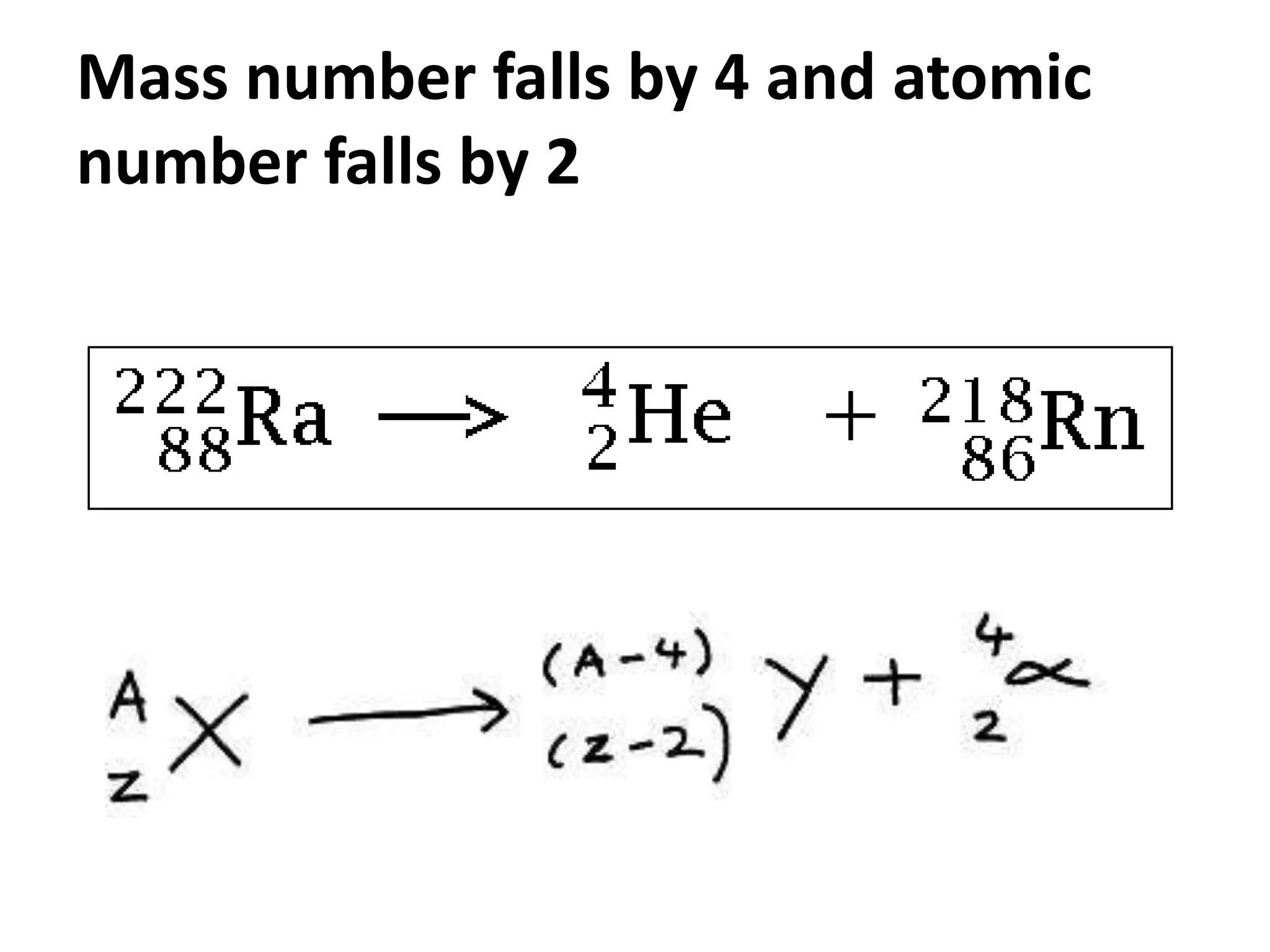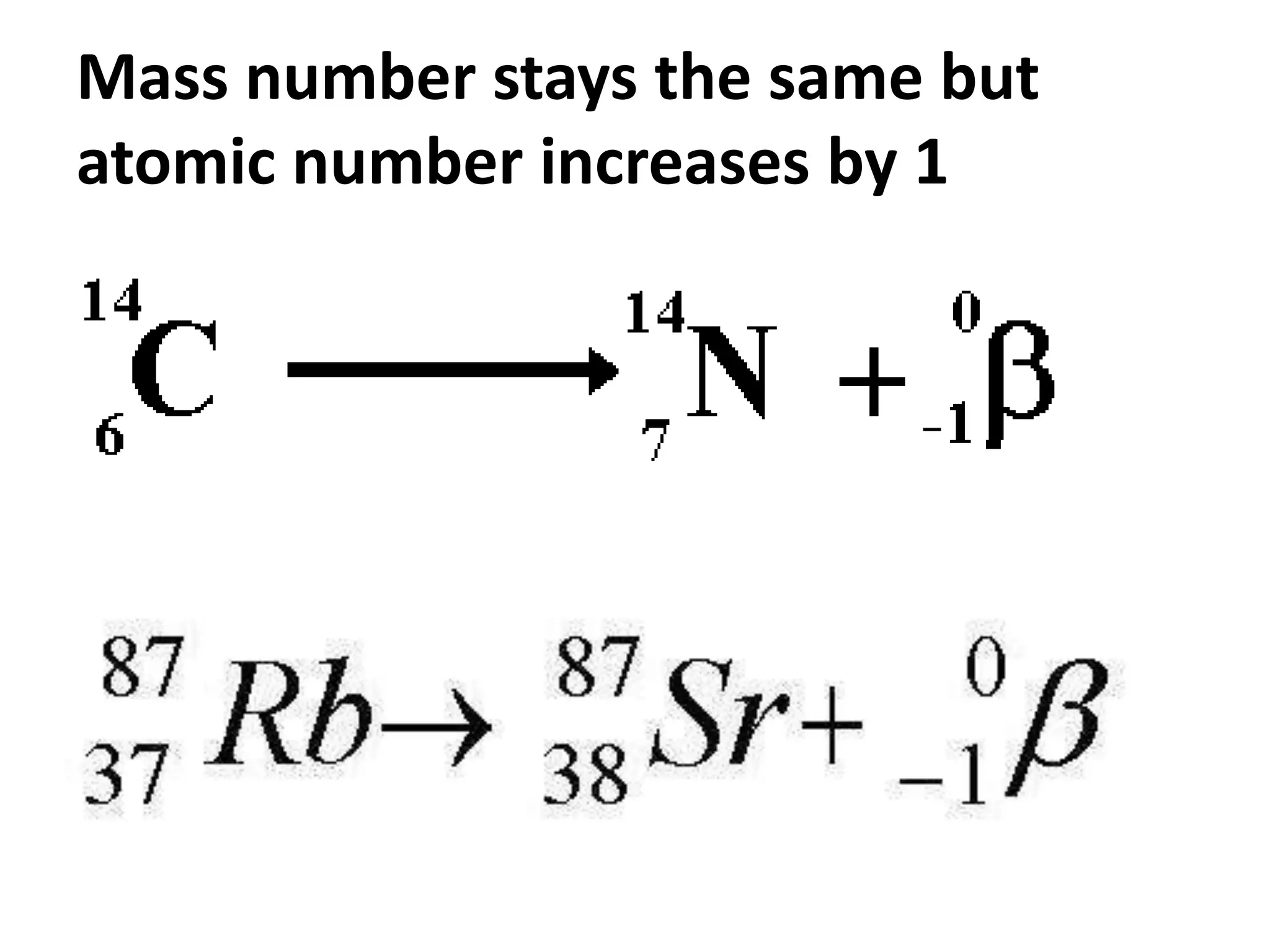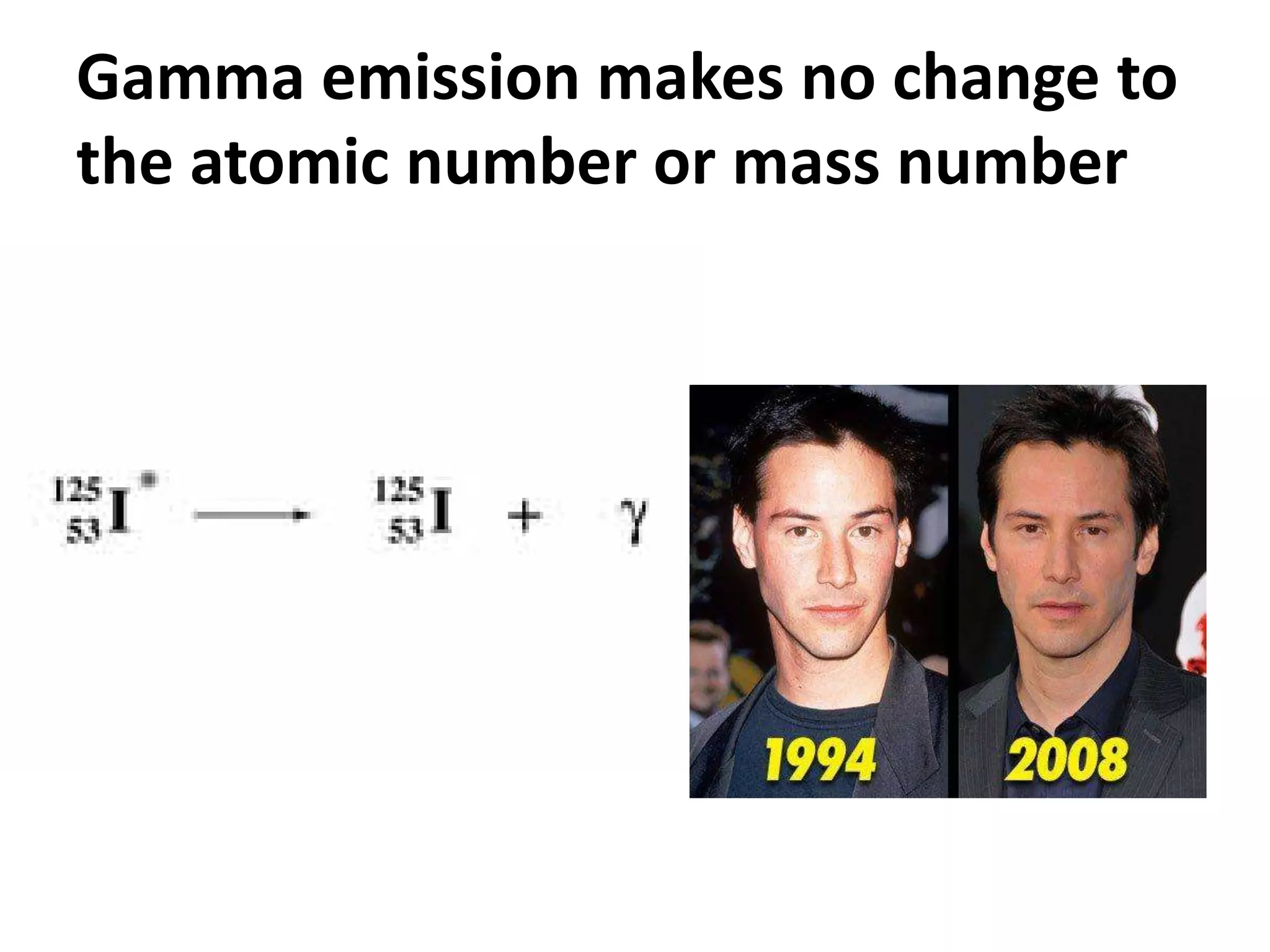Embed