slideshare Channel: http://www.slideshare.net/welingkarDLPEvaluation of training Program

Evaluation is a planned process which provides specific information about a selected session, program for the purpose of determining value or decision making.
Recommended
Training Needs Assessment & Analysis

Training Needs Assessment & AnalysisWe Learn - A Continuous Learning Forum from Welingkar's Distance Learning Program.
Management Training requires Assessment and Analysis which is explained in Effective HR. This presentation explains the significance of ‘needs analyses’ in training. Understand various types of training needs and the processes involved in Training Analysis, know the components of a training Needs Assessment and the methods for collecting data.
For more such innovative content on management studies, join WeSchool PGDM-DLP Program: http://bit.ly/SlideShareEffectHR
Join us on Facebook: http://www.facebook.com/welearnindia
Follow us on Twitter: https://twitter.com/WeLearnIndia
Read our latest blog at: http://welearnindia.wordpress.com
Subscribe to our Slideshare Channel: http://www.slideshare.net/welingkarDLPTraining Functions, Training Needs Assessment, Action Research, Organizationa...

Training Functions, Training Needs Assessment, Action
Research, Organizational Objectives and Training
Designing Training Programs

Designing Training ProgramsWe Learn - A Continuous Learning Forum from Welingkar's Distance Learning Program.
Designing Training Programs is important. The fourth chapter of Effective HR discusses the meaning and significance of training design. In this chapter, Training design models and the factors involved for designing a training program is discussed. Understand the motivation dynamics of trainees and the process of training design from this presentation by Welingkar’s Distance Learning Division.
For more such innovative content on management studies, join WeSchool PGDM-DLP Program: http://bit.ly/SlideShareEffectHR
Join us on Facebook: http://www.facebook.com/welearnindia
Follow us on Twitter: https://twitter.com/WeLearnIndia
Read our latest blog at: http://welearnindia.wordpress.com
Subscribe to our Slideshare Channel: http://www.slideshare.net/welingkarDLPEvaluation of training Program

Evaluation is a planned process which provides specific information about a selected session, program for the purpose of determining value or decision making.
HRD System Design, Assessing HRD Needs, Designing and Implementing HRD Progra...

HRD System Design, Assessing HRD Needs, Designing
and Implementing HRD Programs, Evaluating HRD Program.
Staffing and HRD Function
Training Methods, Techniques & Pedagogy, Training aids & Tools, Facilities fo...

Training Methods, Techniques & Pedagogy, Training aids
& Tools, Facilities for Training
Training Need Analysis Training and Development 

Kannur University MBA slides 3rd Semester, Training Need Analysis, Training and Development Class note for the students
details about the TNA is discussed
Training and its method 

a summary on training and its methods with advantages and disadvantages of every training method
ROLES RESPONSIBILITY AND CHALLENGES OF TRAINNIG MANAGER

ROLES RESPONSIBILITY AND CHALLENGES OF TRAINING MANAGER
Training Need Assessment

" The Difference Between Who You Are and Who You Want to Be, is What You Do..."
Training - Human Resource Management HRM

Training and Developing Employees: Need for Training, Systematic Approach to Training, Types of Training, Training Methods, Evaluation of Training
Training is a planned programme designed to improve performance and bring about measurable changes in knowledge, skills, attitude and social behaviour of employees.
Essential for job success
It can lead to higher production, fewer mistakes, greater job satisfaction and lower turnover
Training Vs. Development, Training Vs. Education, Learning Principles: The Philosophy of Training, Applicability of Training, Job Instruction Training (JIT)
Coaching
Mentoring
Job Rotation
Apprenticeship Training
Committee Assignments
Rao V.S.P “Human Resource Management”, 2nd edition, Pearson –Prentice Hall, New Delhi, 2005
Definition of shrm ( Strategic Human Resource Management)

The role of Strategic HR in overall business.
Training & Development - Designing a training program - key factors, strategi...

This presentation is related to Training and Development which talks about the key factors of designing a training program, the strategies which are faced by the training design managers while designing an effective training program and also the challenges which the design manager has to face on a daily basis while designing the training program.
Hrd & hrm

human resource development and human resource management, and its concept, meaning characteristics and significance in service organizations. it is very useful and resourceful to those who are searching information about the significance of hard and hrm in the service sector.
Training & Development - Systems Approach to Training

This presentation may serve the needs of working managers as well as students of HRM & HRD to some extent.
More Related Content
What's hot
HRD System Design, Assessing HRD Needs, Designing and Implementing HRD Progra...

HRD System Design, Assessing HRD Needs, Designing
and Implementing HRD Programs, Evaluating HRD Program.
Staffing and HRD Function
Training Methods, Techniques & Pedagogy, Training aids & Tools, Facilities fo...

Training Methods, Techniques & Pedagogy, Training aids
& Tools, Facilities for Training
Training Need Analysis Training and Development 

Kannur University MBA slides 3rd Semester, Training Need Analysis, Training and Development Class note for the students
details about the TNA is discussed
Training and its method 

a summary on training and its methods with advantages and disadvantages of every training method
ROLES RESPONSIBILITY AND CHALLENGES OF TRAINNIG MANAGER

ROLES RESPONSIBILITY AND CHALLENGES OF TRAINING MANAGER
Training Need Assessment

" The Difference Between Who You Are and Who You Want to Be, is What You Do..."
Training - Human Resource Management HRM

Training and Developing Employees: Need for Training, Systematic Approach to Training, Types of Training, Training Methods, Evaluation of Training
Training is a planned programme designed to improve performance and bring about measurable changes in knowledge, skills, attitude and social behaviour of employees.
Essential for job success
It can lead to higher production, fewer mistakes, greater job satisfaction and lower turnover
Training Vs. Development, Training Vs. Education, Learning Principles: The Philosophy of Training, Applicability of Training, Job Instruction Training (JIT)
Coaching
Mentoring
Job Rotation
Apprenticeship Training
Committee Assignments
Rao V.S.P “Human Resource Management”, 2nd edition, Pearson –Prentice Hall, New Delhi, 2005
Definition of shrm ( Strategic Human Resource Management)

The role of Strategic HR in overall business.
Training & Development - Designing a training program - key factors, strategi...

This presentation is related to Training and Development which talks about the key factors of designing a training program, the strategies which are faced by the training design managers while designing an effective training program and also the challenges which the design manager has to face on a daily basis while designing the training program.
Hrd & hrm

human resource development and human resource management, and its concept, meaning characteristics and significance in service organizations. it is very useful and resourceful to those who are searching information about the significance of hard and hrm in the service sector.
What's hot (20)
HRD System Design, Assessing HRD Needs, Designing and Implementing HRD Progra...

HRD System Design, Assessing HRD Needs, Designing and Implementing HRD Progra...
Training Methods, Techniques & Pedagogy, Training aids & Tools, Facilities fo...

Training Methods, Techniques & Pedagogy, Training aids & Tools, Facilities fo...
ROLES RESPONSIBILITY AND CHALLENGES OF TRAINNIG MANAGER

ROLES RESPONSIBILITY AND CHALLENGES OF TRAINNIG MANAGER
Definition of shrm ( Strategic Human Resource Management)

Definition of shrm ( Strategic Human Resource Management)
Training & Development - Designing a training program - key factors, strategi...

