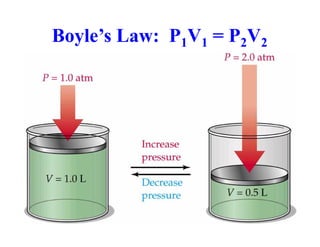
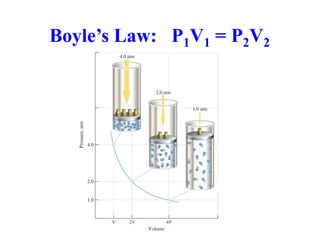
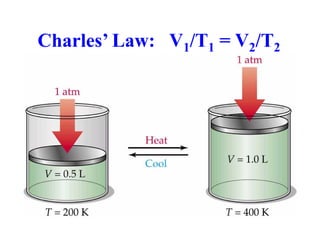
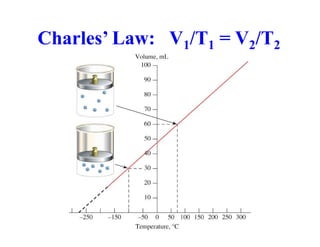
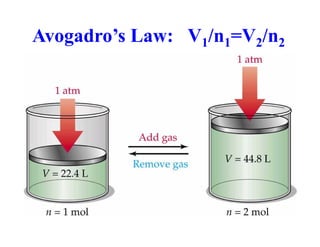
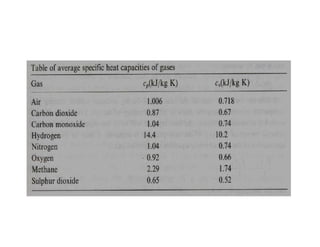
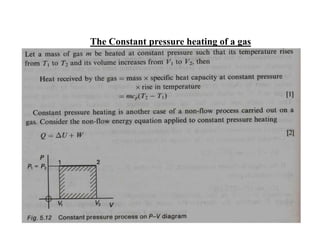
This document discusses several gas laws including Boyle's Law, Charles' Law, Avogadro's Law, Gay-Lussac's Law, Dalton's Law, and properties of gases such as specific heat capacities. Boyle's Law states that the pressure and volume of a gas are inversely related at constant temperature. Charles' Law specifies that the volume of a gas varies directly with the absolute temperature at constant pressure. Avogadro's Law describes how the volume of a gas is directly related to the number of moles at constant temperature and pressure.