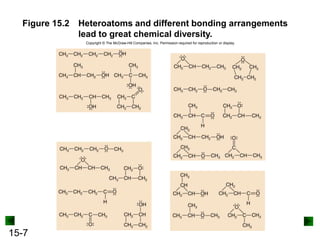
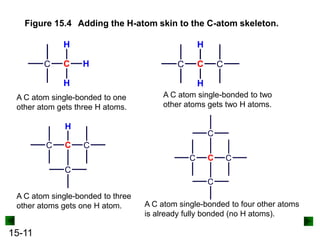
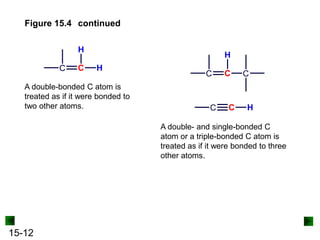
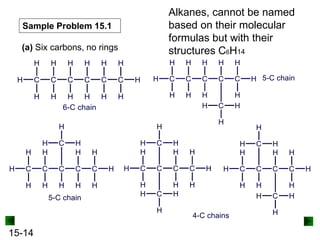
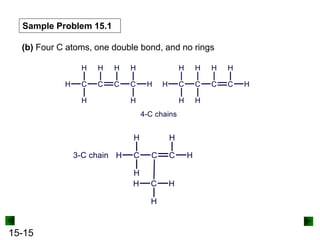
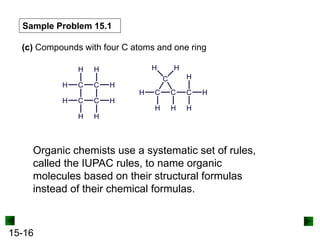
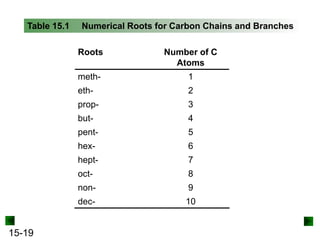
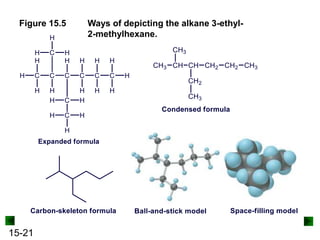
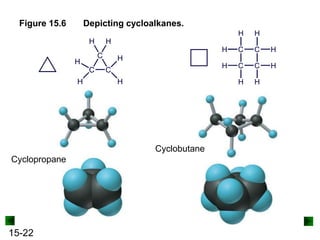
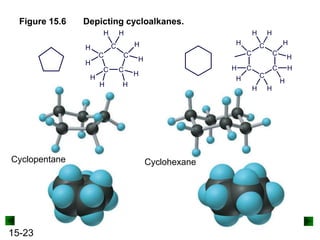
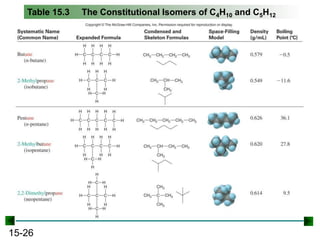
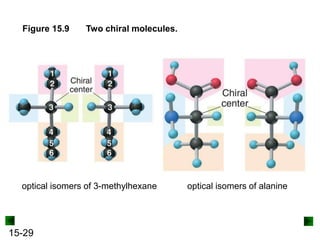
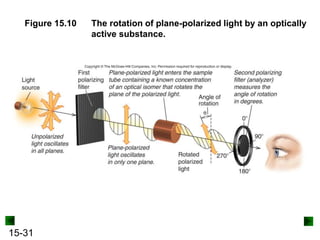
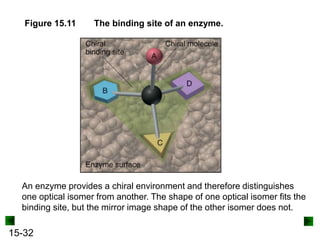
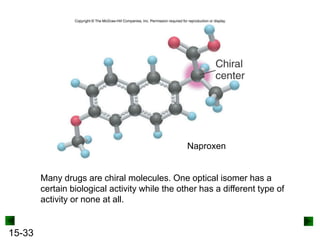
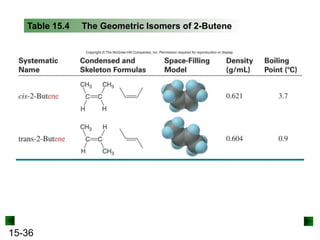
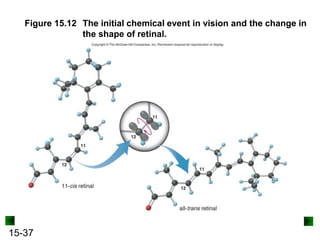
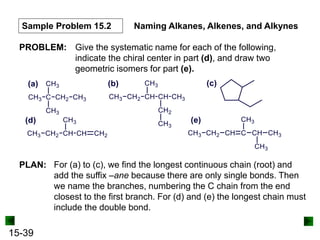
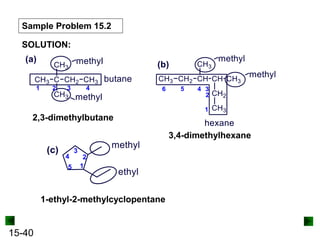
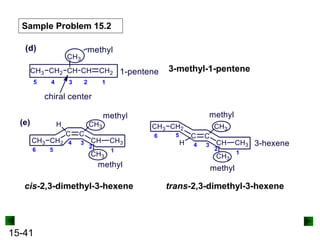