Training & Development - Designing a training program - key factors, strategi...
Viewers also liked
Training & Development - Systems Approach to Training

This presentation may serve the needs of working managers as well as students of HRM & HRD to some extent.
training & development ppt

Role of Training & Development, identifying training needs, process, Reasons to conduct training, Factors influencing Training & Development
Training and Development - Principles of Human Resource Management

Training and development is required for each and every employee in the organization, in this lesson, we have thrown light on various techniques and tools to improve the skills of employees
Strategic Training and Development

Strategic HR and Training and Development strategies for senior practitioners and those now entering the profession
System approach to training programs development

System approach to training programs development, or Useful correspondence between some popular typologies in terms of
Basic Functionality Matrix
Viewers also liked (20)
Training & Development - Systems Approach to Training

Training & Development - Systems Approach to Training
THE IMPORTANCE EMPLOYEES TRAINING AND DEVELOPMENT IN AN ORGANIZATION 

THE IMPORTANCE EMPLOYEES TRAINING AND DEVELOPMENT IN AN ORGANIZATION
Training and Development - Principles of Human Resource Management

Training and Development - Principles of Human Resource Management
Similar to Systems Approach to Training
CPEC Assessment Pilot JIT Training

The LDC-created just-in-time training for team members before a CAP site visit.
Cmmi process overview

CMMi process overview presentations gives brief idea about process implementation in d design house
Practice Makes Perfect

Within the pharmaceutical industry, there is a constant demand for skilled and qualified professionals at all levels. While getting into the pharmaceutical industry and climbing the corporate ladder is not easy, it can be done. Specifically, pharmaceutical companies and CROs should create career paths that strategically position entry level SAS statistical programmers to junior and then to senior/management level positions. Motivated SAS programmers need to be prepared to meet the growing technical and management challenges.
Training Needs Analysis

Training Needs Analysis (TNA) in one of the essential and basic activity of a HR Manager. Unless scientifically drawn, TNA won't bring fruits of productivity and performance improvement.
IT 8076 Software Testing Unit1

Computer science and Engineering , Professional Elective , Vth semester, Anna university Affliated Institution
Business Learning Strategy

Strategic approach to learning and development efforts that aligns with the business and produces measurable results.
Performance management dr. george taylor iii

A performance management presentation given a few years ago
Similar to Systems Approach to Training (20)
More from Arete-Zoe, LLC
Availability of essential medicines in the Czech Republic (2017)

This report analyzes availability of essential medicines as defined in the World Health Organization (WHO) Essential List Medicines (Report of the WHO Expert Committee) in the Czech Republic. The WHO list of essential medicines contains most effective and safe medicines needed to meet the most important needs in health systems, and is frequently used by countries to create their own national lists. Without these drugs, some conditions will not be able to receive optimal therapy. Availability gap represents serious public health concern.
Expert Committee of the World Health Organization selects Essential Medicines Lists in accordance with approved procedures. The Committee evaluates the scientific evidence on the basis of the comparative effectiveness, safety and cost–effectiveness of the medicines. Both lists (adult and pediatric) went through major revisions in 2015, as the Committee considered 77 applications, including 29 treatment regimens for cancer, hepatitis C and tuberculosis (The Selection And Use Of Essential Medicines. Report Of The WHO Expert Committee, 2015).
Local availability is expressed as registration, in the form of total number of licensed products, and number of marketed products, i.e. products that were on the market in Q3 2016. Of the total number of 13,256 individual registrations for essential medicines, only 2,110 (14%) were actively marketed in Q3 2016. Total number of licensed and marketed product equals all strengths, formulations and dosage forms counted separately. The dataset is current as of December 30, 2016. Any revisions to the database made in the period between data download and publication of this report are not considered. Locally available products were compared to the WHO list of essential medicines. The material is presented in graphs and summary tabulations as listed in the table of contents.
Of the 427 essential drugs, 311 are registered in the Czech Republic, 292 were registered and marketed in Q3 2016, 19 were registered but not marketed, and 135 (32%) were unavailable. Most affected classes are antibacterials, antituberculars, antiretrovirals, antiparasitics, and dermatologicals. Essential medicines availability gap overlaps significantly with drugs that are in shortage globally.
The report provides overview of the situation in the Czech Republic. Essential medicines availability gap represents both public health concern and risk of harm to individual patients. Substitute and second line therapies are often less effective, more toxic, or more expensive. Improvisation and the use of less familiar medicines are more likely to lead to medication errors. Mitigation of shortages and creation of shared contingency supplies puts additional strain on understaffed hospitals, in addition to human toll inflicted by social stress. Drug shortages make it impossible to follow evidence-based practice guidelines, and force decisions to prioritize certain group of people over another.
Post-marketing safety surveillance of medical devices and drug-device combina...

ISoP Medical Device SIG Webinar on Post-marketing safety surveillance of medical devices and drug-device combination products
https://isoponline.org/special-interest-groups/medical-devices-group/
PMS is an integral part of a quality management system described in ISO 13485. ISO 13485 references inclusion of applicable regulatory requirements on post market surveillance into the quality management system.
Regulatory requirements are country specific and are continuously evolving. The regulatory processes for devices are significantly different than for drugs. Moreover, the requirements for drug-device combination products are not always clearly articulated.
• In Europe, according to the EU MDR, post-market surveillance shall also allow a comparison to be made between the device and similar products available on the market.
• The first challenge is identifying similar products on the market, that is out of the scope of this webinar. The second challenge is finding relevant information on equivalent and similar products.
• Since EUDAMED does not currently have a post-marketing module, manufacturers have to rely on a large number of national databases. The focus of this webinar is on regulatory requirements in major jurisdictions. There will be another webinar coming soon that will focus on how to obtain the information required to comply with all these requirements.
• With some effort, it is possible to locate information on advisory notices.
• However, adverse events or incidents are not publicly available. This is a major difference from medicinal products.
• In addition, certain AEs are subject to the National Competent Authority Report (NCAR) Exchange. These reports are shared between agencies and can potentially result in FSCA. So even when unable to monitor competitor product adverse event profile, it is important to know about their FSCAs.
Sexual assault (2017)

Sexual assault cases regularly make headlines and can potentially cause serious reputational harm to law enforcement agencies and police departments for mishandling the cases or not pursuing them vigorously enough.
The picture on the left shows the latest developments in a long-term problem of sexual assault on college campuses. In June, Candice Johnson, OCR Acting Assistant Secretary for Civil Rights issued a memo that effectively stalled investigation of civil rights violations including sexual assault on campuses. A month later, Democratic Senators Kirsten Gillibrand from New York State and Claire McCaskill from Missouri urged Secretary of Education Betsy Devos to reverse this decision as unlawful because of failure to protect students under Title IX. Full text of the memo and Title IX, Sec. 1681 Sex are part of your lesson handout.
Similar problem with widespread sexual assault, and especially against minors, is a long-term problem at cruise ships. Because of the nature of cruise ships, there is no immediate response by law enforcement and the ship guards that investigate the matter are the cruise company’s employees therefore often unlikely to be of meaningful help to the victims. Jurisdiction can be federal, state or foreign, depending on the ship’s flag.
Finally, sexual violence in a workplace can be difficult to address because of the unequal relationship between parties and under-reporting. Recently, car company Tesla appeared in the news as a hostile workplace to women.
Mitigating consequences of a drug-facilitated sexual assault .pdf

