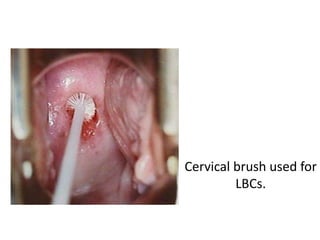
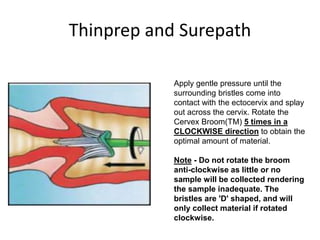
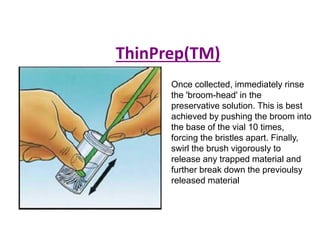
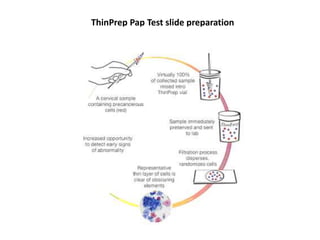
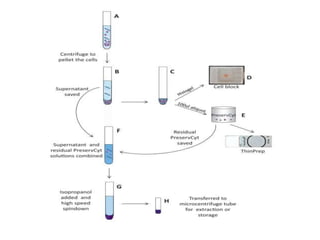
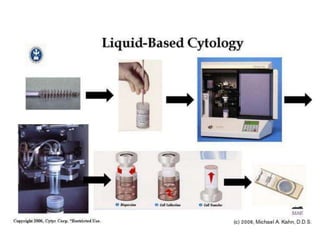
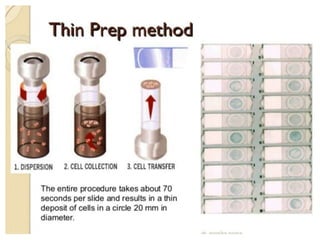
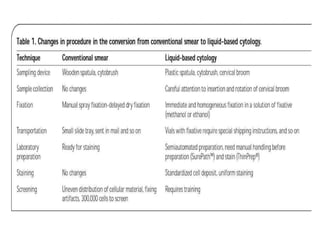
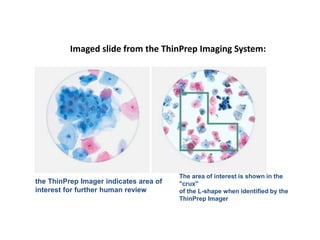
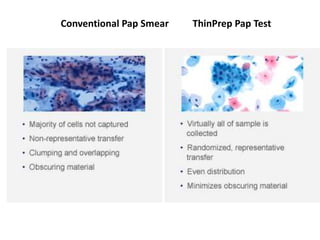
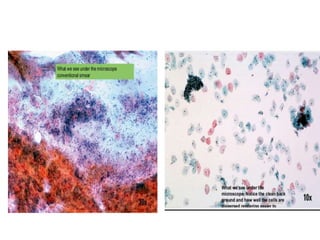
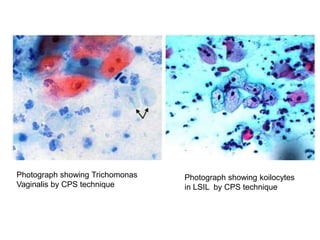
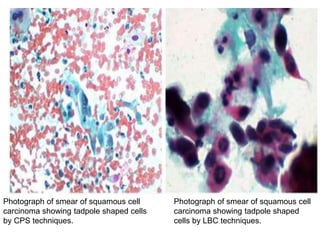
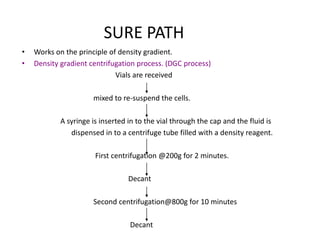
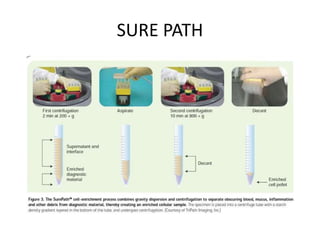
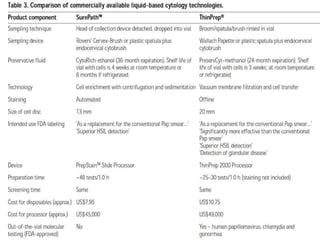
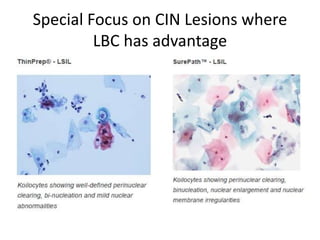
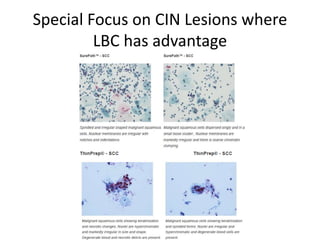
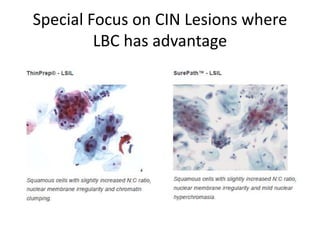
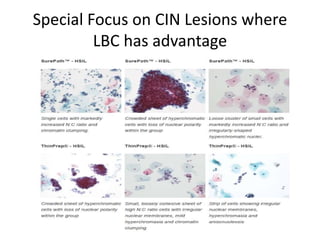
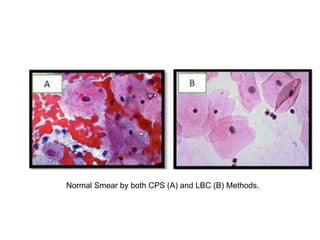
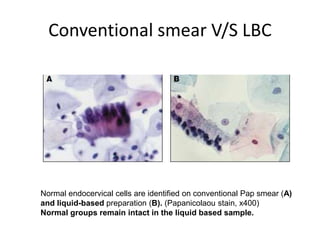
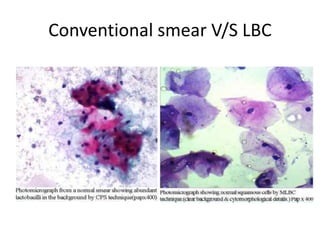
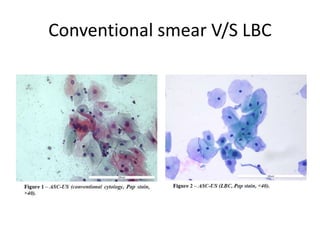
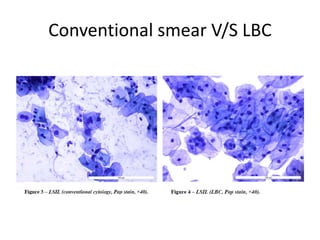
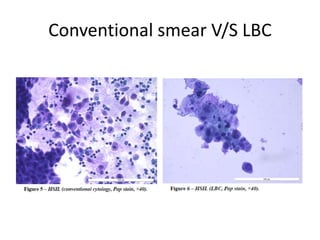
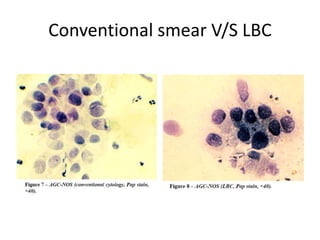
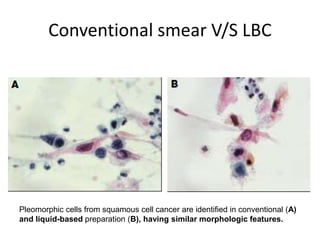
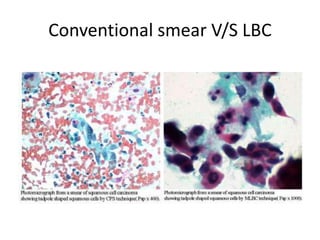