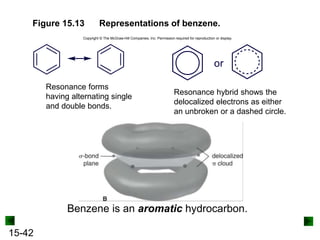
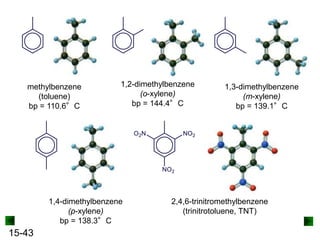
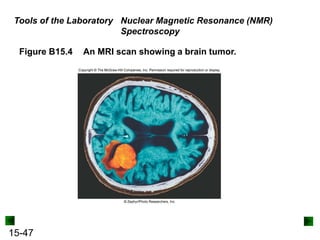
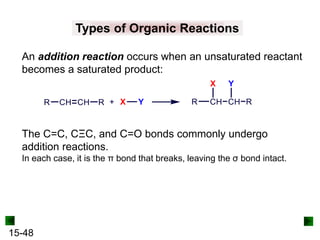
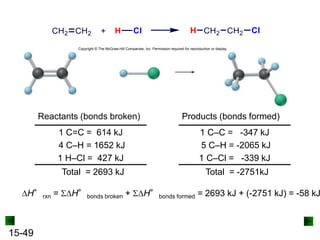
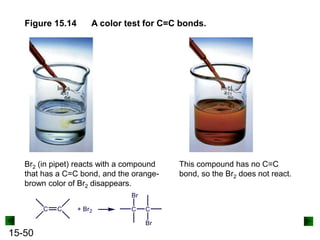
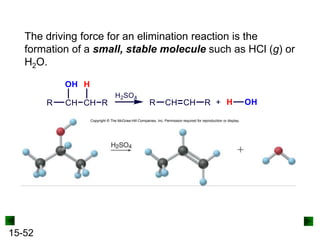
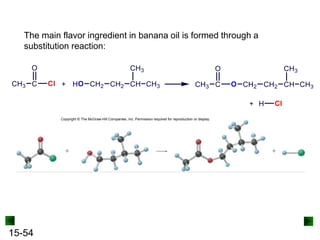
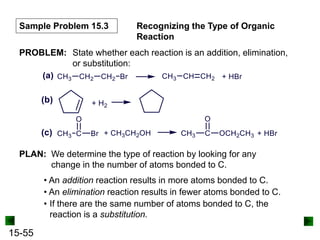
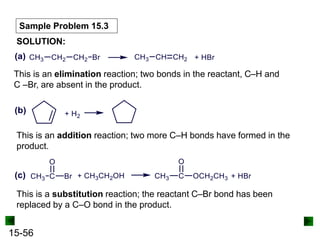
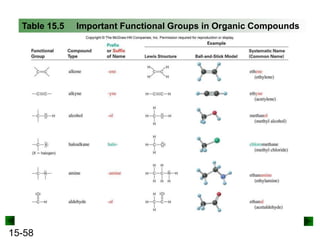
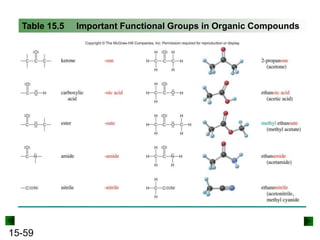
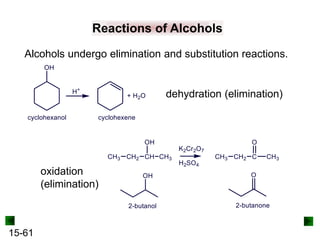
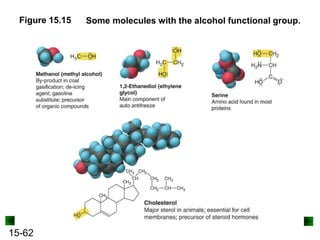
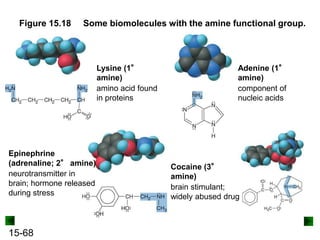
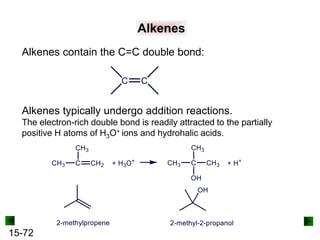
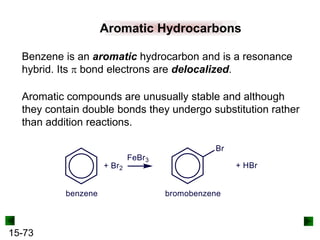
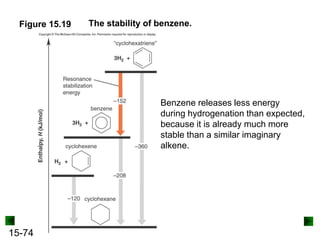
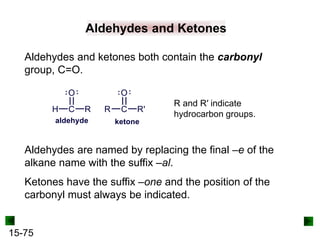
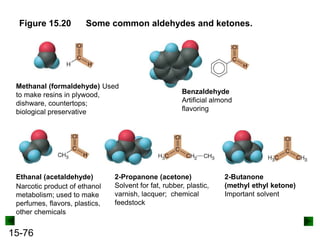
