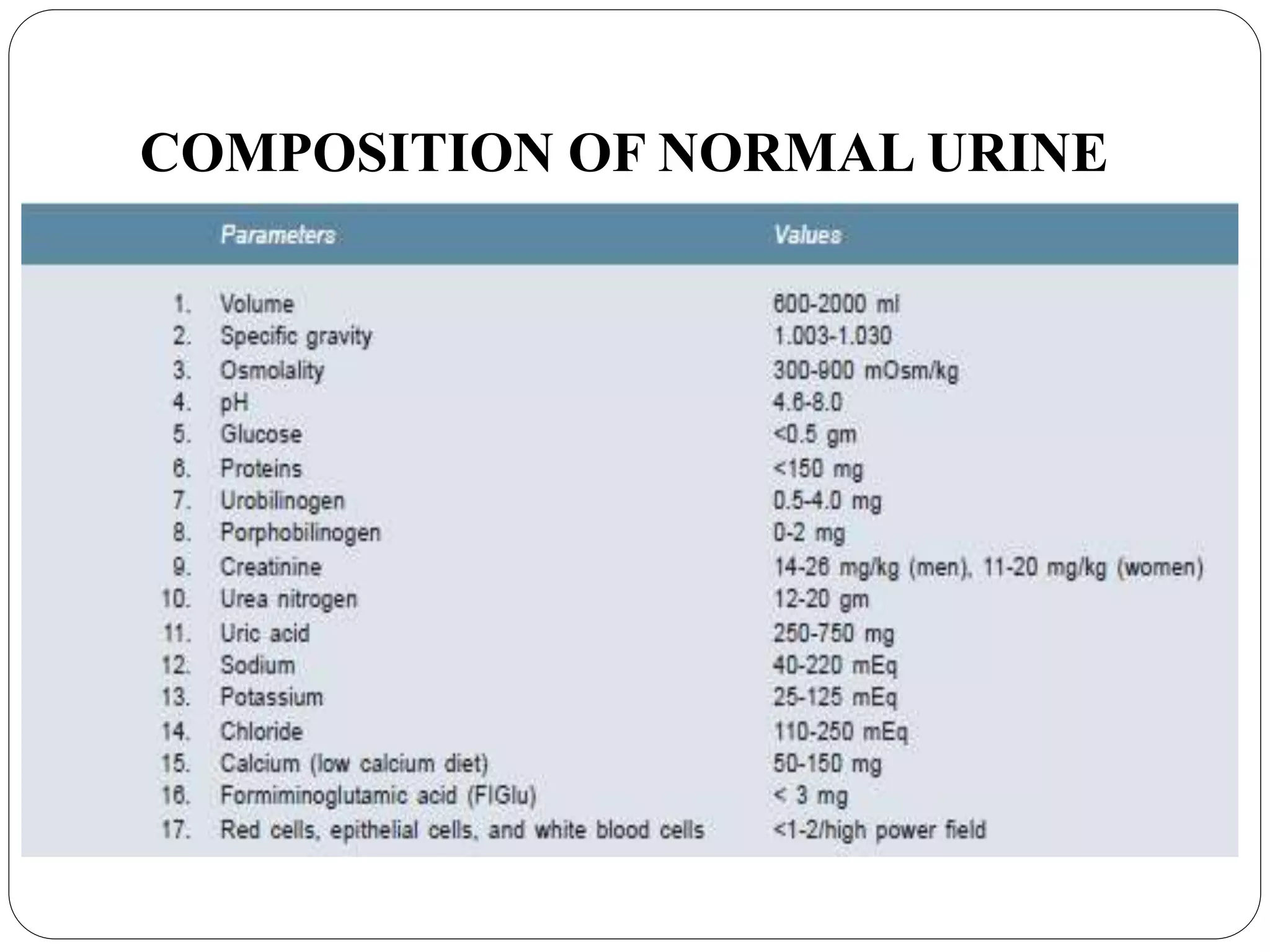
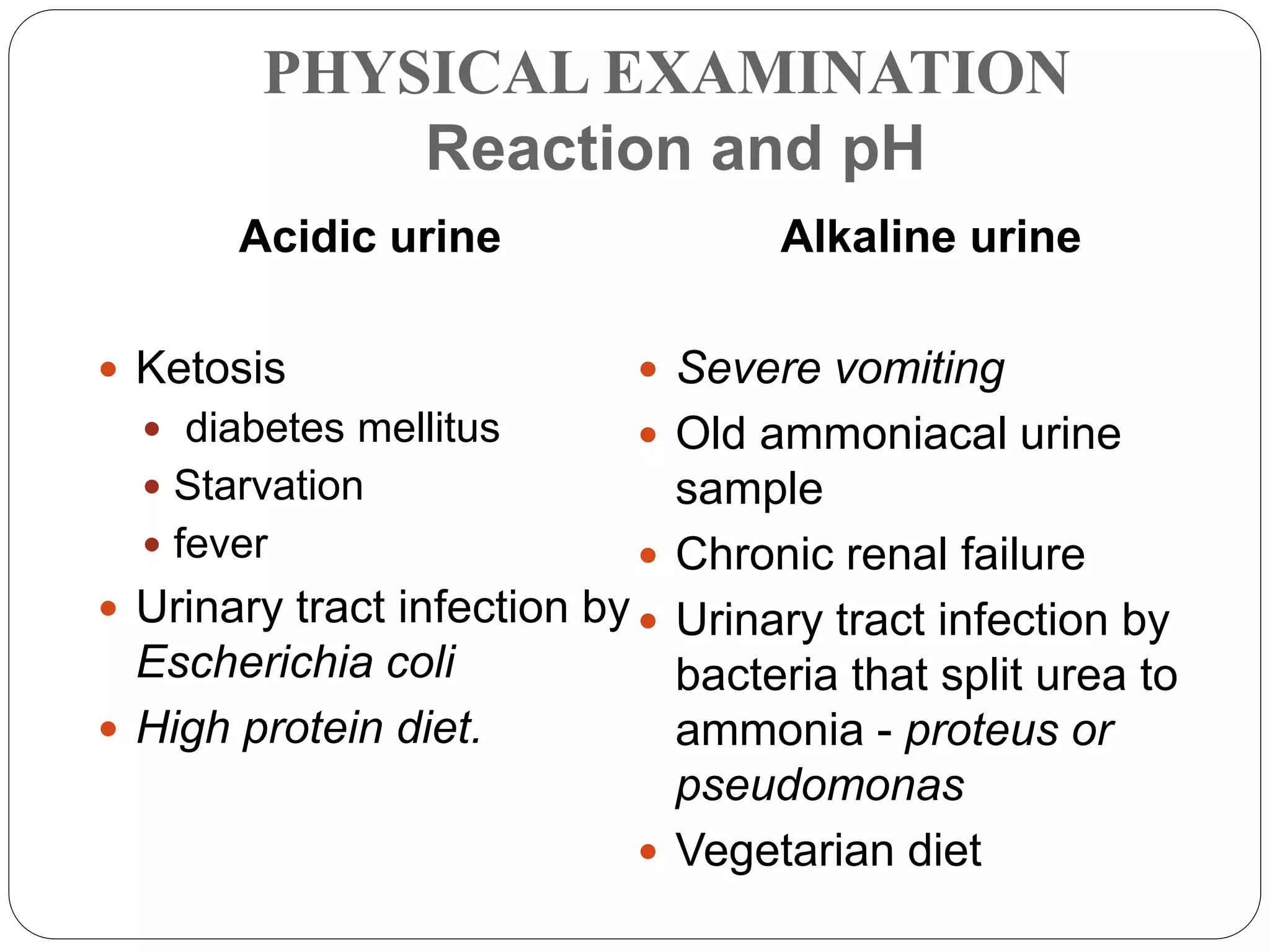
This document provides information about urinalysis, including indications for testing urine, sample collection methods, changes that occur in standing urine, preservation of samples, and the various examinations performed - physical, chemical, and microscopic. The physical examination assesses properties like volume, color, appearance, odor, specific gravity, and pH. The chemical examination tests for proteins, glucose, ketones, bilirubin, and other substances. Microscopic examination analyzes urine sediment. Proper collection and handling of urine samples is important for obtaining accurate test results.