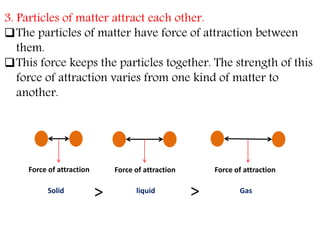
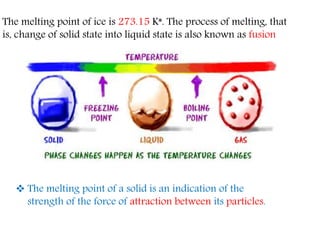
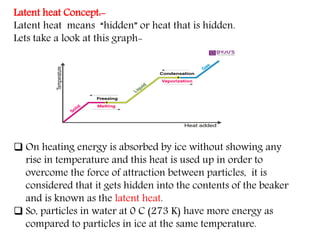
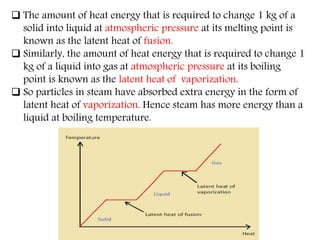
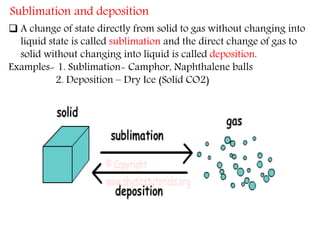
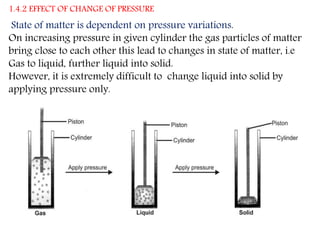
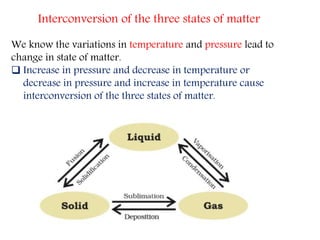
This document provides an overview of matter and its different states. It discusses that matter is made up of particles that are continuously moving, have space between them, and attract each other. The three main states of matter are solids, liquids, and gases, which are determined by the characteristics of the particles. Changes in temperature and pressure can cause matter to change states through processes like melting, boiling, sublimation, and deposition. Evaporation is also explained as the process where liquid particles at the surface gain energy and change to vapor without reaching the boiling point. In summary, the document covers the basic physical properties and behavior of different forms of matter.