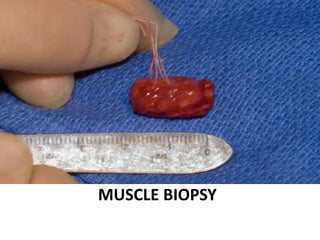
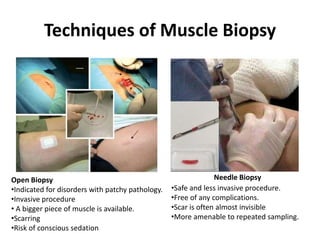
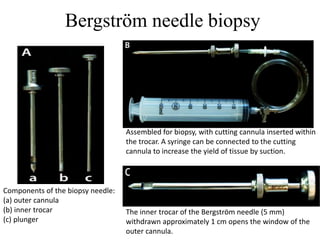
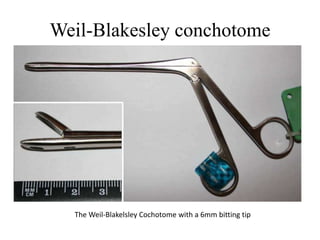
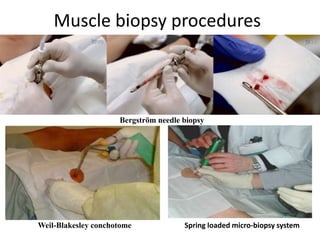
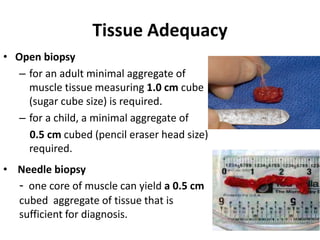
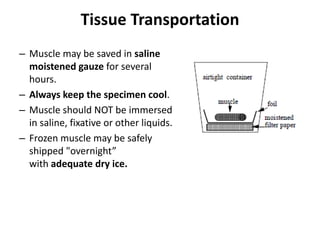
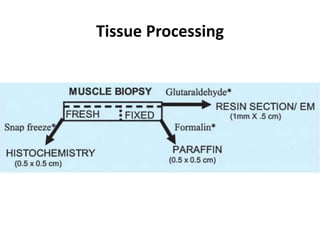
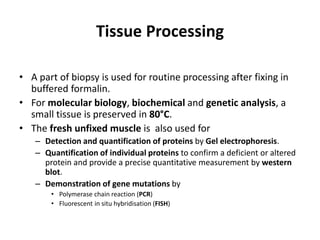
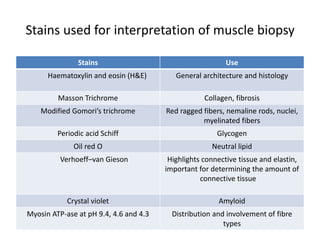
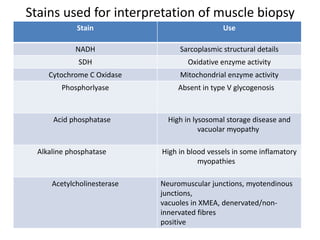
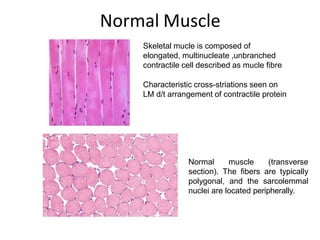
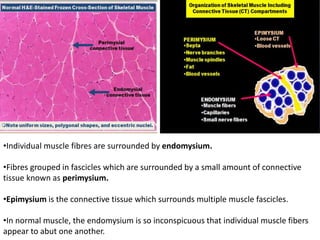
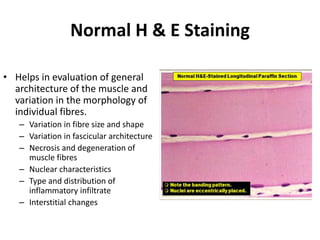
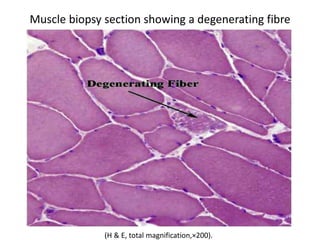
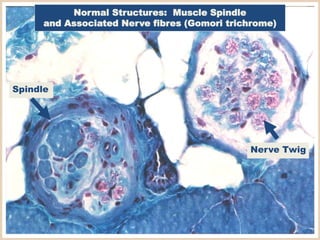
