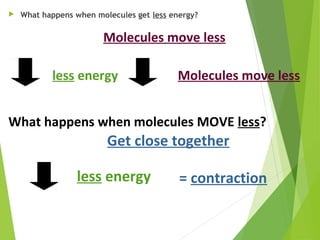
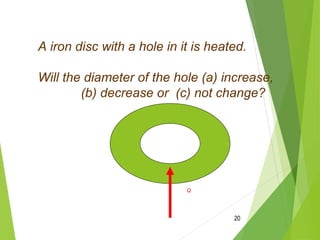
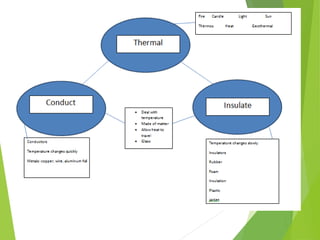
The document discusses the relationship between temperature, kinetic energy, and the thermal expansion and contraction of materials. It explains that when molecules are heated, they gain kinetic energy and move faster, causing them to spread farther apart and materials to expand in size. Conversely, when molecules cool down and lose kinetic energy, they move slower and get closer together, causing materials to contract. Examples provided include liquids expanding in a thermometer when heated and bridges being designed with expansion joints to accommodate changes in length due to varying temperatures.