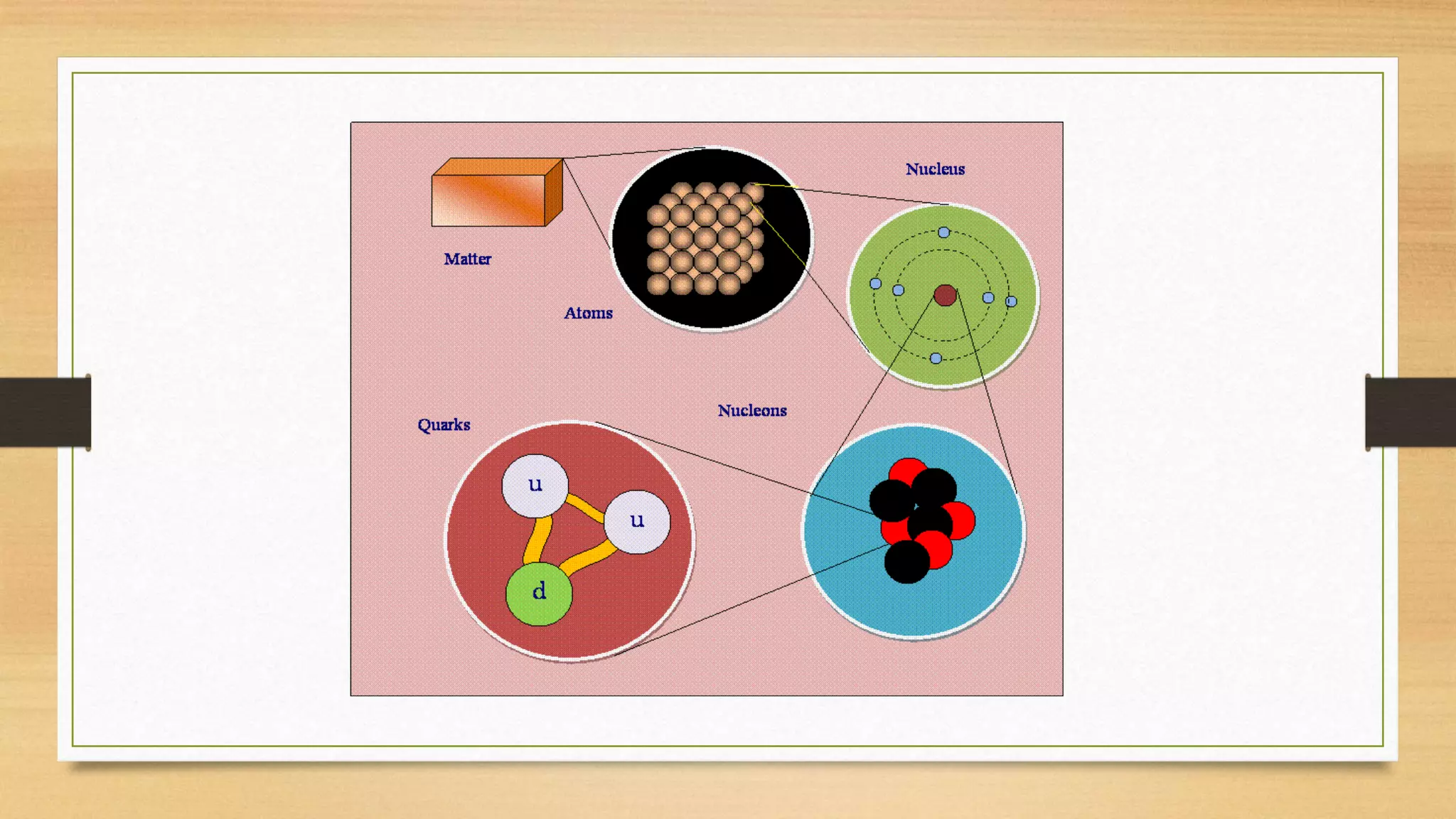
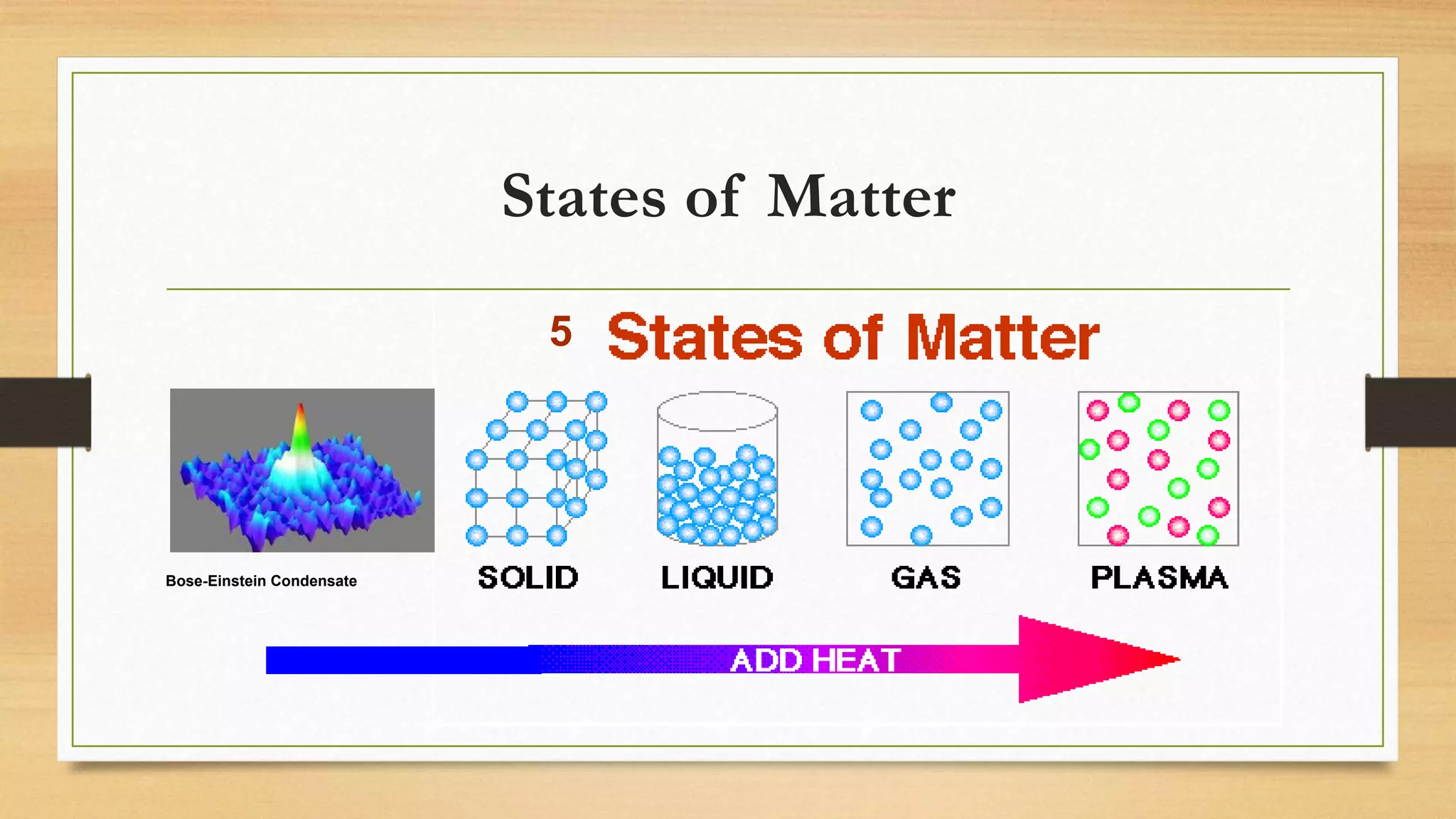
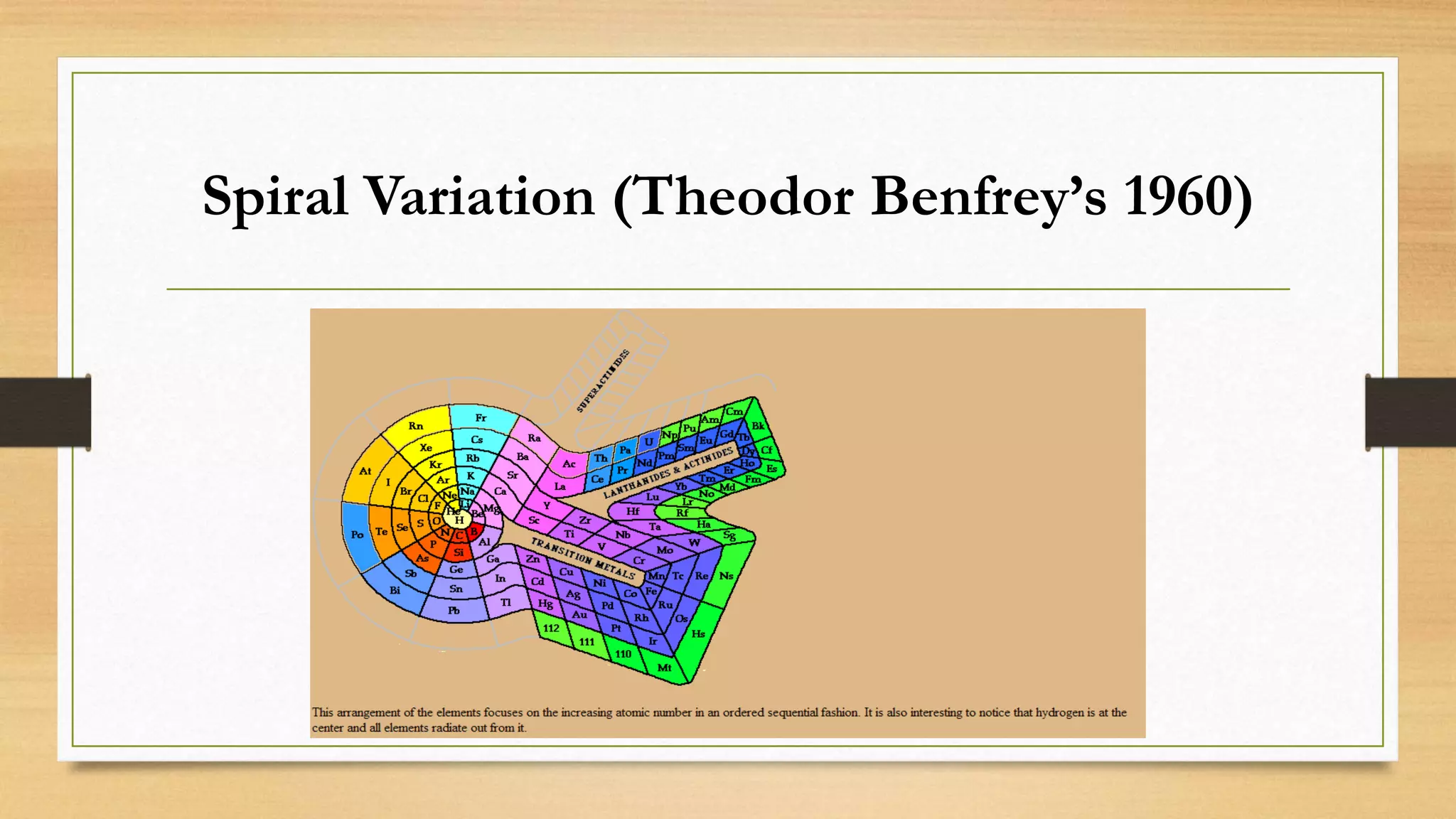