Mitigating consequences of a drug-facilitated sexual assault
First published: 27 Jan 2017
Revised: 19 Jan 2020
Drug-facilitated sexual assault (DFSA) is not just bad sex. It occurs either without the victim’s consent or with consent that cannot be considered valid due to incapacitation of the victim by alcohol or drugs. While opportunistic DFSA is carried out once the victim has been rendered unconscious by own actions, pro-active DFSA describes situations when the perpetrator spikes the victim’s drinks covertly.
The most frequently used drug in DFSA is alcohol. Other drugs often involved include flunitrazepam (Rohypnol), gamma-hydroxybutyrate (GHB), gamma-butyrolactone (GBL), carisoprodol (Soma) and ketamine. Ecstasy (MDMA) and other benzodiazepines are occasionally used also. These drugs rapidly induce drowsiness, sedation and muscle relaxation. Typical symptom is decreased inhibition. Most of the drugs used for DFSA are odorless and tasteless, with the exception of GBL that has a bitter taste. Memoryy loss is common, most victims have little to no recollection of the previous night.
The most common way of obtaining these drugs is through the darknet. Benzodiazepines, GHB (Xyrem), and ketamine (an anesthetic used in human and veterinary medicine) are often diverted from legitimate medical use for illicit purposes.
In 2012, in U.S. v. Caronia became one of the landmark cases in the promotion of prescription drugs for unapproved (off-label) indications. Physicians who prescribe Xyrem (GHB) have to pass special certification to ensure safe prescription, handling, and storage of the drug (REMS).
Sedative or tranquilizer Flunitrazepam is still legally manufactured in Europe and some countries in Latin America. The drug has been reformulated, so it imparts an easily identifiable blue color to clear beverages and haziness to colored drinks. Drugs obtained from illegal manufacturing sources naturally do not display this effect.
DFSAs are increasingly popular in bars, clubs, and raves, but also fraternities and at college campuses. Mishandling of cases of sexual assault at college campuses has been subject to much criticism.
It is very difficult to estimate the total number of DFSAs. The main reason for failure to report sexual assault is the reluctance of the victims to go to the police. Indications exist that the numbers are on the increase. Because of memory loss associated with these drugs, victims often feel embarrassed or guilty. Additional reasons for not reporting sexual assault include need to avoid further stigmatization, especially when the evidence does not seem to be sufficient to support the claim confidently. Forensic evidence is difficult to obtain and often lost after first urination the morning after. All drugs used for DFSA are metabolized rapidly by the body, rendering them undetectable within 24 to 48 hours after ingestion.
Approach to preparing for a biological attack (2017)

Approach to preparing for a biological attack
June 2017
Hospital risk management series
The debate on critical issues in science, health, and security encompasses many controversies and ethical challenges. The difference between a naturally occurring outbreak and criminal act of bioterrorism is often challenging to establish, and emergencies have to be handled as they come, regardless of the origin of the incident. The post-incident forensic analysis may or may not offer satisfactory answers in regards to attribution, liability, and the responsibility for compensation. The underlying issue for all ethical concerns examined in this work is the balance between individual rights and the needs of public health systems to protect others.
Improving the resilience of vulnerable populations

Vulnerable populations in terms of health care disparities include the economically disadvantaged and uninsured, the elderly, and people with chronic health conditions. Low-education status compounds the problem and leads to poorer outcomes than in people with the same disease but higher educational status. Significant disparities include namely risk factors relating to morbidity and mortality and access to healthcare. In the domain of physical health, the worst affected are people with chronic health conditions such as respiratory diseases and metabolic syndrome, including hyperlipidemia and diabetes, and resulting in heart diseases and hypertension. Vulnerable populations often experience accumulation of problems that are multiplied by poor health, yet the medical and non-medical needs of these populations are still underestimated. A significant number of vulnerable people with at least one chronic condition skip purchasing prescription drugs because of the costs involved. The most relevant risk factors that result in poor access to health care include low income and uninsured status, in combination with a lack of regular care. Chronic conditions such as dyslipidemia may not be particularly apparent now, yet represent a high risk of future disability (“Vulnerable Populations: Who Are They?”, 2006).
Pricing transparency at point of care

Medical innovation, increasing the complexity of care, and the relationships between stakeholders gradually lead to the increase in prices of healthcare for consumers. Lack of transparency affects the cost of premiums as well as out-of-pocket expenses. Policymakers in their considerations need to include more indicators than just insurance coverage that, without other measures, will not curb soaring healthcare expenses. Delayed care is a public health concern because of the risk of disability and under-treatment of otherwise treatable conditions. The presentation of data to non-technical audiences, including decision-makers, has to be understandable to convey the information reliably. Systems modeling techniques should be considered to estimate stakeholder behavior in a dynamic system accurately. Currently, many instances of abuse exist within the system. As an example, chargemaster fees apply to uninsured or out-of-network patients. Hospital fees are, however, tackled by state laws rather than at the federal level. Consumers in health care tend to behave differently than in other industries and often think less about the costs involved. Physicians’ education should include the delivery of cost-conscious care to prevent financial harm to their patients. Transparency of cost is one of the most effective mechanisms that enable patients and providers to make informed choices.
Handling a high-risk HIPAA Breach

Handling a high-risk HIPAA Breach Published April 2017 Part of scenarios for patient privacy crisis management Every hospital encounters patients, who for the reason of their social circumstances, dependent status, personal characteristics, or the nature of their condition, are more vulnerable than the general population. While compliance with HIPAA is indeed important, because of the potential to inflict significant liability on the hospital resulting from compliance failure, it should not be the only consideration when caring for vulnerable patients. Mere compliance with the minimum requirements of HIPAA does not guarantee the safety of vulnerable patients. In the case study scenario, the hospital emergency department in a small town admitted a 15-year-old female with emergency labor. After delivery in the emergency room, the mother and the baby were moved to Obstetrics and Neonate. Despite appropriate care, the infant presented with multiple medical problems, which may or may not be resolved in the future. A nurse, who took care of the young mother, accidentally disclosed the patient’s identity and condition to her young daughter, who spread the news in all high schools in the area by the following day. The 15-year-old managed to hide her pregnancy from her family. To complicate matters, the young mother’s mother and aunt work in the same hospital.
Addressing pediatric medication errors in ED setting utilizing Computerized P...