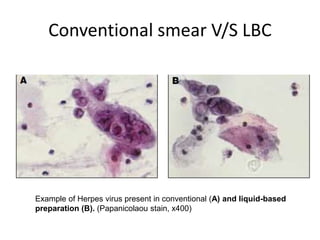
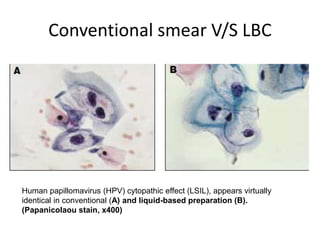
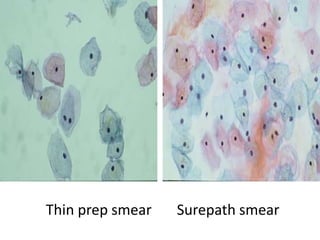
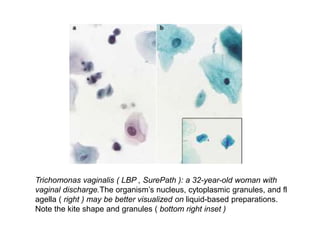
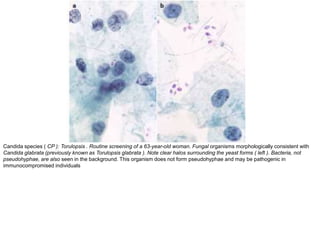
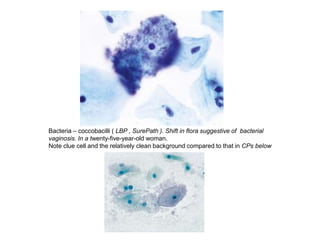
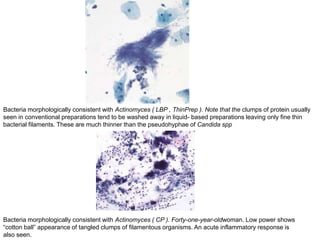
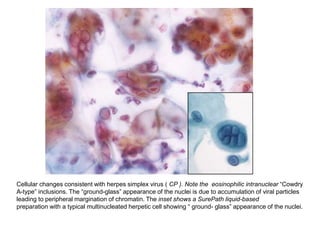
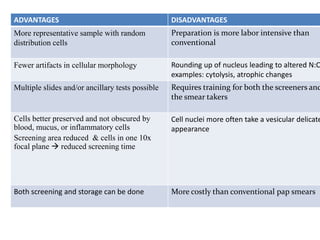
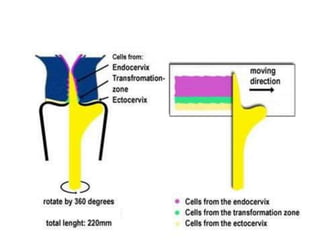
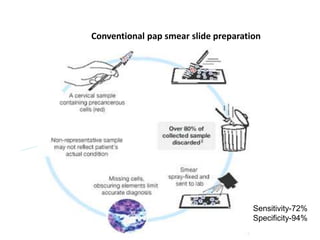
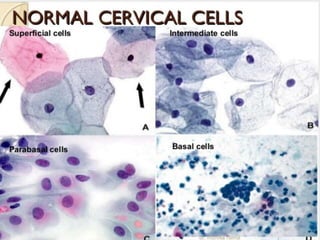
This document discusses liquid based cytology (LBC) and compares it to conventional pap smears. It provides a history of LBC dating back to 1970 and describes two main LBC systems - ThinPrep and SurePath. With ThinPrep, cells are collected in a preservative fluid and then filtered and transferred to a slide. With SurePath, cells are collected in a preservative fluid and separated via density gradient centrifugation before being placed on a slide. Studies have shown LBC reduces inadequate samples and increases specificity compared to conventional pap smears. LBC also better preserves cell morphology. The document focuses on how LBC has advantages over conventional pap smears for detecting cervical intraepithelial neoplasia lesions.






![Reporting according to Bethesda classification[2014]
Specimen type Conventional PAP smear LBC
Adequacy : Epithelial(8000-12,000
cells well preserved and
well visualised.
Glandular
components(10 well
preserved cells in
groups/single)
For LBC -5000 squamous
cells)
Glandular components(10
well preserved cells in
groups/single)
General categorization: Negative for intraepithelial lesion or malignancy[NILM] –
organisms, other non neoplastic findings.
Epithelial cell abnormalities – squamous cells, glandular
cells.](https://image.slidesharecdn.com/liquid-based-cytologyvpg2-180109022337/85/Liquid-based-cytology-10-320.jpg)



![Common trouble shootings -CPS
• Using the incorrect sampler or the right sampler
incorrectly & absence of TZ zone
• Cellular transfer onto slide sampling
error –[airdrying artifact]
• Quality of the smear (thickness,
blood, exudate, mucus,poor fixation)
• Missing the abnormal cells.
•
dense inflammation & hemorrhage screening
• Interpreting the abnormal cells.](https://image.slidesharecdn.com/liquid-based-cytologyvpg2-180109022337/85/Liquid-based-cytology-15-320.jpg)