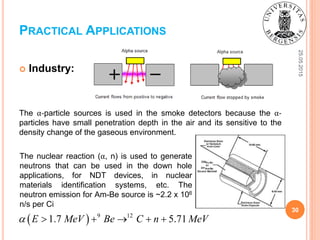
This document provides background information on alpha decay, including its discovery, experimental observations, and theoretical explanations. It discusses how alpha decay was first observed in uranium salts and describes the four main types of radioactivity. The document outlines experiments showing that alpha particles have a charge of +2 and consist of two protons and two neutrons. It also summarizes George Gamow's 1928 quantum tunneling theory of alpha decay, which explained how alpha particles can escape the nucleus despite facing a Coulomb barrier. The theory predicts the relationship between half-life and emission energy that had previously been observed empirically.




![GENERAL INFORMATION ABOUT 𝛂-DECAY
AND HISTORY
The early experiments of Curie and of Rutherford showed that the
radiations from radioactive substances contained components of
different penetrating power, as assessed by their absorption in matter.
25.05.2015
5
The less penetrating rays, which were
completely absorbed by a few cm of
air were called α-rays. The more
penetrating components, which were
absorbed by about 1 mm of lead were
named β-rays. Both the α- and β-rays
were shown to be corpuscular in
character by magnetic deflection
methods.
Fig. 1. Effect of a transverse magnetic field on radiations [1].
B](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-5-320.jpg)


![EXPERIMENTAL OBSERVATIONS
25.05.2015
8
Fig. 1: Experimental values for the alpha decay Q values [2].
The alpha
decay energy
is ranging from
2 to 12 MeV,
the mean value
for all isotopes
is about 6 MeV.](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-8-320.jpg)
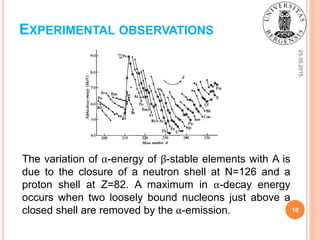
![25.05.2015
Fig. 2: Energy release in the α-decay of the heavy elements, showing the
regularities of the ground-state α-decay energies [1].
EXPERIMENTAL OBSERVATIONS
β-stable isotopes
9](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-9-320.jpg)
![25.05.2015
11
Fig. 4: Decay constant vs. range of a-emitting
nuclei known in 1921 [2].
EXPERIMENTAL OBSERVATIONS
A correlation between the
lifetime and energy of the α-
particle emission was noticed
by Geiger and Nuttall as early
as 1921:
3
log loga b R
R v
](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-11-320.jpg)
![25.05.2015
12
Fig. 5: Fine structure of α-particle spectra of 212Po. α0 and α1 are the most
intense α-lines. [2].
EXPERIMENTAL OBSERVATIONS
212 208
84 82 6.2Po Pb MeV
Fine structure in a-ray spectra
was demonstrated in the high
resolution experiments of
Rosenblum (1929) and of
Rutherford. It is due to the
excitation of levels of residual
nucleus.
The long-range
α-particles are
associated with
disintegrations of
an excited state of
the initial nucleus.](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-12-320.jpg)
![THEORY OF ALPHA DECAY
25.05.2015
13
Classical Physics
cannot explain how
the particles with
energy of up to 12
MeV can penetrate
through the Coulomb
barrier of 20-40 MeV.
Fig. 6: A simplified schematic of the Coulomb barrier in the nucleus.
1/3
238
92
108
52
2 [MeV]
, 29.7
, 21.8
V R ZA
V R U MeV
V R Te MeV
](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-13-320.jpg)

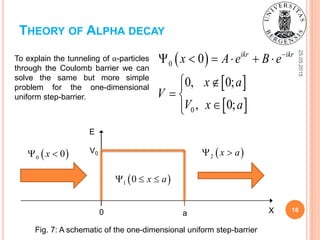
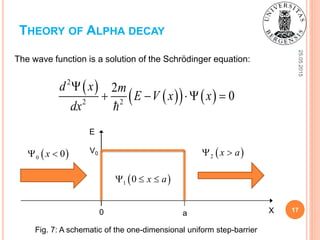
![25.05.2015
15
THEORY OF ALPHA DECAY
Fig. 7: A representation of α-particle as a wave
function the amplitude of which decreases behind
the Coulomb barrier after the tunneling through it
[1].
0 r R
0
ikr ikr
r R A e B e
α-particle in the nucleus:](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-15-320.jpg)


