Phase changes
•Download as PPT, PDF•
5 likes•18,191 views
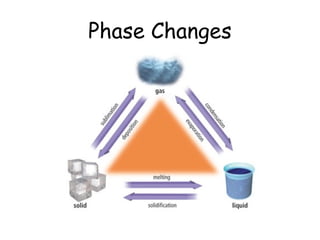
The document discusses the different phases of matter (solid, liquid, gas) and the phase changes between them. When energy is added to a solid, its bonds break and it melts into a liquid where particles can move freely. Adding more energy turns the liquid into a gas where particles move randomly. The phase changes - melting/freezing, vaporization/condensation, sublimation/deposition - describe the changes in particle arrangement that occur when energy is added or removed. Phase changes are classified as endothermic, where energy is absorbed during the change, or exothermic, where energy is released.
Report
Share
Report
Share
Recommended
Particle Nature of Matter

Matter
Characteristics and Properties of Matter
Properties of Solids
Properties of Liquids
Properties of Gases
Phase Changes of Matter
Percentage Composition

It is a powerpoint presentation that discusses about the lesson or topic: Percentage Composition. It also talks about the definition, concepts and examples about the Percentage Composition.
Recommended
Particle Nature of Matter

Matter
Characteristics and Properties of Matter
Properties of Solids
Properties of Liquids
Properties of Gases
Phase Changes of Matter
Percentage Composition

It is a powerpoint presentation that discusses about the lesson or topic: Percentage Composition. It also talks about the definition, concepts and examples about the Percentage Composition.
The Particle Nature of Matter

All matter is particulate in nature. This simply means that matter is made up of tiny bit bits material known as particle.
Elements and compounds grade 7 (first quarter)

K to 12 Science 7 - Instructional Material for Elements and Compounds
Colors of light (Grade 8)

Light behaviors
Properties of Light
Index of Refraction of the Material
Colors
Enhanced Science (physics)
Heat and temperature

Lesson taken from the book Science 7 of Phoenix Pub. House by Lilia G. Venco et.al, 2012.
Properties of Mixtures and Pure Substances

Matter can be classifies as a pure substance or mixture. They differ based from their properties and arrangement of particles.
The Kinetic Molecular Theory

Includes the principles of the KMT and their application to molecular behavior.
**More good stuff available at:
www.wsautter.com
and
http://www.youtube.com/results?search_query=wnsautter&aq=f
Properties of Liquids

Lecture Note Presentation on the properties of liquids. Designed for HS Chemistry & Earth Systems.
Redesign- Make a prediction relating heat to the movement of molecules.docx

Redesign: Make a prediction relating heat to the movement of molecules. How does this relate to fluorescent lighting? Gas Liquid
Solution
generally the ,
Solids - vibrational (and rotational); moving very slowly.
Liquids - vibrational, rotational, and translational; moving quickly.
Gases - vibrational, rotational, and translational; moving very quickly.
Explanation:
Particles are always moving. No matter what. There is a certain point where they move so slowly it is considered minuscule, but they are in fact moving.
Thus, in solids, liquids, and gases, you can expect particles to move in a certain trend. That trend is average kinetic energy of the particles and the forces holding them together (attraction).
Solids
When you have little to no heat, the particles are moving very slowly, so slowly that they atomic attractions result in bonds that are difficult to break without adding energy. At this stage, this is known as a solid.
In a solid, the particles are vibrating. Only some solids have the ability to rotate. They cannot translate regardless.
Liquids
When you add energy (heat) to a solid, you \"excite\" the particles so much that they break their bond; their attraction is too weak to hold them together - yet strong enough to keep a form
As a result, you get a liquid.
In a liquid, the particles are not only vibrating (their bonds are not present but they still vibrate), but they are also rotating and translating. The particles are moving much faster than in a solid.
Gases
When you add even more energy to the substance, you increase the kinetic energy of those particles so much, that they lose their state form, becoming a gas.
In a gas, particles have vibrational, rotational, and translational motion, allowing them to \"bounce\" off of one another. Particles in a gas state move much faster than a liquid.
Light is a form of energy that can be released by an atom. It is made up of many small particle-like packets that have energy and momentum but no mass. These particles, called light photons, are the most basic units of light.
When the atom gains or loses energy, the change is expressed by the movement of electrons. When something passes energy on to an atom -- heat , for example -- an electron may be temporarily boosted to a higher orbital (farther away from the nucleus). The electron only holds this position for a tiny fraction of a second; almost immediately, it is drawn back toward the nucleus, to its original orbital. As it returns to its original orbital, the electron releases the extra energy in the form of a photon, in some cases a light photon
.
More Related Content
What's hot
The Particle Nature of Matter