Pediatric patients who are treated in general acute care hospitals are at increased risk of medication errors. The main reasons are the lack of experience with the special needs of pediatric patients, their lower ability to tolerate medication errors, medication-related problems such as forms and packaging designed primarily for adults and labeling with insufficient information on the dosing of pediatric patients. Medication errors can be reduced significantly by appropriate medication management systems. Computerized Provider Order Entry (CPOE) systems reduce the frequency of medication errors in all stages of the process. IT technology introduces an additional vulnerability in the form of IT-related medication errors. Nurses are the last individuals in the medication management process who can detect and intercept a medication error and prevent incorrect medication orders from reaching and harming their patients. To be able to do so, nurses have to be familiar with the medication management system in their hospital and escalate incorrect orders as appropriate and relevant.
Let's talk causality attribution: Current practices and path forward 

Consistent and reliable causality attribution at the case level is the cornerstone of confident signal detection.
The current practice relies on study investigators to establish causal relationships based on their observations. The Sponsor (Company) can add their assessment based on additional information about the drug. The current industry standard, E2B (R3), accounts for multiple assessment methods and presents the data elements for each drug-event pair evaluated by multiple sources in a matrix.
There are many causality assessment methods used within the industry, some universal, others more specialized. Most commonly used methods include WHO-UMC, Naranjo, Roussel-Uclaf (RUCAM) - to detect drug-associated liver injury, Karch and Lasagna, the French PV Algorithm, Bayesian Adverse Reactions Diagnostic Instrument (BARDI), MacBARDI, and Updated Logistic method. Expert judgment remains the most common method used.
Serious challenges prevent the practical implementation of existing algorithms by the industry. Many of the algorithms cannot be applied rigorously because of missing data. Additionally, an accurate definition of clinical harm is often lacking (e.g., peripheral neuropathy, vasculitis). Brighton Collaboration Case Definitions partly address this component.
Algorithms do not consider medication errors and are not easy to use with interactions, contributory causation, or secondary harms. Information obtained from the reporter is usually insufficient to establish a causal relationship, and follow-up requests for information must be sent, often repeatedly. The result is a very high share of unassessable reports and poor internal consistency of existing assessments.
I suggest modifying the ADE reporting to incorporate components enabling structured causality assessment directly by the reporting physician (postmarket) or investigator (clinical trials). Guiding questions would assist the reporting physician in determining causal relationships and facilitate algorithmic attribution upon submission:
Temporal relationship is a key component of causality assessment. Safety databases routinely calculate latency and last dose latency that feed the algorithm.
Dechallenge and Rechallenge represent key concepts in pharmacovigilance. This information is typically missing from reports. A series of questions regarding Outcome and Response (Action taken with drug) guide the reporting physician through a checklist for all suspect and interacting drugs, reliably and consistently calculating dechallenge/rechallenge for each drug-event pair.
Biological plausibility is a complex component requiring knowledge of the drug and the patient's medical condition.
Finally, it is important to ask the reporting physician about any underlying diseases that could have contributed to the event. A clear answer to this question is an essential component of the causality assessment algorithms.
Clinical evaluation: Supporting medical device product life-cycle. Applicable...

Clinical Evaluation: Applicable Standards
MDR 2017/745 – Article 61
MDR 2017/745 - Annex XIV
MDR 2017/745 - Annex I
MDCG 2020-5, MDCG 2020-6, MDCG 2020-13
MDCG 2020-7, MDCG 2020-8, MDCG 2020-9
MEDDEV 2.7/1 Rev4, MEDDEV 2.12/2 rev2, MEDDEV 2.7/4 Rev. 4
IMDRF MDCE WG/N56 FINAL:2019
IMDRF MDCE WG/N55 FINAL:2019
Biological evaluation – ISO 10993-1, ISO 10993-18
Clinical Investigations – ISO 14155
Information supplied - ISO 15223-1:5/2017
Risk management – ISO 14971:6/2020, ISO/TR 24971:2020
Quality Management System – ISO 13485:ed2:12/2016
Clinical documentation for medical devices 

Clinical documentation for medical devices
Medical Devices Regulation (EU) 2017/745
We prepare EU MDR-compliant clinical documentation for medical device manufacturers for submission to notified bodies and national regulatory authorities.
EU MDR-compliant clinical documentation (English, Czech):
- Clinical evaluation (plan, report)
- Post-Market Clinical Follow-Up, -
- PMCF (plan, report, study design)
- Post-Market Surveillance System (plan, report)
- Clinical investigation design to complement existing evidence
- Biological Evaluation
- Literature review
Consulting
- Strategy how to generate clinical evidence
- Design of PMCF studies and clinical investigations
Additional support:
- Clinical expert for multiple medical specialties
- Risk management specialist
- Technical documentation
Zpracování klinické dokumentace dle EU MDR 2017/745 

Zpracování klinické dokumentace dle EU MDR 2017/745
- Strategie generování klinického důkazu
- Zpracování klinické dokumentace
- Design PMCF studií a zkoušek
- Návrhy aktualizací existující dokumentace
Služby
Poradenství
Strategie generování klinického důkazu
Design PMCF studií a zkoušek
Zpracování klinické dokumentace (ČJ, AJ)
Klinické hodnocení (plán/zpráva)
PMCF, PMS (plán/zpráva), PSUR
Biologické hodnocení
Návrh aktualizace související dokumentace
Stavba týmu dle potřeb zákazníka:
Klinický expert relevantní pro daný lékařský obor
Specialista na management rizika
Laboratoř na testování software, včetně AI/ML
Zpracování ostatních částí technické dokumentace
Klinické hodnocení (Plán, Zpráva)
Protokol literární rešerše
Biologické hodnocení
Post-Market Clinical Follow-Up (PMCF) (Plán, Zpráva)
Post-Market Surveillance (PMS) (Plán, Zpráva)
Periodic Safety Update Report (PSUR)
COVID-19 Vaccines (Pfizer/BioNTech) 

VAERS Explorer: Resource gateway
The Pfizer-BioNTech COVID-19 Vaccine (Tozinameran)
COMIRNATY- covid-19 vaccine, mRNA injection, suspension
Disease summary
Product approval
Vaccine description
Mechanism of action
Indications and usage
Contraindications
Warnings and precautions
Adverse reactions - experience
from clinical trials
Adverse reactions - postmarketing experience
Mandatory reporting
Adverse events of special interest (AESI)
Pregnancy and breastfeeding
Usage in other populations
Completed clinical trials
Ongoing clinical trials
Resources
VAERS Explorer https://www.aretezoe.com/vaers-explorer
Vaccine labels
https://www.aretezoe.com/vaccine-labels
Anthrax vaccine 

Anthrax is a serious infectious disease caused by the bacteria Bacillus anthracis. People or animals can contract anthrax from contact with infected animals or contaminated animal products. Bacillus anthracis forms spores than can survive in the environment, especially soil or animal products (e.g., rawhide) for decades. The most common route of exposure is via skin scrapes when working with infected animals resulting in cutaneous anthrax. Gastrointestinal infection occurs following eating raw or undercooked infected or contaminated meat. The most dangerous form of anthrax follows after inhalation of aerosolized anthrax spores, typically during industrial processing of infected animal products (e.g., rawhide, wool). In the United States, anthrax is very rare. Vaccination of livestock is recommended in areas with historical occurrences of anthrax. Moreover, all food animals are examined before slaughter (Mayo Clinic, Guide to Understanding Anthrax, ACIP).
Anthrax spores had been mass-produced as a bioweapon by the Soviet Union (STAT News). In 2001, anthrax was also used as a bioweapon when letters laced with anthrax were mailed to several news media offices and Democratic Senators Tom Daschle and Patrick Leahy, killing five and sickening 17 (Amerithrax investigation). Anthrax vaccine BioThrax is given to adults at increased risk of exposure in five doses, with a booster dose each year. It is also used as post-exposure prophylaxis in combination with antibiotics.
VAERS Explorer https://www.aretezoe.com/vaers-explorer
You can follow VAERS Explorer on social media:
Facebook: https://www.facebook.com/VAERSExplorer/
LinkedIn: https://www.linkedin.com/showcase/vaers-explorer/
Twitter: https://twitter.com/AreteZoe
MeWe: https://mewe.com/i/veronikavaldova1
Gab: https://gab.com/VAERSExplorer
TruthSocial: https://truthsocial.com/@vaersexplorer
Parler: https://parler.com/VAERSExplorer
YouTube: https://www.youtube.com/playlist?list=PLB6cShh65rNpo1dHiAQJ3-QEHyyM9zSJ1
Rumble: https://rumble.com/user/VAERSExplorer
Adenovirus vaccine