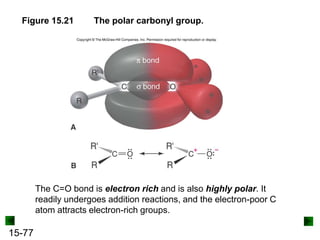
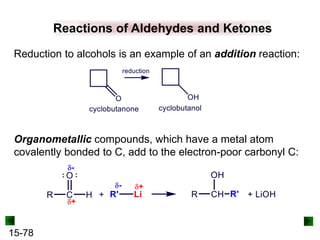
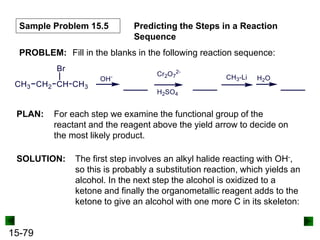
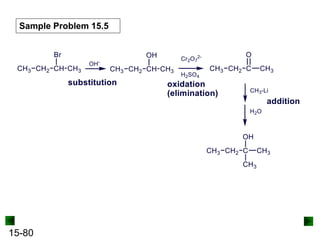
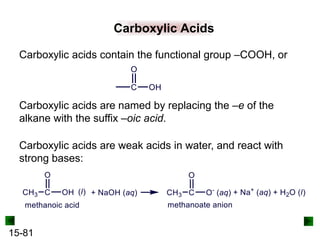
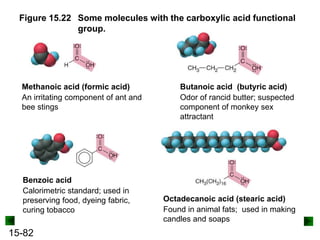
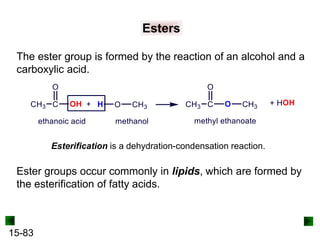
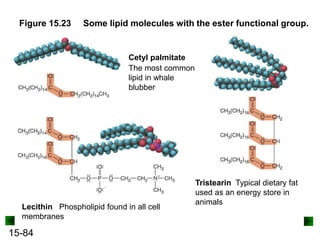
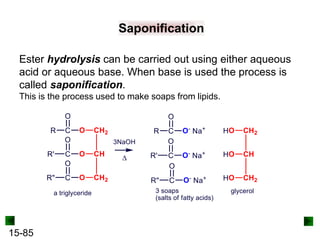
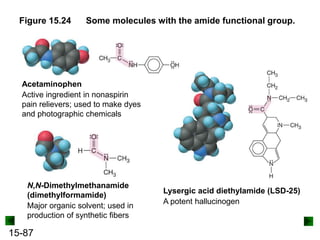
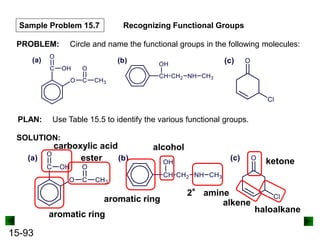
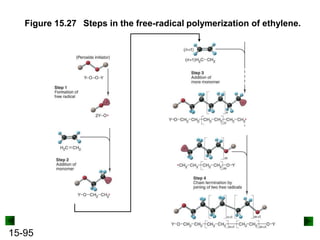
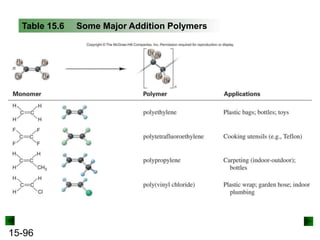
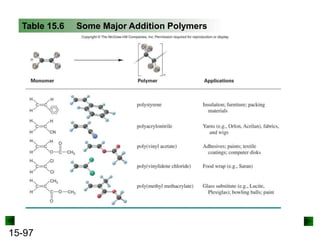
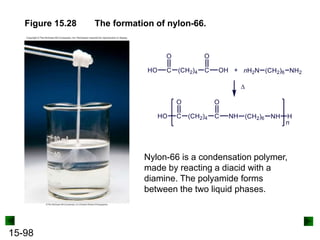
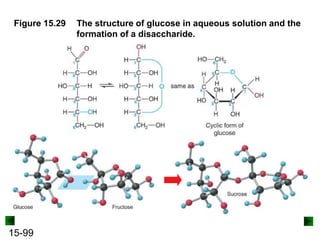
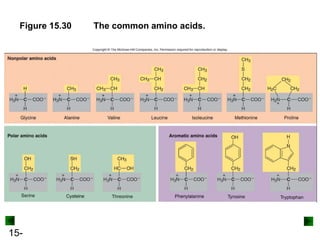
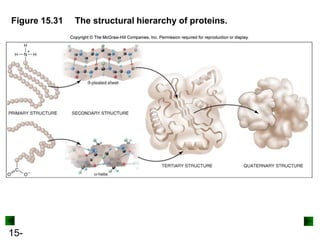
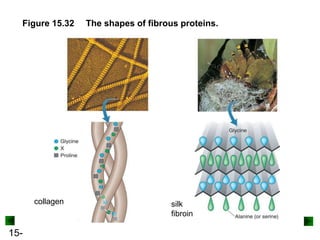
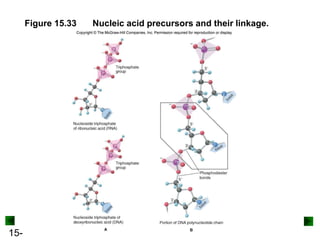
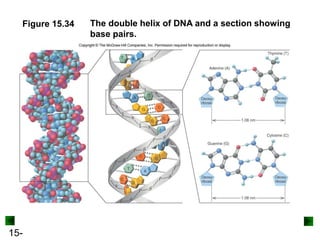