All matter is particulate in nature. This simply means that matter is made up of tiny bit bits material known as particle.
Elements and compounds grade 7 (first quarter)

K to 12 Science 7 - Instructional Material for Elements and Compounds
Colors of light (Grade 8)

Light behaviors
Properties of Light
Index of Refraction of the Material
Colors
Enhanced Science (physics)
Heat and temperature

Lesson taken from the book Science 7 of Phoenix Pub. House by Lilia G. Venco et.al, 2012.
Properties of Mixtures and Pure Substances

Matter can be classifies as a pure substance or mixture. They differ based from their properties and arrangement of particles.
The Kinetic Molecular Theory

Includes the principles of the KMT and their application to molecular behavior.
**More good stuff available at:
www.wsautter.com
and
http://www.youtube.com/results?search_query=wnsautter&aq=f
Properties of Liquids

Lecture Note Presentation on the properties of liquids. Designed for HS Chemistry & Earth Systems.
What's hot (20)
Similar to Phase changes
Redesign- Make a prediction relating heat to the movement of molecules.docx

Redesign: Make a prediction relating heat to the movement of molecules. How does this relate to fluorescent lighting? Gas Liquid
Solution
generally the ,
Solids - vibrational (and rotational); moving very slowly.
Liquids - vibrational, rotational, and translational; moving quickly.
Gases - vibrational, rotational, and translational; moving very quickly.
Explanation:
Particles are always moving. No matter what. There is a certain point where they move so slowly it is considered minuscule, but they are in fact moving.
Thus, in solids, liquids, and gases, you can expect particles to move in a certain trend. That trend is average kinetic energy of the particles and the forces holding them together (attraction).
Solids
When you have little to no heat, the particles are moving very slowly, so slowly that they atomic attractions result in bonds that are difficult to break without adding energy. At this stage, this is known as a solid.
In a solid, the particles are vibrating. Only some solids have the ability to rotate. They cannot translate regardless.
Liquids
When you add energy (heat) to a solid, you \"excite\" the particles so much that they break their bond; their attraction is too weak to hold them together - yet strong enough to keep a form
As a result, you get a liquid.
In a liquid, the particles are not only vibrating (their bonds are not present but they still vibrate), but they are also rotating and translating. The particles are moving much faster than in a solid.
Gases
When you add even more energy to the substance, you increase the kinetic energy of those particles so much, that they lose their state form, becoming a gas.
In a gas, particles have vibrational, rotational, and translational motion, allowing them to \"bounce\" off of one another. Particles in a gas state move much faster than a liquid.
Light is a form of energy that can be released by an atom. It is made up of many small particle-like packets that have energy and momentum but no mass. These particles, called light photons, are the most basic units of light.
When the atom gains or loses energy, the change is expressed by the movement of electrons. When something passes energy on to an atom -- heat , for example -- an electron may be temporarily boosted to a higher orbital (farther away from the nucleus). The electron only holds this position for a tiny fraction of a second; almost immediately, it is drawn back toward the nucleus, to its original orbital. As it returns to its original orbital, the electron releases the extra energy in the form of a photon, in some cases a light photon
.
Matter, Properties, & Phases

Lesson teaches students about Matter (grades 6-8) & talks about the atomic models & the history behind the way it has been established through physics.
This Lesson Also Includes:
1. Physical & Chemical Changes
2. States of Matter & Phase Changes in Matter
3. Molecular Movements in a Solid, Liquid, & Vapor
Particulate nature of matter

Describe the structure of solids, liquids and gases in terms of particle separation, arrangement and types of motion
State the distinguishing properties of solids, liquids and gases
• Describe and explain diffusion (1 core)
• Describe evidence for the movement of particles in gases and liquids (1 core)
Describe changes of state in terms of melting, boiling, evaporation, freezing, condensation and sublimation
Explain changes of state in terms of the kinetic
theory
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

Anything which occupies space or volume, has mass and can be perceived by our senses is called matter.
Similar to Phase changes (20)
Redesign- Make a prediction relating heat to the movement of molecules.docx

Redesign- Make a prediction relating heat to the movement of molecules.docx
PHYSICAL CHANGES IN TERMS OF THE arrangement and.pptx