Adenoviruses commonly cause respiratory illnesses ranging from the common cold to pneumonia, croup, and bronchitis, but also gastroenteritis, conjunctivitis, cystitis, or neurological disease. Adenoviruses have been a common cause of acute respiratory illness in military recruits. As non-enveloped viruses, adenoviruses are relatively resistant to common disinfectants. There are more than 50 types of immunologically distinct adenoviruses. People with weakened immune systems are at high risk of developing a severe disease caused by adenovirus infection (CDC, Health.mil). The vaccine is mandatory for all enlisted military recruits (Army Regulation 40-562).
Vaccine: Adenovirus Type 4 and Type 7 Vaccine, Live, Oral (US)
Pharmacovigilance Workshop: Case Studies 

Pharmacovigilance Workshop
The workshop is designed to introduce pharmacovigilance to graduate students and working professionals interested in drug safety as a career field. After a brief introduction of publicly available data sources, each team received a case study that detailed a specific safety concern that resulted in a significant safety update of product labeling or product withdrawal.
Medicines may be withdrawn from markets because of risks to patients or business reasons. Change of benefit: risk profile is usually prompted by adverse effects that were either not previously detected, are more frequent, or more severe than anticipated based on the results of Phase III clinical trials. Market withdrawals are triggered by adverse events that were only made apparent from postmarketing surveillance data collected from population-wide use over more extended periods of time. The sources of information the regulatory agencies use when deciding on market withdrawal include meta-analyses and pooled analyses of data from clinical trials, clinical trials, spontaneous case reports, laboratory studies, observational studies, animal studies, and reviews of existing safety data.
In the U.S., individual case safety reports are collected in the FDA Adverse Event Reporting System (FAERS). In Europe, medication side effects are sent to national regulatory authorities and in the EU pharmacovigilance database EudraVigilance. The participants learned where to find clinical trials, market authorizations, and product labeling.
After the introductory presentation, each team received a case study that detailed a specific safety issue that resulted in a significant safety update of product labeling or product withdrawal. Each case study received product labeling and several scientific publications that discussed the safety concern in depth. Each team prepared a presentation with detailed overview of their assigned case study.
Are you interested in drug safety?
Try this for yourself!
Case studies:
Mylotarg (Gemtuzumab ozogamicin): no benefit, risk of death
Roaccutane (isotretinoin): teratogenic effect
Lariam (mefloquine): neuropsychiatric side effects
Zyprexa (olanzapine): stroke in patients with dementia
Avandia (rosiglitazone): myocardial infarction, death due to cardiovascular causes
Seroxat (paroxetine): suicidality
Xyrem (sodium oxybate): diversion, abuse
Coumadin (warfarin): bleeding
https://www.aretezoe.com/pharmacovigilance-workshop
Pharmacovigilance workshop 

The workshop is designed to introduce pharmacovigilance to graduate students and working professionals interested in drug safety as a career field. After a brief introduction of publicly available data sources, each team received a case study that detailed a specific safety concern that resulted in a significant safety update of product labeling or product withdrawal.
Medicines may be withdrawn from markets because of risks to patients or business reasons. Change of benefit: risk profile is usually prompted by adverse effects that were either not previously detected, are more frequent, or more severe than anticipated based on the results of Phase III clinical trials. Market withdrawals are triggered by adverse events that were only made apparent from postmarketing surveillance data collected from population-wide use over more extended periods of time. The sources of information the regulatory agencies use when deciding on market withdrawal include meta-analyses and pooled analyses of data from clinical trials, clinical trials, spontaneous case reports, laboratory studies, observational studies, animal studies, and reviews of existing safety data.
In the U.S., individual case safety reports are collected in the FDA Adverse Event Reporting System (FAERS). In Europe, medication side effects are sent to national regulatory authorities and in the EU pharmacovigilance database EudraVigilance. The participants learned where to find clinical trials, market authorizations, and product labeling.
After the introductory presentation, each team received a case study that detailed a specific safety issue that resulted in a significant safety update of product labeling or product withdrawal. Each case study received product labeling and several scientific publications that discussed the safety concern in depth. Each team prepared a presentation with detailed overview of their assigned case study.
Are you interested in drug safety?
Try this for yourself!
https://www.aretezoe.com/pharmacovigilance-workshop
Ethical Dilemmas in Gerontology (2017)

Published April 2017
Part of hospital test scenarios, escalation to ethics committee
Patients with a terminal illness who communicate their wish to die to a nurse shall receive appropriate care that is in line with institutional procedures, local laws, and their personal preferences. A nurse should be able to rely on the support of the institution he or she works for in terms of training, clear line of responsibility for such decisions, and unambiguously communicated expectations defined in organizational procedures. Assisted suicide is legal in Switzerland and several other European countries, in several states in the U.S., and in Canada. The mental capacity of the patient has to be considered in addition to locally applicable laws. Medical Power of Attorney is helpful if the patient previously described his or her wishes regarding end-of-life decisions and became incapacitated in the meantime. Financial toxicity, in addition to dubious effectiveness, contributes to the reluctance of some patients to undergo aggressive and invasive therapies. German physician Albert Moll in his book Medical Ethics (1902), argues that aggressive care in incurably ill patients is unethical. Healthcare staff, including nurses, can conscientiously object to assisting with suicide.
Deteriorating Patient with Sepsis: Early Diagnosis and Intervention (2017)

Deteriorating Patient with Sepsis: Early Diagnosis and Intervention
First published April 2017
Part of test scenarios for implementation of new sepsis guidelines
More from Arete-Zoe, LLC (20)
Availability of essential medicines in the Czech Republic (2017)

Availability of essential medicines in the Czech Republic (2017)
Post-marketing safety surveillance of medical devices and drug-device combina...

Post-marketing safety surveillance of medical devices and drug-device combina...
Mitigating consequences of a drug-facilitated sexual assault .pdf

Mitigating consequences of a drug-facilitated sexual assault .pdf
Approach to preparing for a biological attack (2017)

Approach to preparing for a biological attack (2017)
Improving the resilience of vulnerable populations

Improving the resilience of vulnerable populations
Addressing pediatric medication errors in ED setting utilizing Computerized P...