![25.05.2015
20
THEORY OF ALPHA DECAY
0
2
0
,
2
,
4
V r R
V r Ze
r R
r
The potential energy of the α-particle in Coulomb barrier is:
And the barrier penetration coefficient is [3]:
2 2
0 0
2
exp 2 ,
2 2
b
R
Ze Ze
D m E dr b
r E
1/3
2
exp 2 arccos 1
2 [MeV]
V R V R V RR
D mV R
E E E
V R ZA
](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-20-320.jpg)
![25.05.2015
21
THEORY OF ALPHA DECAY
1 ,
2
l l R
mE
If α decay takes place to or from an excited state, the angular
momentum of the α-particle may equal to different values limited by
the nucleus size:
where λ and l ≤ 10 are the de Broglie wave of the α-particle and the
orbital moment, respectively. It leads to the increase of the total
potential barrier due to the additional component – angular momentum
barrier of the α-particle [3]:
2
2
1
,
2
,
0.002 1
l
l
Coulomb
l l
V r l
mr
V R l
l l
V R
](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-21-320.jpg)
![25.05.2015
22
THEORY OF ALPHA DECAY
The barrier penetration coefficient , D, depends on the both barriers:
The following approximation can be used for the range of orbital
moment l<7 [3]:
2 2
2
0
1 2
,
2 4
l l Ze
V r R l
mr r
2
0
2
exp 2 , ,
2
b
R
Ze
D m V r l E dr b
E
0 0 1/63
238
0 92 0
1
exp 2.027
exp 0.0849 1
l l
l l
l l
D D
Z A
D U D l l
](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-22-320.jpg)

![25.05.2015
24
The odd nucleon α-emitters, especially in ground
state transitions, decay at a slower rate than that
suggested by the simple one-body theory.
The decays of the odd nuclei are referred to as
“hindered decays” and a “hindrance factor” may be
defined as the ratio of the measured partial half-life to
the calculated one.
THEORY OF ALPHA DECAY
.
1/2
1/2
[1;10000]
Meas
Theory
T
HF
T
The hindered decays can be explained by the detailed
quantum mechanical analysis of the formation of a-
particles in the nuclei with different energies and
orbital momenta.](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-24-320.jpg)


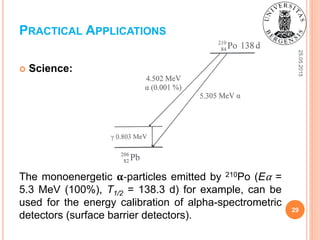
![25.05.2015
27
Energy:
As an example, the composed nuclear battery VERIIT was
developed in Kharkiv Institute of Physics and Technology,
2011, Ukraine [4]. It is based on the transformation of the
kinetic energy of α-particles into the charge through the
ionization process in several thin Me-layers of the battery.
The source is 210Po (Eα = 5.3 MeV, T1/2 = 138.3 d);
The efficiency is about 10%;
Pe = 89 μW, Uout = 15 V.
PRACTICAL APPLICATIONS
eff
X Y Q q
dQ
i
dt w
](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-27-320.jpg)

![REFERENCES
25.05.2015
31
[1] W.E. Burcham. Nuclear physics: an introduction.
Longman.; 1973.
[2] B.A. Brown. Lecture notes in nuclear structure physics.
Michigan State University.; 2005
[3] K.N. Mukhin. Nuclear physics. Macdonald & Co.; 1970.
[4] V.I. Karas, S.I. Kononenko, V.I. Muratov, V.T. Tolok, New
type radionuclide battery VERIIT for the space
applications (Report), Kharkiv Institute of Physics and
Technology, 2011.](https://image.slidesharecdn.com/sofiienkoandriialphadecayuib2015-150525194555-lva1-app6891/85/Alpha-decay-physical-background-and-practical-applications-31-320.jpg)
