The document discusses the discovery of radioactivity and the different types of radioactive decay:
- Alpha, beta, and gamma decay were discovered through experiments by Henri Becquerel, Marie and Pierre Curie, and Ernest Rutherford in the late 19th century.
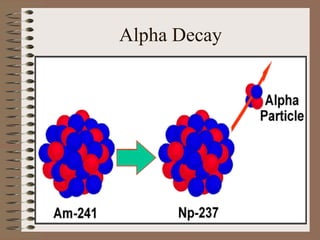
- Alpha decay involves emitting an alpha particle (helium nucleus), beta decay involves emitting an electron or positron, and gamma decay involves emitting high-energy photons.
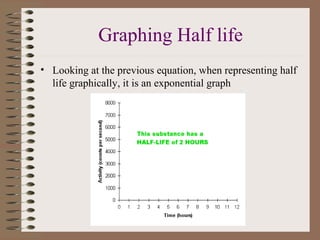
- The decays result in the transmutation of elements and conservation of nucleon number. Radioactive decay occurs at exponential rates described by half-lives and can be used to date materials.