Addressing pediatric medication errors in ED setting utilizing Computerized P...
Let's talk causality attribution: Current practices and path forward 

Let's talk causality attribution: Current practices and path forward
Clinical evaluation: Supporting medical device product life-cycle. Applicable...

Clinical evaluation: Supporting medical device product life-cycle. Applicable...
Zpracování klinické dokumentace dle EU MDR 2017/745 

Zpracování klinické dokumentace dle EU MDR 2017/745
Deteriorating Patient with Sepsis: Early Diagnosis and Intervention (2017)

Deteriorating Patient with Sepsis: Early Diagnosis and Intervention (2017)
Recently uploaded
Premium MEAN Stack Development Solutions for Modern Businesses

Stay ahead of the curve with our premium MEAN Stack Development Solutions. Our expert developers utilize MongoDB, Express.js, AngularJS, and Node.js to create modern and responsive web applications. Trust us for cutting-edge solutions that drive your business growth and success.
Know more: https://www.synapseindia.com/technology/mean-stack-development-company.html
Meas_Dylan_DMBS_PB1_2024-05XX_Revised.pdf

Personal Brand Statement:
As an Army veteran dedicated to lifelong learning, I bring a disciplined, strategic mindset to my pursuits. I am constantly expanding my knowledge to innovate and lead effectively. My journey is driven by a commitment to excellence, and to make a meaningful impact in the world.
5 Things You Need To Know Before Hiring a Videographer

Dive into this presentation to discover the 5 things you need to know before hiring a videographer in Toronto.
Improving profitability for small business

In this comprehensive presentation, we will explore strategies and practical tips for enhancing profitability in small businesses. Tailored to meet the unique challenges faced by small enterprises, this session covers various aspects that directly impact the bottom line. Attendees will learn how to optimize operational efficiency, manage expenses, and increase revenue through innovative marketing and customer engagement techniques.
Digital Transformation in PLM - WHAT and HOW - for distribution.pdf

Presentation describing the Digital Transformation for PLM in the context of a Dassault Systemes portfolio.
India Orthopedic Devices Market: Unlocking Growth Secrets, Trends and Develop...

According to TechSci Research report, “India Orthopedic Devices Market -Industry Size, Share, Trends, Competition Forecast & Opportunities, 2030”, the India Orthopedic Devices Market stood at USD 1,280.54 Million in 2024 and is anticipated to grow with a CAGR of 7.84% in the forecast period, 2026-2030F. The India Orthopedic Devices Market is being driven by several factors. The most prominent ones include an increase in the elderly population, who are more prone to orthopedic conditions such as osteoporosis and arthritis. Moreover, the rise in sports injuries and road accidents are also contributing to the demand for orthopedic devices. Advances in technology and the introduction of innovative implants and prosthetics have further propelled the market growth. Additionally, government initiatives aimed at improving healthcare infrastructure and the increasing prevalence of lifestyle diseases have led to an upward trend in orthopedic surgeries, thereby fueling the market demand for these devices.
Enterprise Excellence is Inclusive Excellence.pdf

Enterprise excellence and inclusive excellence are closely linked, and real-world challenges have shown that both are essential to the success of any organization. To achieve enterprise excellence, organizations must focus on improving their operations and processes while creating an inclusive environment that engages everyone. In this interactive session, the facilitator will highlight commonly established business practices and how they limit our ability to engage everyone every day. More importantly, though, participants will likely gain increased awareness of what we can do differently to maximize enterprise excellence through deliberate inclusion.
What is Enterprise Excellence?
Enterprise Excellence is a holistic approach that's aimed at achieving world-class performance across all aspects of the organization.
What might I learn?
A way to engage all in creating Inclusive Excellence. Lessons from the US military and their parallels to the story of Harry Potter. How belt systems and CI teams can destroy inclusive practices. How leadership language invites people to the party. There are three things leaders can do to engage everyone every day: maximizing psychological safety to create environments where folks learn, contribute, and challenge the status quo.
Who might benefit? Anyone and everyone leading folks from the shop floor to top floor.
Dr. William Harvey is a seasoned Operations Leader with extensive experience in chemical processing, manufacturing, and operations management. At Michelman, he currently oversees multiple sites, leading teams in strategic planning and coaching/practicing continuous improvement. William is set to start his eighth year of teaching at the University of Cincinnati where he teaches marketing, finance, and management. William holds various certifications in change management, quality, leadership, operational excellence, team building, and DiSC, among others.
RMD24 | Retail media: hoe zet je dit in als je geen AH of Unilever bent? Heid...

Grote partijen zijn al een tijdje onderweg met retail media. Ondertussen worden in dit domein ook de kansen zichtbaar voor andere spelers in de markt. Maar met die kansen ontstaan ook vragen: Zelf retail media worden of erop adverteren? In welke fase van de funnel past het en hoe integreer je het in een mediaplan? Wat is nu precies het verschil met marketplaces en Programmatic ads? In dit half uur beslechten we de dilemma's en krijg je antwoorden op wanneer het voor jou tijd is om de volgende stap te zetten.
Putting the SPARK into Virtual Training.pptx

This 60-minute webinar, sponsored by Adobe, was delivered for the Training Mag Network. It explored the five elements of SPARK: Storytelling, Purpose, Action, Relationships, and Kudos. Knowing how to tell a well-structured story is key to building long-term memory. Stating a clear purpose that doesn't take away from the discovery learning process is critical. Ensuring that people move from theory to practical application is imperative. Creating strong social learning is the key to commitment and engagement. Validating and affirming participants' comments is the way to create a positive learning environment.
The Parable of the Pipeline a book every new businessman or business student ...

The-Parable of the Pipeline a book every new businessman or business student must read before diving into real world of business.
3.0 Project 2_ Developing My Brand Identity Kit.pptx

A personal brand exploration presentation summarizes an individual's unique qualities and goals, covering strengths, values, passions, and target audience. It helps individuals understand what makes them stand out, their desired image, and how they aim to achieve it.
Memorandum Of Association Constitution of Company.ppt

www.seribangash.com
A Memorandum of Association (MOA) is a legal document that outlines the fundamental principles and objectives upon which a company operates. It serves as the company's charter or constitution and defines the scope of its activities. Here's a detailed note on the MOA:
Contents of Memorandum of Association:
Name Clause: This clause states the name of the company, which should end with words like "Limited" or "Ltd." for a public limited company and "Private Limited" or "Pvt. Ltd." for a private limited company.
https://seribangash.com/article-of-association-is-legal-doc-of-company/
Registered Office Clause: It specifies the location where the company's registered office is situated. This office is where all official communications and notices are sent.
Objective Clause: This clause delineates the main objectives for which the company is formed. It's important to define these objectives clearly, as the company cannot undertake activities beyond those mentioned in this clause.
www.seribangash.com
Liability Clause: It outlines the extent of liability of the company's members. In the case of companies limited by shares, the liability of members is limited to the amount unpaid on their shares. For companies limited by guarantee, members' liability is limited to the amount they undertake to contribute if the company is wound up.
https://seribangash.com/promotors-is-person-conceived-formation-company/
Capital Clause: This clause specifies the authorized capital of the company, i.e., the maximum amount of share capital the company is authorized to issue. It also mentions the division of this capital into shares and their respective nominal value.
Association Clause: It simply states that the subscribers wish to form a company and agree to become members of it, in accordance with the terms of the MOA.
Importance of Memorandum of Association:
Legal Requirement: The MOA is a legal requirement for the formation of a company. It must be filed with the Registrar of Companies during the incorporation process.
Constitutional Document: It serves as the company's constitutional document, defining its scope, powers, and limitations.
Protection of Members: It protects the interests of the company's members by clearly defining the objectives and limiting their liability.
External Communication: It provides clarity to external parties, such as investors, creditors, and regulatory authorities, regarding the company's objectives and powers.
https://seribangash.com/difference-public-and-private-company-law/
Binding Authority: The company and its members are bound by the provisions of the MOA. Any action taken beyond its scope may be considered ultra vires (beyond the powers) of the company and therefore void.
Amendment of MOA:
While the MOA lays down the company's fundamental principles, it is not entirely immutable. It can be amended, but only under specific circumstances and in compliance with legal procedures. Amendments typically require shareholder
Sustainability: Balancing the Environment, Equity & Economy