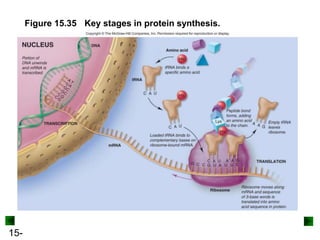
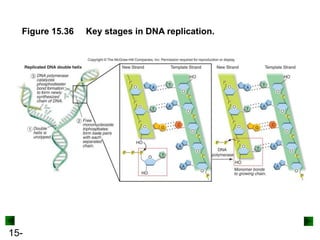
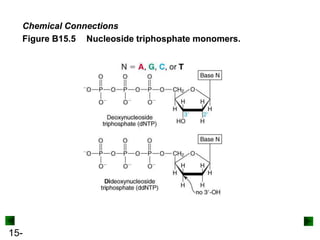
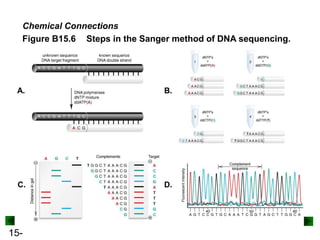
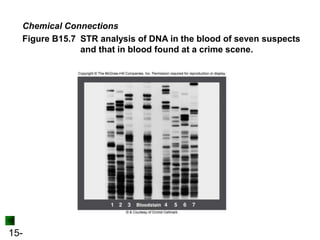
This chapter discusses organic compounds and carbon chemistry. It covers the properties of carbon that allow it to form large, complex molecules through catenation. The structures and classes of hydrocarbons like alkanes, alkenes, and alkynes are examined. Important classes of organic reactions such as addition, elimination, and substitution are described. The chapter also explores functional groups and how they determine a molecule's reactivity and properties. Alcohols are highlighted as one functional group and their naming conventions and intermolecular hydrogen bonding are discussed.


![15-3