presentation
Downloaded 42 times


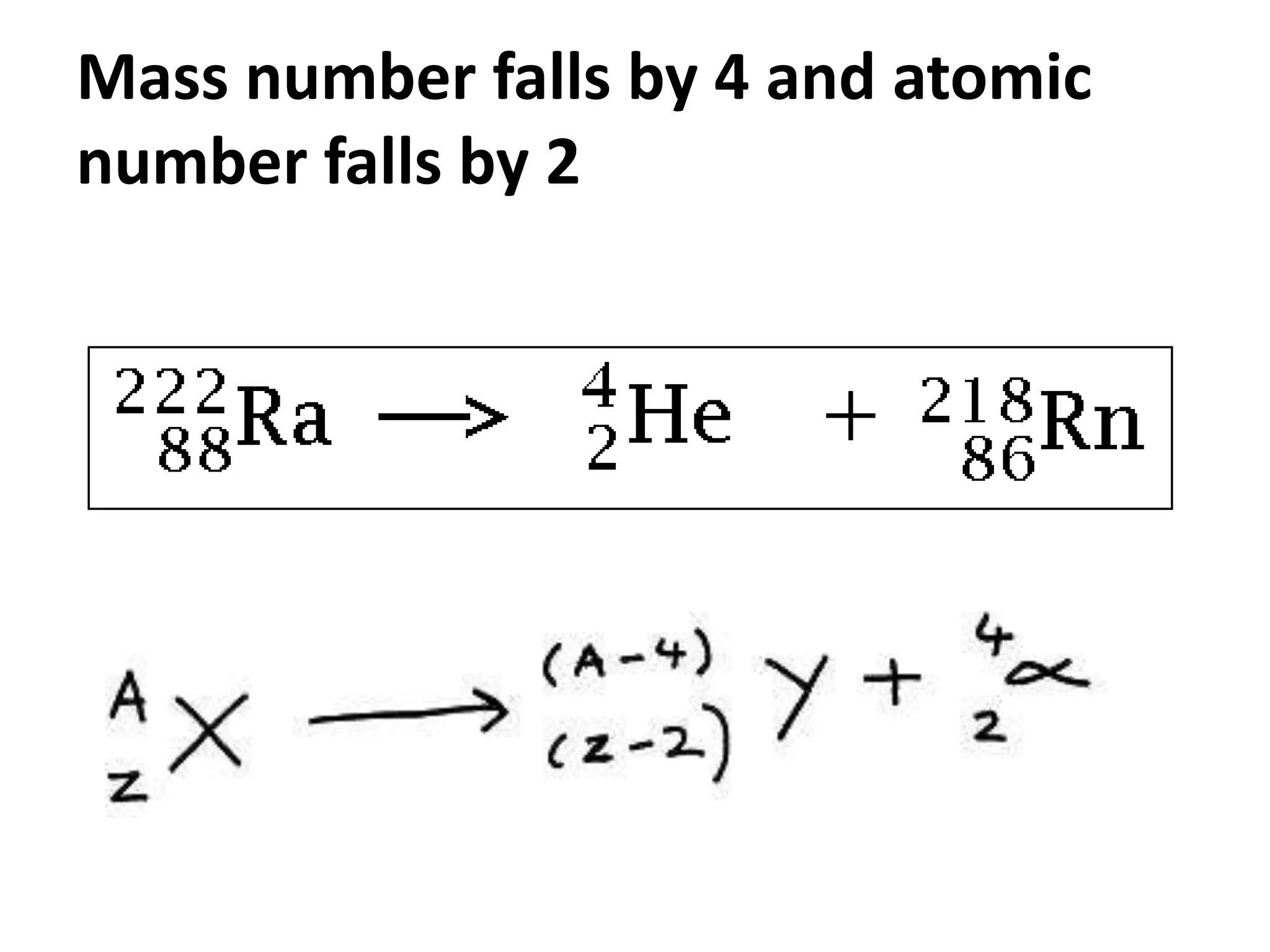


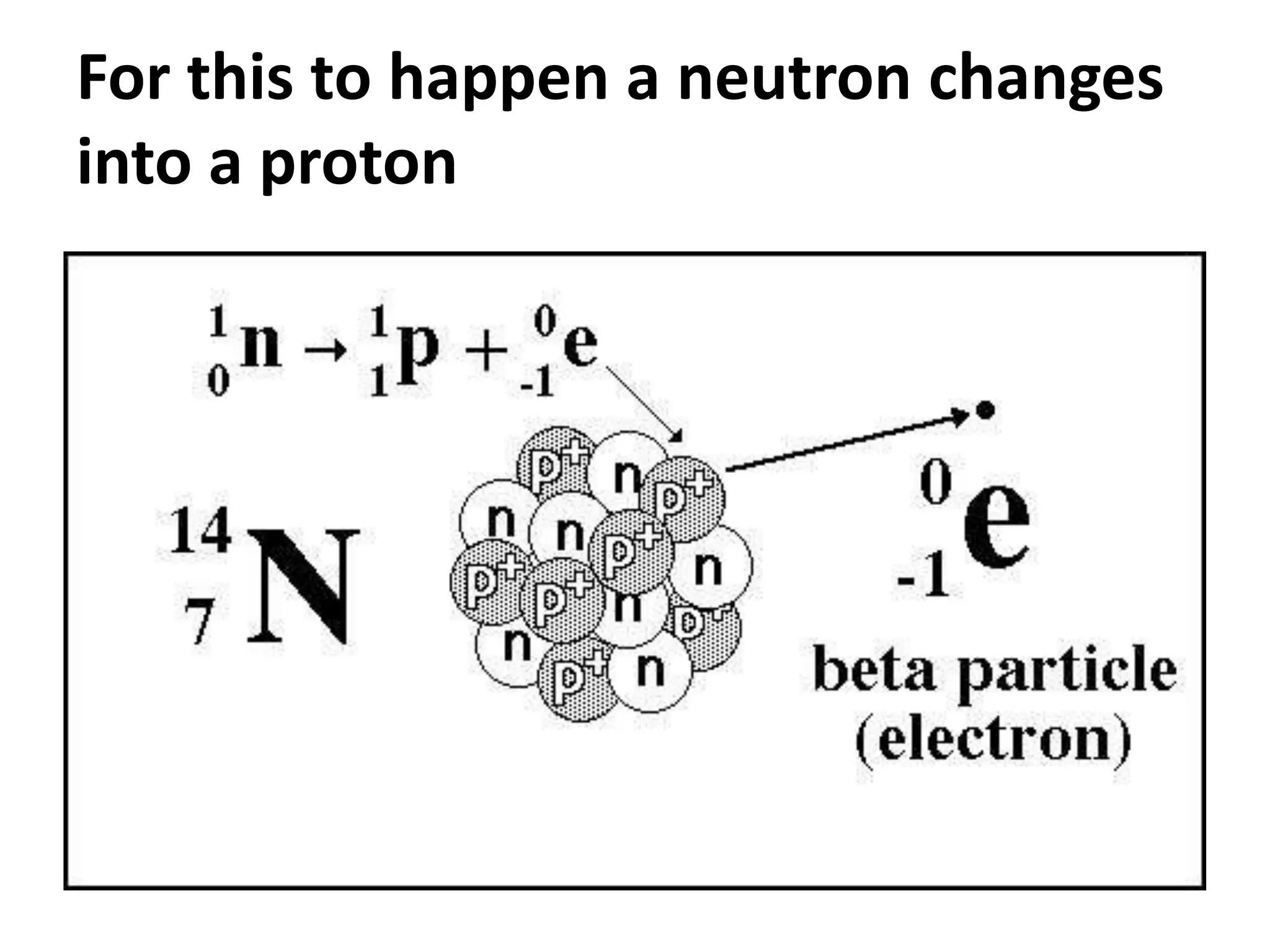
