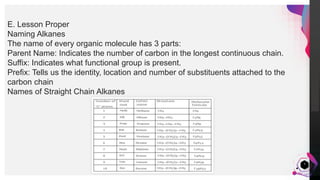
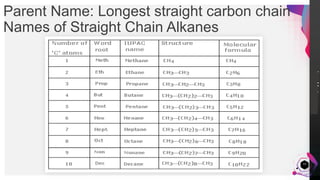
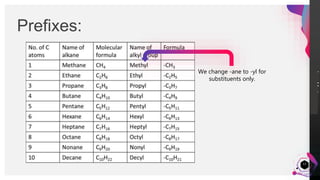
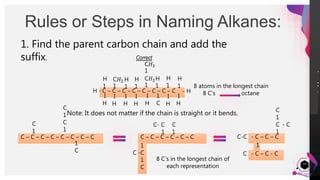
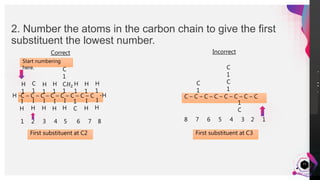
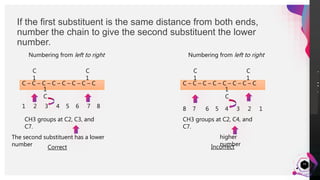
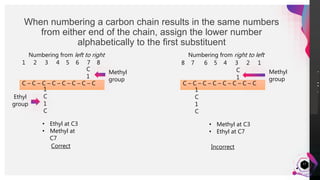
This document outlines a lesson plan on naming alkanes. The objectives are to identify organic molecule parts, name straight chain alkanes, discuss alkane naming steps, and participate in classroom activities. The lesson proper explains that alkane names have three parts - the parent name indicating the longest carbon chain, a suffix of "-ane" for alkanes, and prefixes for substituents. It provides examples of naming straight chain alkanes and lists the steps to name alkanes, including numbering carbons, naming substituents, and combining all parts of the name. An assessment with example alkane structures is included to check student understanding.