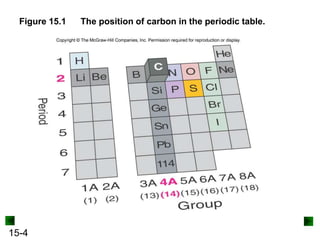
Bonding Properties of Carbon
• Carbon forms covalent bonds in all its elemental forms
and compounds.
– The ground state electron configuration of C is [He]2s22p2; the
formation of carbon ions is therefore energetically unfavorable.
– C has an electronegativity of 2.5, which is midway between that
of most metals and nonmetals. C prefers to share electrons.
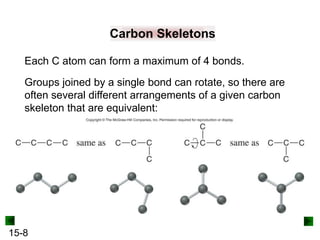
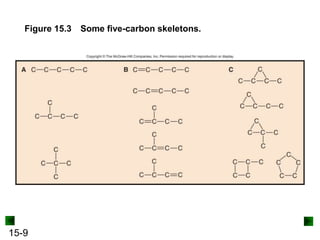
• Carbon exhibits catenation, the ability to bond to itself
and form stable chain, ring, and branched compounds.
– The small size of the C atom allows it to form short, strong
bonds.
– The tetrahedral shape of the C atom allows catenation.](https://image.slidesharecdn.com/organicchemistry-230212132937-6ca0bac4/85/Organic-Chemistry-ppt-3-320.jpg)