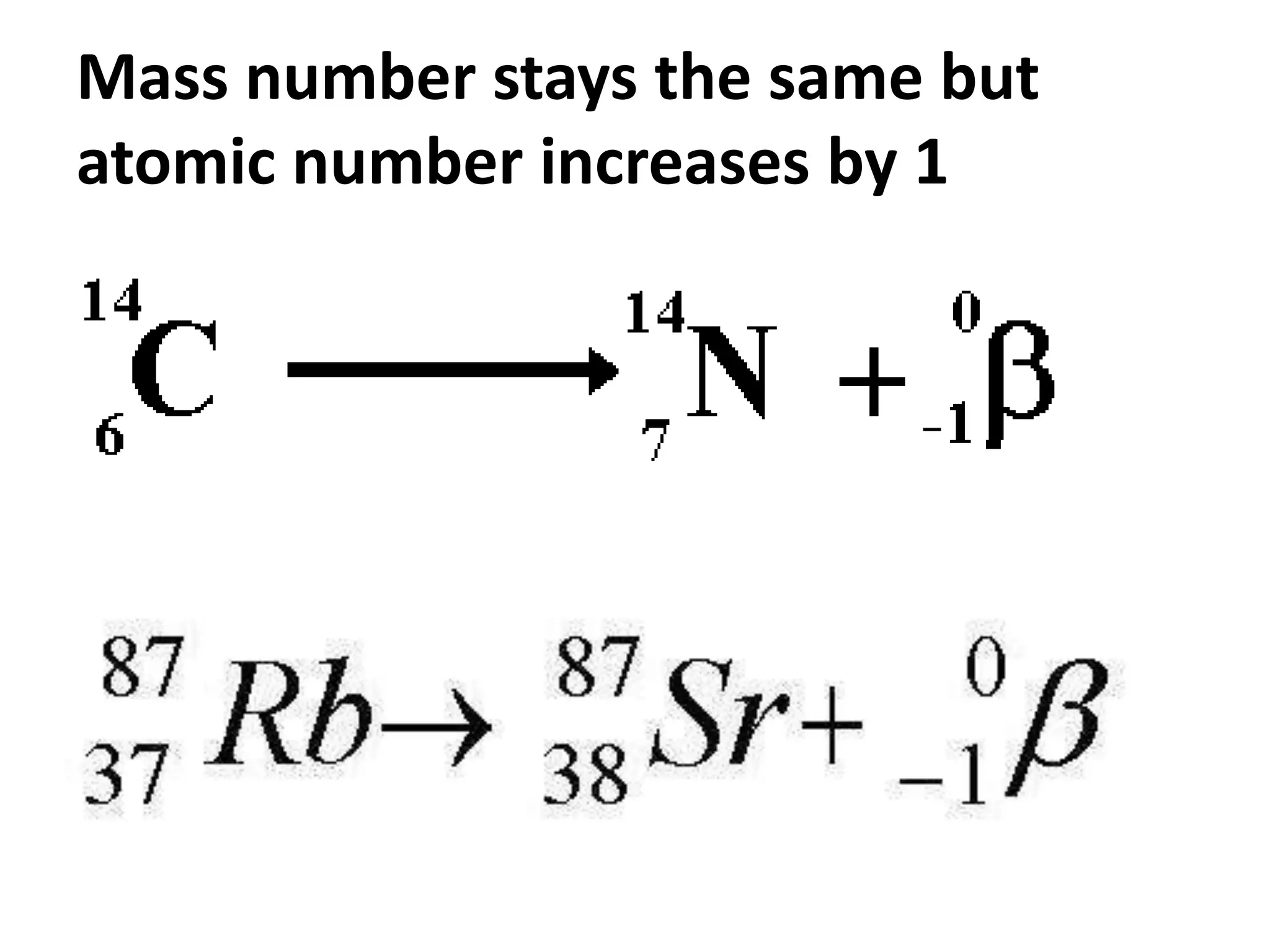

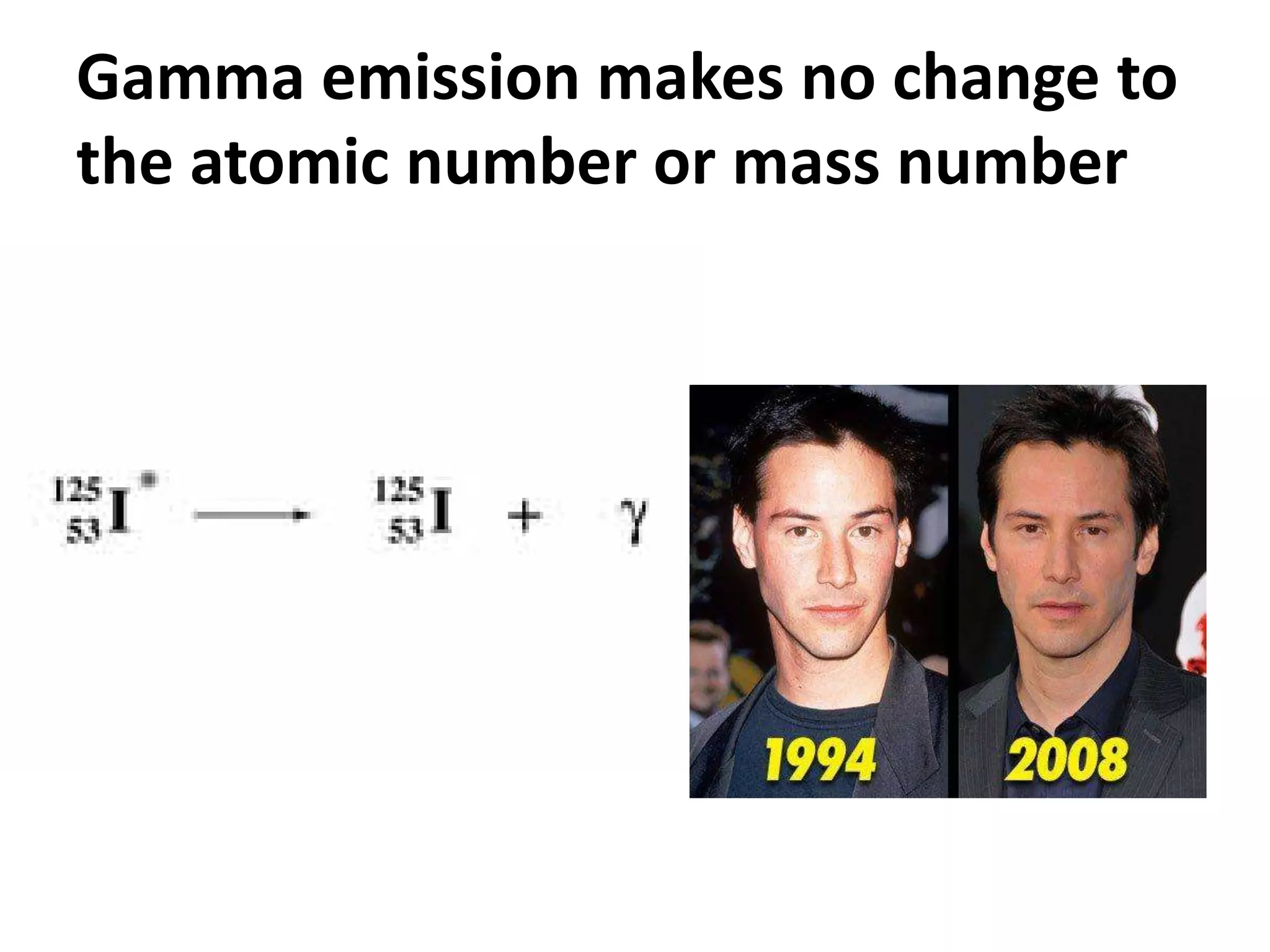

This document discusses three types of radioactive decay: alpha, beta, and gamma. Alpha decay involves emitting two protons and two neutrons from the nucleus, represented by helium, which decreases the mass and atomic number by 4 and 2 respectively. Beta decay occurs when an electron is ejected from the nucleus, which changes a neutron to a proton, keeping the mass the same but increasing the atomic number by 1. Gamma decay simply releases excess energy without changing the mass or atomic number.