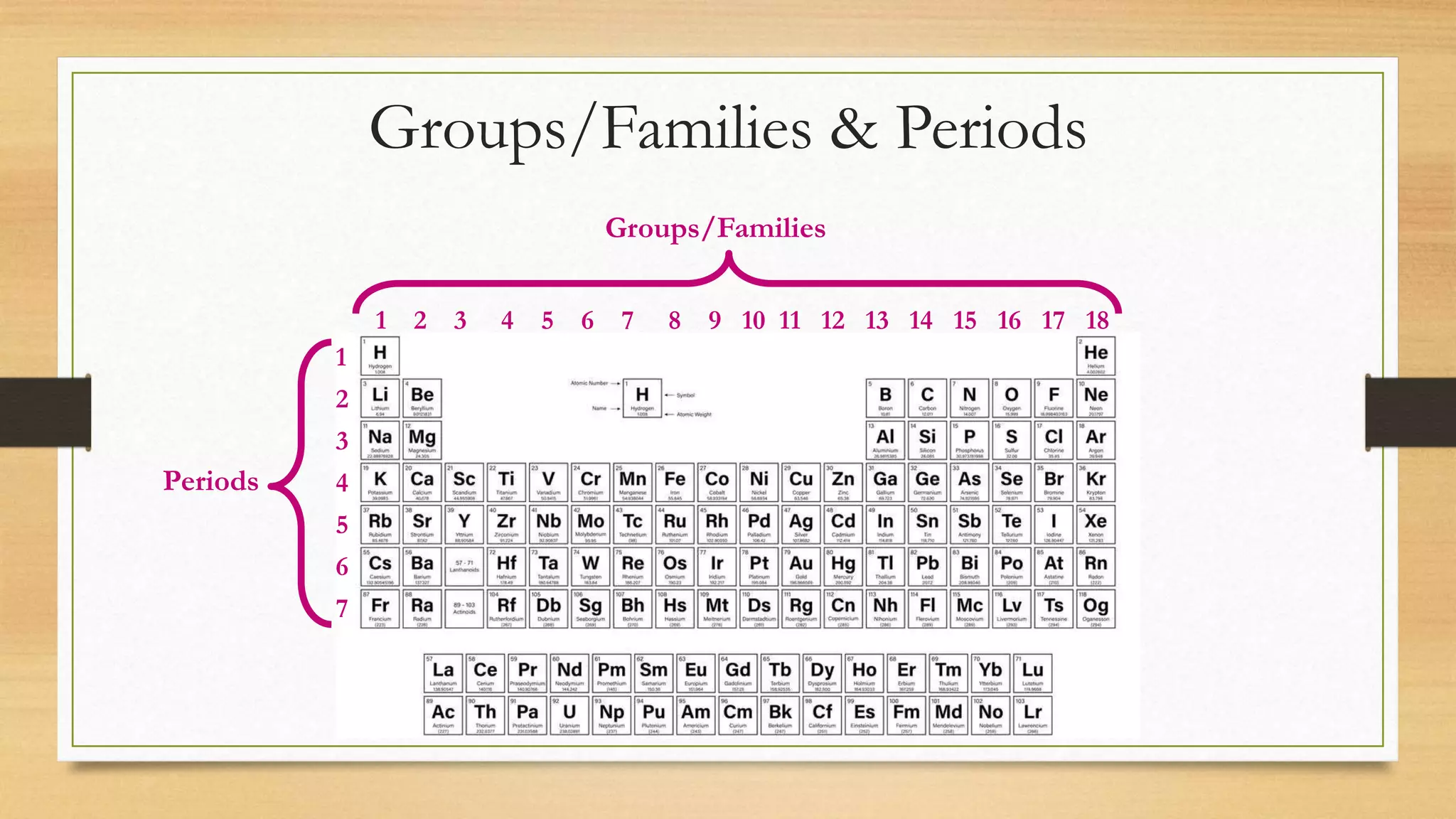
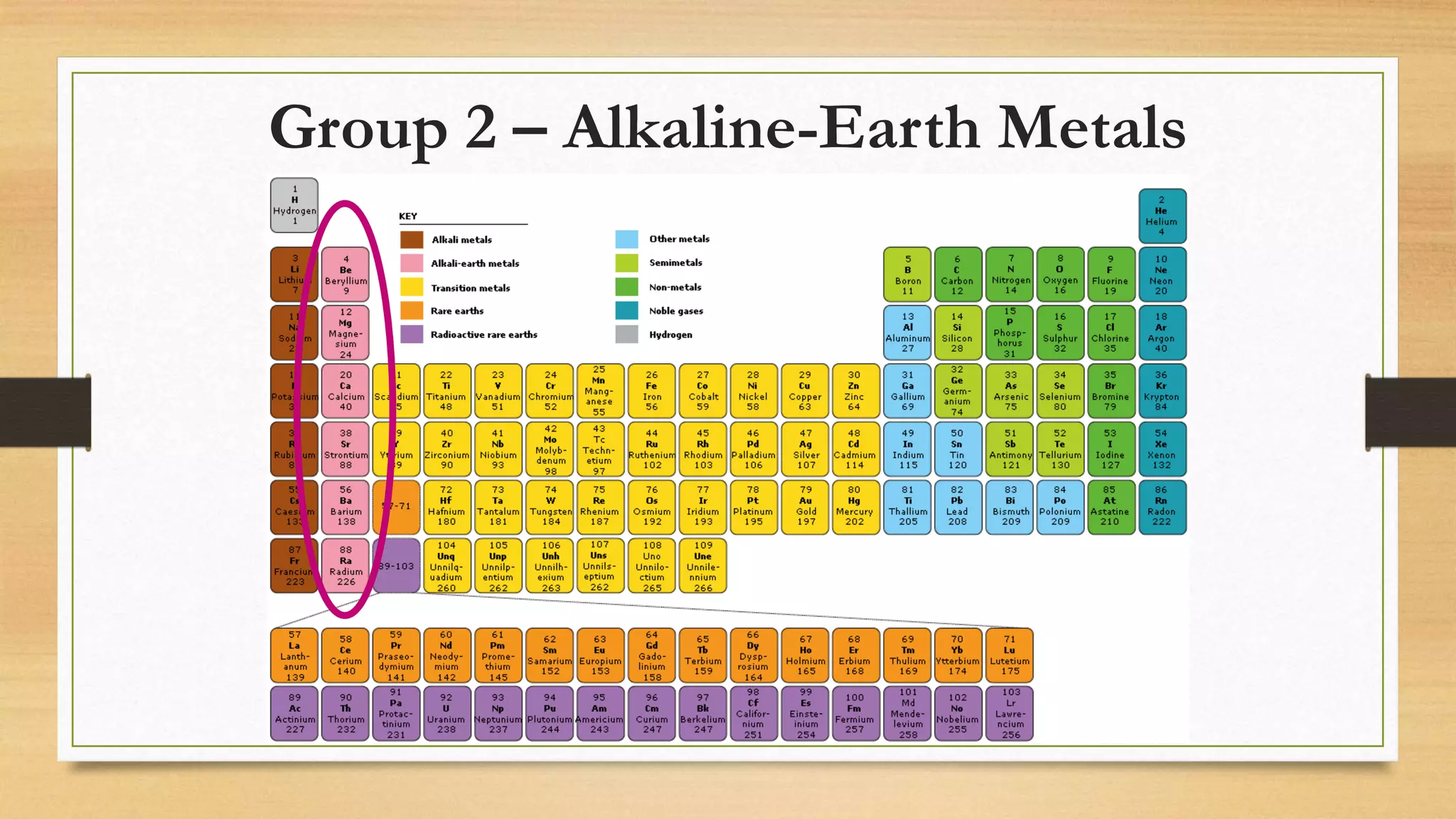
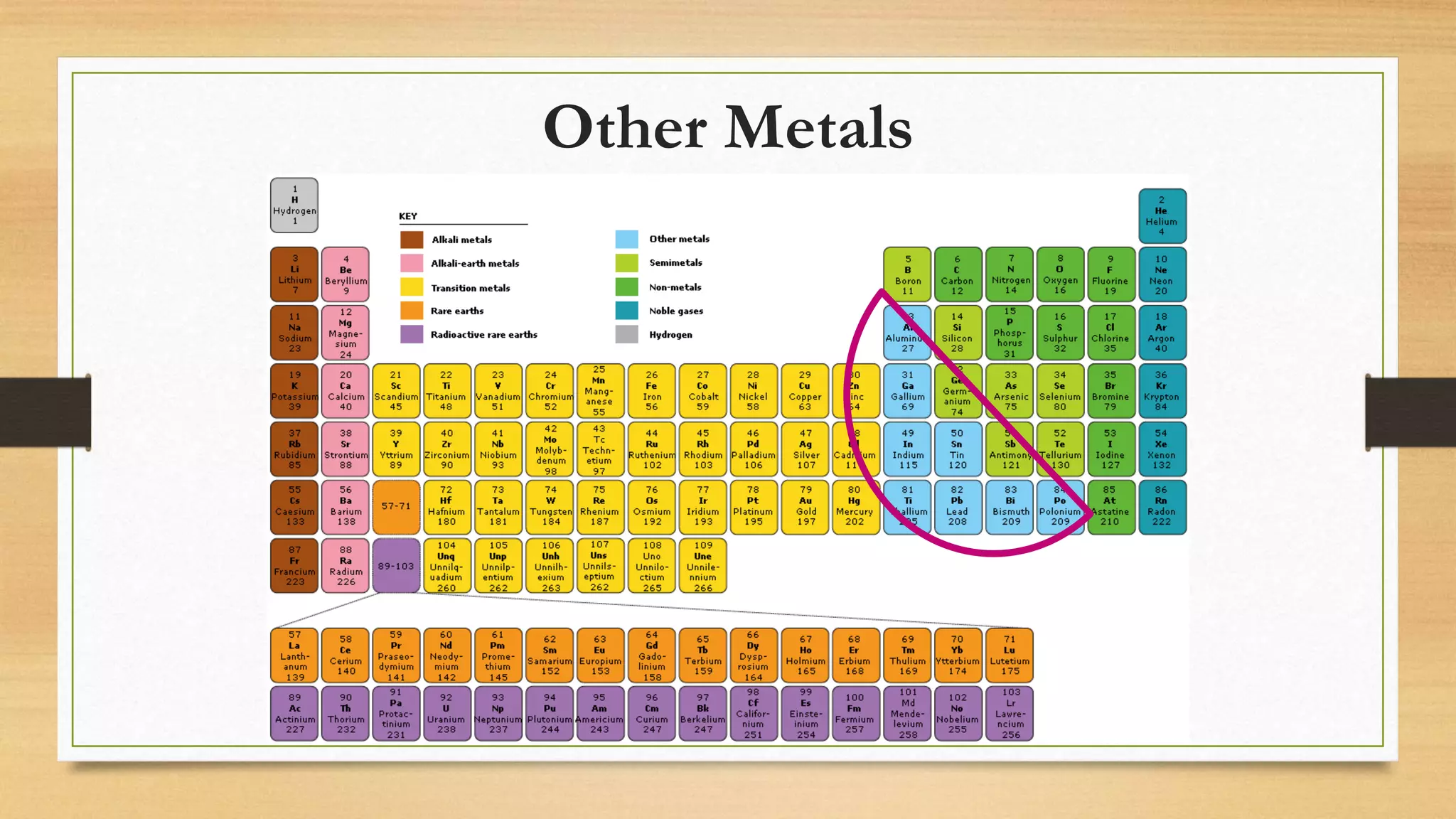
