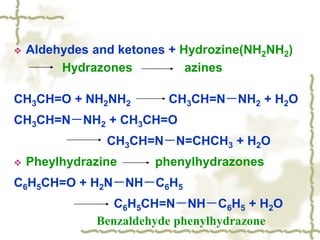
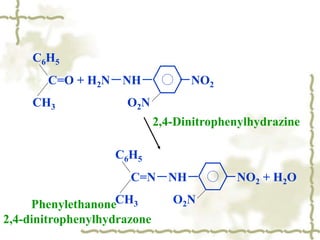
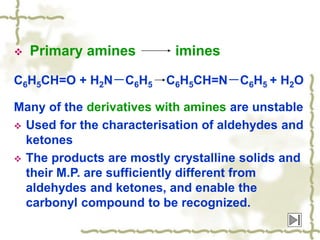
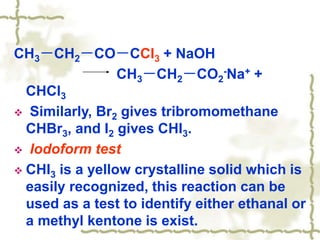
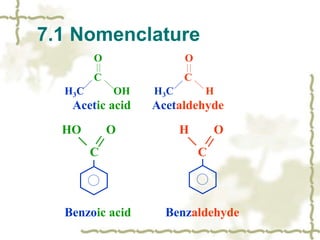
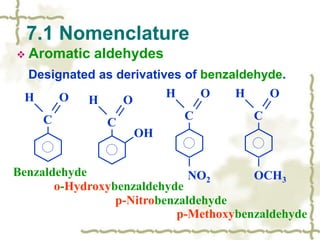
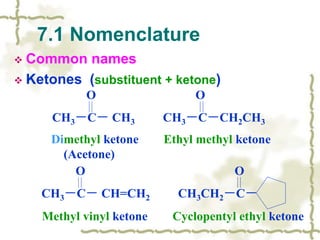
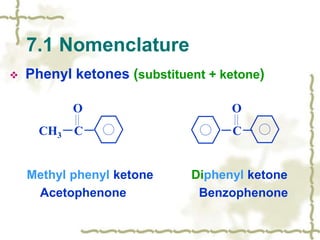
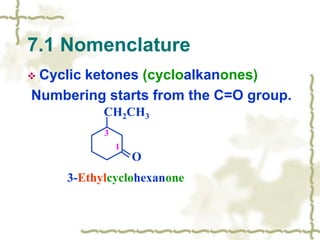
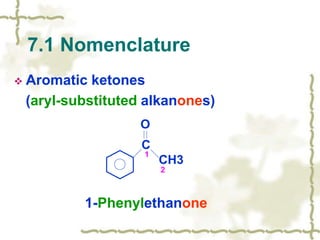
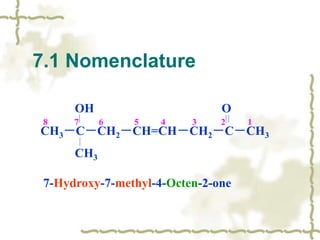
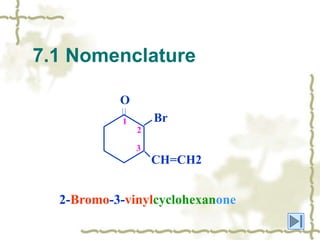
Chapter 7 discusses aldehydes and ketones, emphasizing the significance of the carbonyl group (C=O) in organic chemistry and its various applications in industries and biological functions. It covers nomenclature, structures, chemical properties, and reactions, including oxidation and nucleophilic additions, detailing how these compounds are named and their physical properties. Additionally, it highlights the importance of these compounds in creating flavors, aromas, and their use as reagents and solvents in chemical synthesis.
























![SO3
2- as nucleophilic reagent
Theoretically, the SO3
2- reacts more rapidly,
but HSO3
- is a weak acid
HSO3
- + H2O SO3
2- + H3O+
Ka = 1.1×10-7
Usually, [HSO3
-] is much greater than [SO3
2-]
O CH3 O CH3
-O-S C=O O=S-C-O -
O CH3 O CH3](https://image.slidesharecdn.com/oc-ch7aldehydeandketone-220920120934-76decc99/85/oc-ch7-Aldehyde-and-ketone-ppt-33-320.jpg)