[Note: This is a partial preview. To download this presentation, visit:
https://www.oeconsulting.com.sg/training-presentations]
Sustainability has become an increasingly critical topic as the world recognizes the need to protect our planet and its resources for future generations. Sustainability means meeting our current needs without compromising the ability of future generations to meet theirs. It involves long-term planning and consideration of the consequences of our actions. The goal is to create strategies that ensure the long-term viability of People, Planet, and Profit.
Leading companies such as Nike, Toyota, and Siemens are prioritizing sustainable innovation in their business models, setting an example for others to follow. In this Sustainability training presentation, you will learn key concepts, principles, and practices of sustainability applicable across industries. This training aims to create awareness and educate employees, senior executives, consultants, and other key stakeholders, including investors, policymakers, and supply chain partners, on the importance and implementation of sustainability.
LEARNING OBJECTIVES
1. Develop a comprehensive understanding of the fundamental principles and concepts that form the foundation of sustainability within corporate environments.
2. Explore the sustainability implementation model, focusing on effective measures and reporting strategies to track and communicate sustainability efforts.
3. Identify and define best practices and critical success factors essential for achieving sustainability goals within organizations.
CONTENTS
1. Introduction and Key Concepts of Sustainability
2. Principles and Practices of Sustainability
3. Measures and Reporting in Sustainability
4. Sustainability Implementation & Best Practices
To download the complete presentation, visit: https://www.oeconsulting.com.sg/training-presentations
Lookback Analysis

Explore our most comprehensive guide on lookback analysis at SafePaaS, covering access governance and how it can transform modern ERP audits. Browse now!
What is the TDS Return Filing Due Date for FY 2024-25.pdf

It is crucial for the taxpayers to understand about the TDS Return Filing Due Date, so that they can fulfill your TDS obligations efficiently. Taxpayers can avoid penalties by sticking to the deadlines and by accurate filing of TDS. Timely filing of TDS will make sure about the availability of tax credits. You can also seek the professional guidance of experts like Legal Pillers for timely filing of the TDS Return.
Skye Residences | Extended Stay Residences Near Toronto Airport

Experience unparalleled EXTENDED STAY and comfort at Skye Residences located just minutes from Toronto Airport. Discover sophisticated accommodations tailored for discerning travelers.
Website Link :
https://skyeresidences.com/
https://skyeresidences.com/about-us/
https://skyeresidences.com/gallery/
https://skyeresidences.com/rooms/
https://skyeresidences.com/near-by-attractions/
https://skyeresidences.com/commute/
https://skyeresidences.com/contact/
https://skyeresidences.com/queen-suite-with-sofa-bed/
https://skyeresidences.com/queen-suite-with-sofa-bed-and-balcony/
https://skyeresidences.com/queen-suite-with-sofa-bed-accessible/
https://skyeresidences.com/2-bedroom-deluxe-queen-suite-with-sofa-bed/
https://skyeresidences.com/2-bedroom-deluxe-king-queen-suite-with-sofa-bed/
https://skyeresidences.com/2-bedroom-deluxe-queen-suite-with-sofa-bed-accessible/
#Skye Residences Etobicoke, #Skye Residences Near Toronto Airport, #Skye Residences Toronto, #Skye Hotel Toronto, #Skye Hotel Near Toronto Airport, #Hotel Near Toronto Airport, #Near Toronto Airport Accommodation, #Suites Near Toronto Airport, #Etobicoke Suites Near Airport, #Hotel Near Toronto Pearson International Airport, #Toronto Airport Suite Rentals, #Pearson Airport Hotel Suites
20240425_ TJ Communications Credentials_compressed.pdf

"𝑩𝑬𝑮𝑼𝑵 𝑾𝑰𝑻𝑯 𝑻𝑱 𝑰𝑺 𝑯𝑨𝑳𝑭 𝑫𝑶𝑵𝑬"
𝐓𝐉 𝐂𝐨𝐦𝐬 (𝐓𝐉 𝐂𝐨𝐦𝐦𝐮𝐧𝐢𝐜𝐚𝐭𝐢𝐨𝐧𝐬) is a professional event agency that includes experts in the event-organizing market in Vietnam, Korea, and ASEAN countries. We provide unlimited types of events from Music concerts, Fan meetings, and Culture festivals to Corporate events, Internal company events, Golf tournaments, MICE events, and Exhibitions.
𝐓𝐉 𝐂𝐨𝐦𝐬 provides unlimited package services including such as Event organizing, Event planning, Event production, Manpower, PR marketing, Design 2D/3D, VIP protocols, Interpreter agency, etc.
Sports events - Golf competitions/billiards competitions/company sports events: dynamic and challenging
⭐ 𝐅𝐞𝐚𝐭𝐮𝐫𝐞𝐝 𝐩𝐫𝐨𝐣𝐞𝐜𝐭𝐬:
➢ 2024 BAEKHYUN [Lonsdaleite] IN HO CHI MINH
➢ SUPER JUNIOR-L.S.S. THE SHOW : Th3ee Guys in HO CHI MINH
➢FreenBecky 1st Fan Meeting in Vietnam
➢CHILDREN ART EXHIBITION 2024: BEYOND BARRIERS
➢ WOW K-Music Festival 2023
➢ Winner [CROSS] Tour in HCM
➢ Super Show 9 in HCM with Super Junior
➢ HCMC - Gyeongsangbuk-do Culture and Tourism Festival
➢ Korean Vietnam Partnership - Fair with LG
➢ Korean President visits Samsung Electronics R&D Center
➢ Vietnam Food Expo with Lotte Wellfood
"𝐄𝐯𝐞𝐫𝐲 𝐞𝐯𝐞𝐧𝐭 𝐢𝐬 𝐚 𝐬𝐭𝐨𝐫𝐲, 𝐚 𝐬𝐩𝐞𝐜𝐢𝐚𝐥 𝐣𝐨𝐮𝐫𝐧𝐞𝐲. 𝐖𝐞 𝐚𝐥𝐰𝐚𝐲𝐬 𝐛𝐞𝐥𝐢𝐞𝐯𝐞 𝐭𝐡𝐚𝐭 𝐬𝐡𝐨𝐫𝐭𝐥𝐲 𝐲𝐨𝐮 𝐰𝐢𝐥𝐥 𝐛𝐞 𝐚 𝐩𝐚𝐫𝐭 𝐨𝐟 𝐨𝐮𝐫 𝐬𝐭𝐨𝐫𝐢𝐞𝐬."
Maksym Vyshnivetskyi: PMO Quality Management (UA)