PHYSICAL CHANGES IN TERMS OF THE arrangement and.pptx
0708_matter and classification,states, phases of matter.ppt

0708_matter and classification,states, phases of matter.ppt
2.1 form 3 simple kinetic molecular model of matter

2.1 form 3 simple kinetic molecular model of matter
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

ICSE Class VIII Chemistry Matter Around Us - TopperLearning
More from Grover Cleveland Middle School
More from Grover Cleveland Middle School (20)
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Ocean acidification will-the_reef_survive-non-narrated[1]
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Ocean acidification will-the_reef_survive-non-narrated[1]
Recently uploaded
PHP Frameworks: I want to break free (IPC Berlin 2024)

In this presentation, we examine the challenges and limitations of relying too heavily on PHP frameworks in web development. We discuss the history of PHP and its frameworks to understand how this dependence has evolved. The focus will be on providing concrete tips and strategies to reduce reliance on these frameworks, based on real-world examples and practical considerations. The goal is to equip developers with the skills and knowledge to create more flexible and future-proof web applications. We'll explore the importance of maintaining autonomy in a rapidly changing tech landscape and how to make informed decisions in PHP development.
This talk is aimed at encouraging a more independent approach to using PHP frameworks, moving towards a more flexible and future-proof approach to PHP development.
JMeter webinar - integration with InfluxDB and Grafana

Watch this recorded webinar about real-time monitoring of application performance. See how to integrate Apache JMeter, the open-source leader in performance testing, with InfluxDB, the open-source time-series database, and Grafana, the open-source analytics and visualization application.
In this webinar, we will review the benefits of leveraging InfluxDB and Grafana when executing load tests and demonstrate how these tools are used to visualize performance metrics.
Length: 30 minutes
Session Overview
-------------------------------------------
During this webinar, we will cover the following topics while demonstrating the integrations of JMeter, InfluxDB and Grafana:
- What out-of-the-box solutions are available for real-time monitoring JMeter tests?
- What are the benefits of integrating InfluxDB and Grafana into the load testing stack?
- Which features are provided by Grafana?
- Demonstration of InfluxDB and Grafana using a practice web application
To view the webinar recording, go to:
https://www.rttsweb.com/jmeter-integration-webinar
How world-class product teams are winning in the AI era by CEO and Founder, P...

How world-class product teams are winning in the AI era by CEO and Founder, Product School
Knowledge engineering: from people to machines and back

Keynote at the 21st European Semantic Web Conference
GraphRAG is All You need? LLM & Knowledge Graph

Guy Korland, CEO and Co-founder of FalkorDB, will review two articles on the integration of language models with knowledge graphs.
1. Unifying Large Language Models and Knowledge Graphs: A Roadmap.
https://arxiv.org/abs/2306.08302
2. Microsoft Research's GraphRAG paper and a review paper on various uses of knowledge graphs:
https://www.microsoft.com/en-us/research/blog/graphrag-unlocking-llm-discovery-on-narrative-private-data/
Transcript: Selling digital books in 2024: Insights from industry leaders - T...

The publishing industry has been selling digital audiobooks and ebooks for over a decade and has found its groove. What’s changed? What has stayed the same? Where do we go from here? Join a group of leading sales peers from across the industry for a conversation about the lessons learned since the popularization of digital books, best practices, digital book supply chain management, and more.
Link to video recording: https://bnctechforum.ca/sessions/selling-digital-books-in-2024-insights-from-industry-leaders/
Presented by BookNet Canada on May 28, 2024, with support from the Department of Canadian Heritage.
GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using Deplo...

Effective Application Security in Software Delivery lifecycle using Deployment Firewall and DBOM
The modern software delivery process (or the CI/CD process) includes many tools, distributed teams, open-source code, and cloud platforms. Constant focus on speed to release software to market, along with the traditional slow and manual security checks has caused gaps in continuous security as an important piece in the software supply chain. Today organizations feel more susceptible to external and internal cyber threats due to the vast attack surface in their applications supply chain and the lack of end-to-end governance and risk management.
The software team must secure its software delivery process to avoid vulnerability and security breaches. This needs to be achieved with existing tool chains and without extensive rework of the delivery processes. This talk will present strategies and techniques for providing visibility into the true risk of the existing vulnerabilities, preventing the introduction of security issues in the software, resolving vulnerabilities in production environments quickly, and capturing the deployment bill of materials (DBOM).
Speakers:
Bob Boule
Robert Boule is a technology enthusiast with PASSION for technology and making things work along with a knack for helping others understand how things work. He comes with around 20 years of solution engineering experience in application security, software continuous delivery, and SaaS platforms. He is known for his dynamic presentations in CI/CD and application security integrated in software delivery lifecycle.
Gopinath Rebala
Gopinath Rebala is the CTO of OpsMx, where he has overall responsibility for the machine learning and data processing architectures for Secure Software Delivery. Gopi also has a strong connection with our customers, leading design and architecture for strategic implementations. Gopi is a frequent speaker and well-known leader in continuous delivery and integrating security into software delivery.
AI for Every Business: Unlocking Your Product's Universal Potential by VP of ...