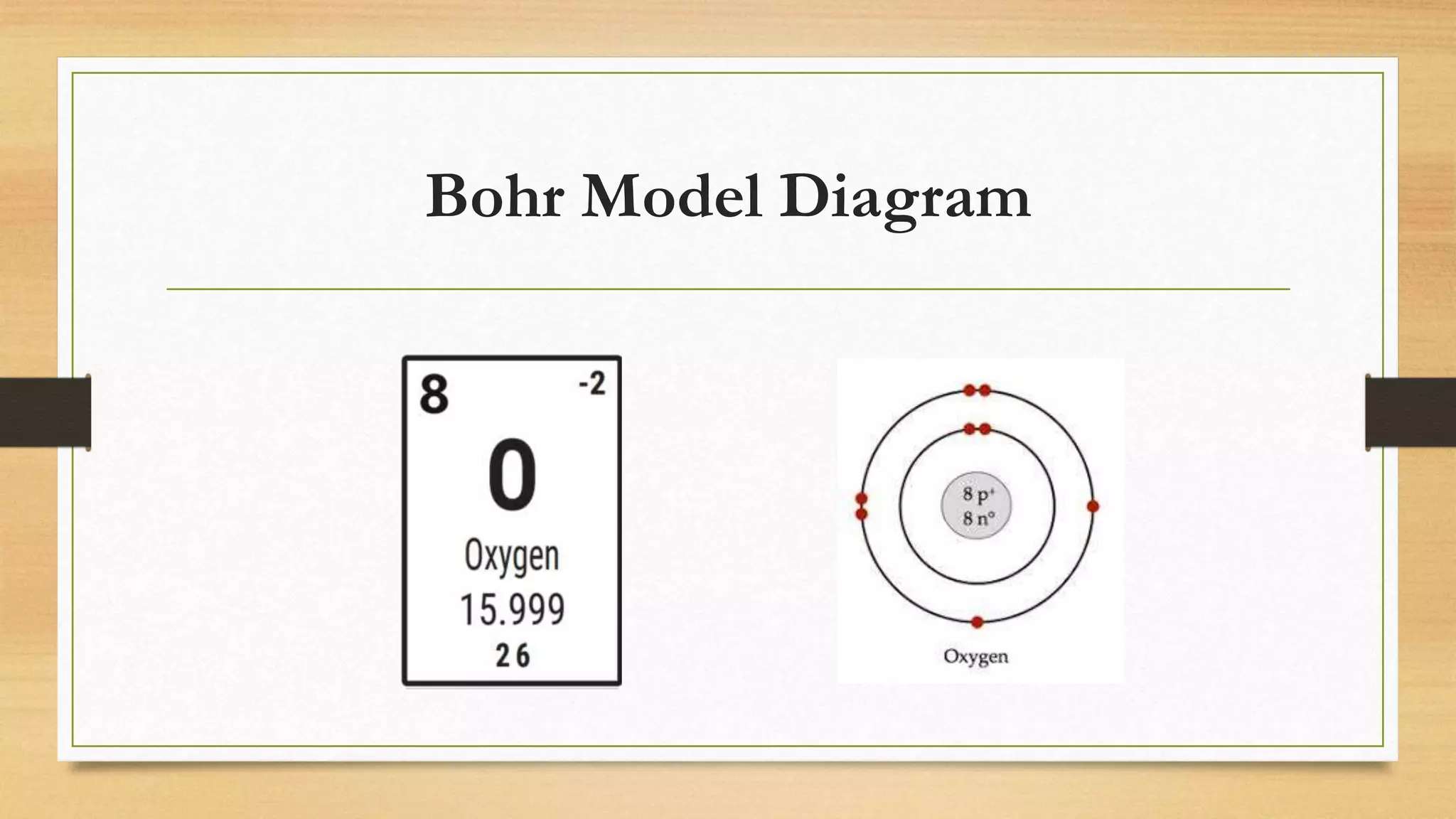
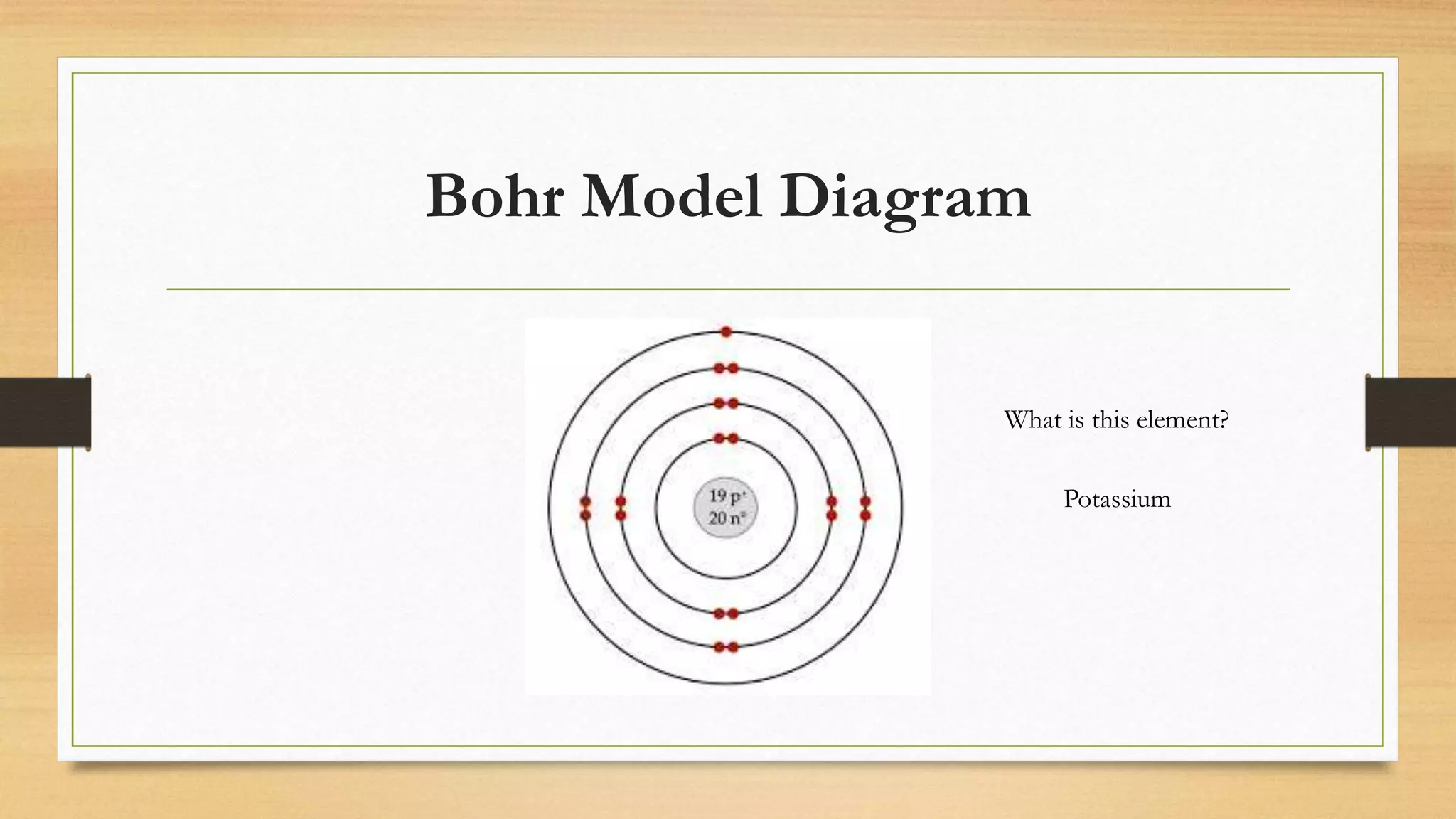
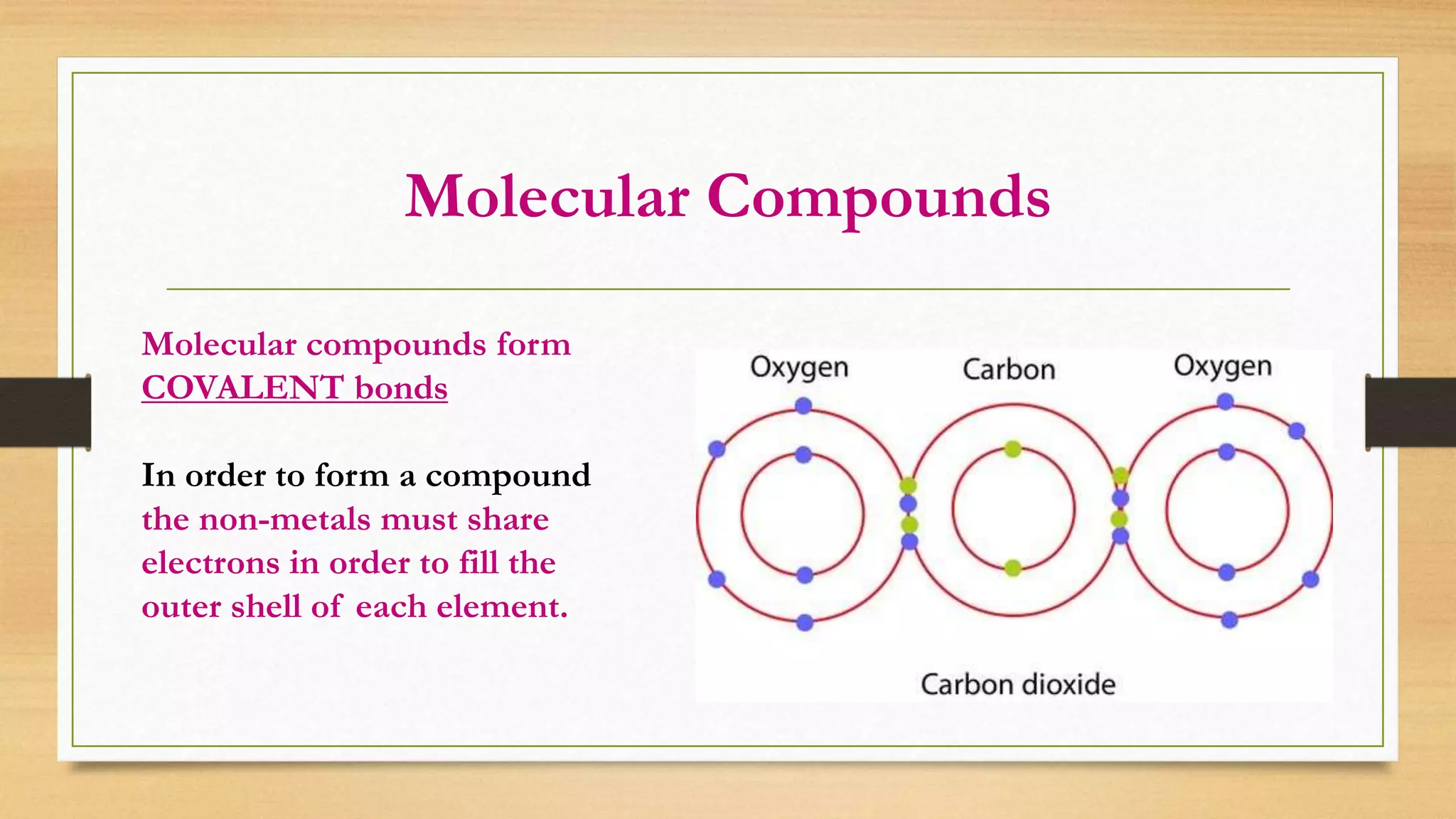
The document discusses matter and chemical change. It defines matter as anything that has mass and takes up space. Matter exists in different states - solid, liquid, and gas - which are determined by the motion and spacing of particles. Changes between states of matter involve adding or removing heat. Chemical and physical properties are also discussed, where chemical changes alter the chemical composition and physical changes do not. The document provides examples of physical and chemical properties and changes. Classifying matter as elements, compounds, or mixtures is also covered.