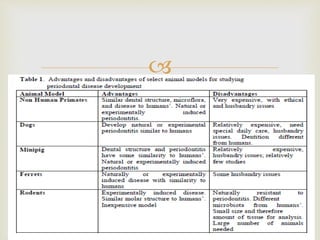
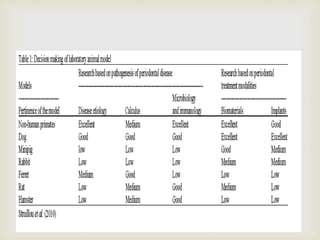
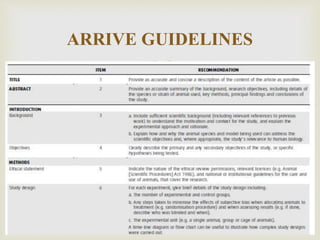
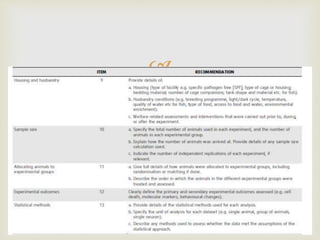
This document provides an overview of animal models used in periodontal research. It discusses the definition and history of animal models, the need for animal models in periodontal research given limitations of human studies, and various categories and classifications of animal models. The document then examines specific animal models used in periodontal research, including rats, mice, and hamsters, describing their anatomy, how periodontal disease presents in each, and advantages and limitations of each model.














![
CLASSIFICATION
(Page &Schroeder 1982)
I] Small rodents
E. g: Mice, Rats, Hamsters, Minks.
II] Larger animals
E. g: Dogs and sheep.
III] Non-Human primates
E. g: Baboon, Macaca, Chimpanzee and Gorilla
IV] Others
E. g: Apes, Cats, Horses, Guinea pigs, Mongooses, Wolves, Foxes,
Rabbits, Ferret etc.](https://image.slidesharecdn.com/animalmodels-160617185419/85/Animal-models-15-320.jpg)