AI for Every Business: Unlocking Your Product's Universal Potential by VP of Product, Slack
DevOps and Testing slides at DASA Connect

My and Rik Marselis slides at 30.5.2024 DASA Connect conference. We discuss about what is testing, then what is agile testing and finally what is Testing in DevOps. Finally we had lovely workshop with the participants trying to find out different ways to think about quality and testing in different parts of the DevOps infinity loop.
GenAISummit 2024 May 28 Sri Ambati Keynote: AGI Belongs to The Community in O...

“AGI should be open source and in the public domain at the service of humanity and the planet.”
FIDO Alliance Osaka Seminar: The WebAuthn API and Discoverable Credentials.pdf

FIDO Alliance Osaka Seminar
State of ICS and IoT Cyber Threat Landscape Report 2024 preview

The IoT and OT threat landscape report has been prepared by the Threat Research Team at Sectrio using data from Sectrio, cyber threat intelligence farming facilities spread across over 85 cities around the world. In addition, Sectrio also runs AI-based advanced threat and payload engagement facilities that serve as sinks to attract and engage sophisticated threat actors, and newer malware including new variants and latent threats that are at an earlier stage of development.
The latest edition of the OT/ICS and IoT security Threat Landscape Report 2024 also covers:
State of global ICS asset and network exposure
Sectoral targets and attacks as well as the cost of ransom
Global APT activity, AI usage, actor and tactic profiles, and implications
Rise in volumes of AI-powered cyberattacks
Major cyber events in 2024
Malware and malicious payload trends
Cyberattack types and targets
Vulnerability exploit attempts on CVEs
Attacks on counties – USA
Expansion of bot farms – how, where, and why
In-depth analysis of the cyber threat landscape across North America, South America, Europe, APAC, and the Middle East
Why are attacks on smart factories rising?
Cyber risk predictions
Axis of attacks – Europe
Systemic attacks in the Middle East
Download the full report from here:
https://sectrio.com/resources/ot-threat-landscape-reports/sectrio-releases-ot-ics-and-iot-security-threat-landscape-report-2024/
ODC, Data Fabric and Architecture User Group

Let's dive deeper into the world of ODC! Ricardo Alves (OutSystems) will join us to tell all about the new Data Fabric. After that, Sezen de Bruijn (OutSystems) will get into the details on how to best design a sturdy architecture within ODC.
The Art of the Pitch: WordPress Relationships and Sales

Clients don’t know what they don’t know. What web solutions are right for them? How does WordPress come into the picture? How do you make sure you understand scope and timeline? What do you do if sometime changes?
All these questions and more will be explored as we talk about matching clients’ needs with what your agency offers without pulling teeth or pulling your hair out. Practical tips, and strategies for successful relationship building that leads to closing the deal.
Slack (or Teams) Automation for Bonterra Impact Management (fka Social Soluti...

Sidekick Solutions uses Bonterra Impact Management (fka Social Solutions Apricot) and automation solutions to integrate data for business workflows.
We believe integration and automation are essential to user experience and the promise of efficient work through technology. Automation is the critical ingredient to realizing that full vision. We develop integration products and services for Bonterra Case Management software to support the deployment of automations for a variety of use cases.
This video focuses on the notifications, alerts, and approval requests using Slack for Bonterra Impact Management. The solutions covered in this webinar can also be deployed for Microsoft Teams.
Interested in deploying notification automations for Bonterra Impact Management? Contact us at sales@sidekicksolutionsllc.com to discuss next steps.
Dev Dives: Train smarter, not harder – active learning and UiPath LLMs for do...

