Embed presentation

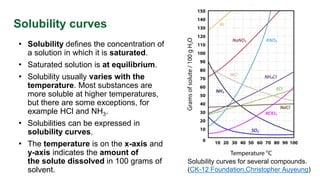
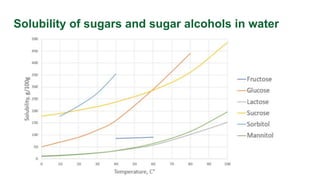




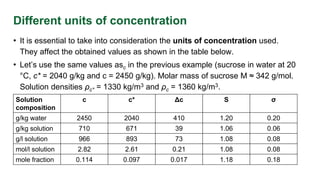

This document discusses supersaturation and solubility. It defines key terms like solubility, saturated solutions, and supersaturated solutions. Supersaturation occurs when the concentration of a solute is higher than the solubility limit. This drives crystallization. The document presents different ways to express the level of supersaturation, like concentration driving force and relative supersaturation. It provides an example calculation for sucrose in water. Finally, it notes that concentration units like g/kg solution can impact supersaturation values.