Maksym Vyshnivetskyi: PMO Quality Management (UA)
Lemberg PMO School 2024
Website – https://lembs.com/pmoschool
Youtube – https://www.youtube.com/startuplviv
FB – https://www.facebook.com/pmdayconference
Recently uploaded (20)
Premium MEAN Stack Development Solutions for Modern Businesses

Premium MEAN Stack Development Solutions for Modern Businesses
Set off and carry forward of losses and assessment of individuals.pptx

Set off and carry forward of losses and assessment of individuals.pptx
5 Things You Need To Know Before Hiring a Videographer

5 Things You Need To Know Before Hiring a Videographer
Digital Transformation in PLM - WHAT and HOW - for distribution.pdf

Digital Transformation in PLM - WHAT and HOW - for distribution.pdf
India Orthopedic Devices Market: Unlocking Growth Secrets, Trends and Develop...

India Orthopedic Devices Market: Unlocking Growth Secrets, Trends and Develop...
RMD24 | Retail media: hoe zet je dit in als je geen AH of Unilever bent? Heid...

RMD24 | Retail media: hoe zet je dit in als je geen AH of Unilever bent? Heid...
The Parable of the Pipeline a book every new businessman or business student ...

The Parable of the Pipeline a book every new businessman or business student ...
3.0 Project 2_ Developing My Brand Identity Kit.pptx

3.0 Project 2_ Developing My Brand Identity Kit.pptx
Memorandum Of Association Constitution of Company.ppt

Memorandum Of Association Constitution of Company.ppt
Sustainability: Balancing the Environment, Equity & Economy

Sustainability: Balancing the Environment, Equity & Economy
What is the TDS Return Filing Due Date for FY 2024-25.pdf

What is the TDS Return Filing Due Date for FY 2024-25.pdf
Skye Residences | Extended Stay Residences Near Toronto Airport

Skye Residences | Extended Stay Residences Near Toronto Airport
20240425_ TJ Communications Credentials_compressed.pdf

20240425_ TJ Communications Credentials_compressed.pdf
Systems Approach to Training
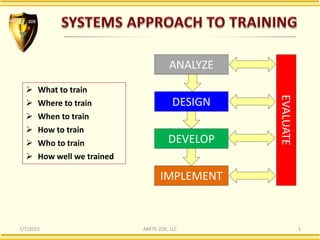
- 1. 7/7/2015 ARETE-ZOE, LLC 1 EVALUATE ANALYZE DESIGN DEVELOP IMPLEMENT What to train Where to train When to train How to train Who to train How well we trained
- 2. Input Process Evaluation data Changed situation Different needs Changed requirements Reorganization Output Roles & Responsibilities Analysis of functional needs is necessary to produce Design of the system as appropriate/relevant for its current and estimated future scope of activities. The products of analysis will be the foundation of what constitutes a successful design. Design translates analysis data into a structure or blueprint of both organization and process. PERFORMANCE data is collected to produce a comprehensive list of tasks and performance requirements, Roles & Responsibilities and draft Training Plan 7/7/2015 ARETE-ZOE, LLC 2 Job analysis Task analysis Performance requirements Responsibilities Determine setting Materials Training Plan MISSION ANALYSIS | COLLECTIVE TASK ANALYSIS | JOB ANALYSIS | INDIVIDUAL TASK ANALYSIS
- 3. The outputs of the ANALYSIS Phase become the inputs to the DESIGN Phase. The objective of training is to simulate functioning of the system as closely as possible to the actual environment. Input Process Output Roles & Responsibilities Training Plan Target population Learning objectives Learning analysis hierarchies Sequence training Select training site Learning strategy Learning activities Design test Target population The outputs of the Design phase include: Charts with organizational structure, task list inventory by staff position and function, flowchart describing data flow including all interfaces, a list of SOPs, Working Instructions, and Training Materials which will have to be created including definition of scope and for whom they will be relevant. 7/7/2015 ARETE-ZOE, LLC 3 TARGET POPULATION| LEARNING OBJECTIVES| LEARNING STRATEGIES & METHODS| TESTS Test items Methods, Media Flowcharts, charts List of SOPs, WIs List of TMs
- 4. Input Process Target Population Description TEST items Methods & Media Charts, flowcharts List of SOPs and WIs, Training materials list Output System Master File Development involves turning the design outputs into the products, programs, and materials required to implement clinical research activities at organizational and individual level. Products include SOPs and Working Instructions according to scope and definition described in the Design phase. Evaluation of all developed materials is a critical part of the development process because it validates their practicality. FLOWCHARTS, SOPs, manuals, risk management plans, training structure and materials, and construction and validation of tests to determine effectiveness of the system prior to its implementation. 7/7/2015 ARETE-ZOE, LLC 4 Review design outputs Revise current material Establish a team of subject matter experts Plan instructor training Plan key personnel training In-Process Reviews (IPRs) Review developed products Staff preliminary products Validate products and tests Make necessary revisions Obtain approval/authorization Reproduce products Acquire resources
- 5. Input Process System Master File Output Trained staff, faculty and cadre Implementation is preparing for, training and conducting all activities according to SOPs and WIs, using materials, documents, platforms, templates, and methods designed and developed in previous phases. Key activities: Train relevant people on activities relevant to them as defined in previous phases using training materials designed phase I and developed in phase II; conduct activities as described in Master File; Collect and collate data on performance of the system as whole necessary for its continuing evaluation, in order to detect, revise, and correct any occurring inefficiencies. Document key decisions and maintain audit trail to support any decisions made and actions taken during the implementation process. Transition of a blueprint into real-world environment and conducting instruction, tests, and evaluations according to SMF to enable personnel to achieve their individual and collective objectives. 7/7/2015 ARETE-ZOE, LLC 5 Train staff, faculty and cadre for their duties Maintain records Conduct training Use validated materials Develop schedules Collect and collate data Maintain audit trail Training records Documentation of training modifications and rationale Audit trail
- 6. Input Process Test Data Data collected on performance of the system Output Evaluation summary Evaluation is essential to determine if the system as designed, developed, and implemented meets its business needs. Evaluation provides the data to determine if individuals can perform the tasks assigned to them to the expected standard, and if individual sites and the organization as whole can accomplish their objectives to the required standards and at within established time lines. Evaluation also provides data if the system as whole is appropriate and cost effective. Internal and external audit validates the system as whole with regards to regulatory compliance in target markets. Needs analysis shall verify a training problem and identify non-training solutions to a performance problem, such as doctrine, leadership, organization, and material required capabilities. EVALUATION is based on criterion-referenced checks which ensure continuous improvement of a newly developed system. 7/7/2015 ARETE-ZOE, LLC 6 Plan evaluation Internal evaluation External evaluation Analyze data Manage data Revisions as necessary Needs analysis