💥 Speed, accuracy, and scaling – discover the superpowers of GenAI in action with UiPath Document Understanding and Communications Mining™:
See how to accelerate model training and optimize model performance with active learning
Learn about the latest enhancements to out-of-the-box document processing – with little to no training required
Get an exclusive demo of the new family of UiPath LLMs – GenAI models specialized for processing different types of documents and messages
This is a hands-on session specifically designed for automation developers and AI enthusiasts seeking to enhance their knowledge in leveraging the latest intelligent document processing capabilities offered by UiPath.
Speakers:
👨🏫 Andras Palfi, Senior Product Manager, UiPath
👩🏫 Lenka Dulovicova, Product Program Manager, UiPath
Recently uploaded (20)
PHP Frameworks: I want to break free (IPC Berlin 2024)

PHP Frameworks: I want to break free (IPC Berlin 2024)
JMeter webinar - integration with InfluxDB and Grafana

JMeter webinar - integration with InfluxDB and Grafana
How world-class product teams are winning in the AI era by CEO and Founder, P...

How world-class product teams are winning in the AI era by CEO and Founder, P...
Knowledge engineering: from people to machines and back

Knowledge engineering: from people to machines and back
Transcript: Selling digital books in 2024: Insights from industry leaders - T...

Transcript: Selling digital books in 2024: Insights from industry leaders - T...
GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using Deplo...

GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using Deplo...
AI for Every Business: Unlocking Your Product's Universal Potential by VP of ...

AI for Every Business: Unlocking Your Product's Universal Potential by VP of ...
FIDO Alliance Osaka Seminar: FIDO Security Aspects.pdf

FIDO Alliance Osaka Seminar: FIDO Security Aspects.pdf
GenAISummit 2024 May 28 Sri Ambati Keynote: AGI Belongs to The Community in O...

GenAISummit 2024 May 28 Sri Ambati Keynote: AGI Belongs to The Community in O...
FIDO Alliance Osaka Seminar: The WebAuthn API and Discoverable Credentials.pdf

FIDO Alliance Osaka Seminar: The WebAuthn API and Discoverable Credentials.pdf
State of ICS and IoT Cyber Threat Landscape Report 2024 preview

State of ICS and IoT Cyber Threat Landscape Report 2024 preview
The Art of the Pitch: WordPress Relationships and Sales

The Art of the Pitch: WordPress Relationships and Sales
Slack (or Teams) Automation for Bonterra Impact Management (fka Social Soluti...

Slack (or Teams) Automation for Bonterra Impact Management (fka Social Soluti...
Dev Dives: Train smarter, not harder – active learning and UiPath LLMs for do...

Dev Dives: Train smarter, not harder – active learning and UiPath LLMs for do...
Phase changes
- 2. Do Now: What are the names for the phases that matter goes through when changing from solid to liquid to gas, and then back again?
- 3. States of Matter - Review By adding energy, usually in the form of heat, the bonds that hold a solid together can slowly break apart. The object goes from one state, or phase, to another. These changes are called PHASE CHANGES.
- 4. Solids A solid is an ordered arrangement of particles that have very little movement. The particles vibrate back and forth but remain closely attracted to each other.
- 5. Liquids A liquid is an arrangement of less ordered particles that have gained energy and can move about more freely. Their attraction is less than a solid.
- 6. Compare the two states:
- 7. Gases By adding energy, solids can change from an ordered arrangement to a less ordered arrangement (liquid), and finally to a very random arrangement of the particles (the gas phase).
- 8. Each phase change has a specific name, based on what is happening to the particles of matter.
- 9. Melting and Freezing The solid, ordered molecules gain energy, collide more, and become less ordered. The less ordered liquid molecules loose energy, slow down, and become more ordered.
- 10. Condensation and Vaporization The less ordered gas molecules loose energy, slow down, and become more ordered. The more ordered liquid molecules gain energy, speed up, and become less ordered. Evaporation only occurs at the surface of a liquid. Boiling occurs throughout the liquid.
- 11. Sublimation and Deposition The more ordered solid gains energy, the molecules speed up and become less ordered gas. Liquid phase is bypassed. The less ordered gas looses energy, molecules slow down, and become a solid (frost). Liquid phase is bypassed
- 12. Phase changes are classified as one of two types: 1. ENDOTHERMIC – The object absorbs energy from the surroundings. Think of ENDO as “entering”: energy comes into the object. What is happening to the particles? 2. EXOTHERMIC – The object releases energy to the surroundings. Think of EXO as “exiting”: energy goes out of the object. What is happening to the particles now?
- 13. Now we can complete the remaining two columns of the study guide.