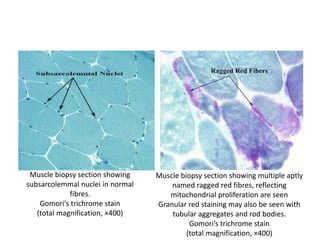
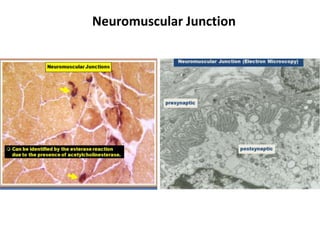
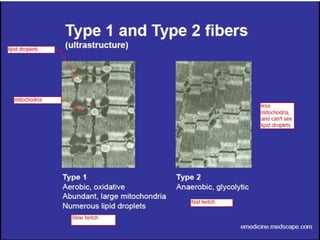
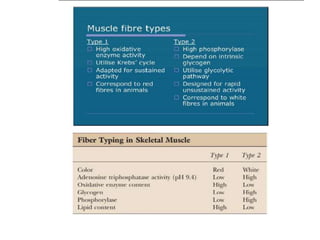
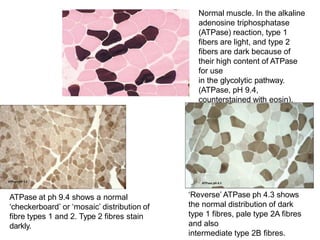
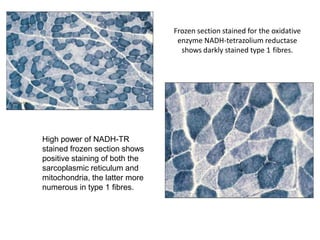
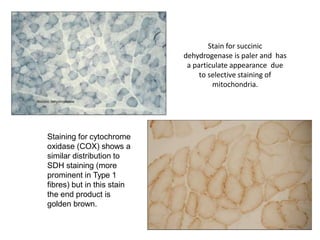
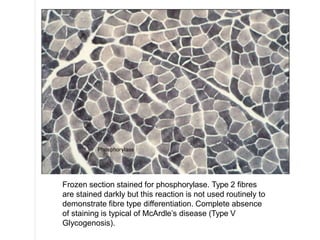
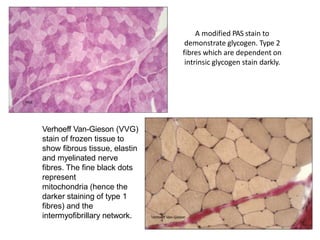
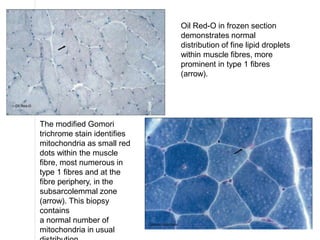
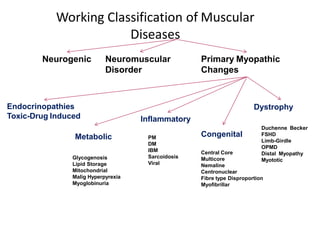
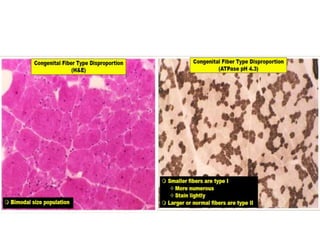
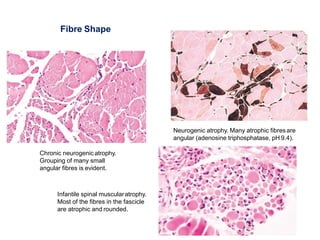
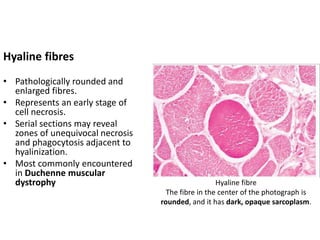
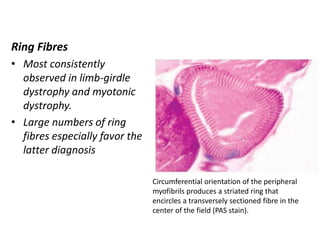
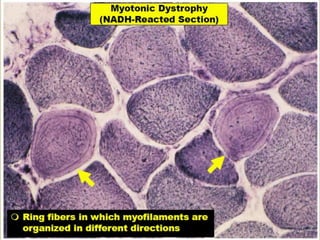
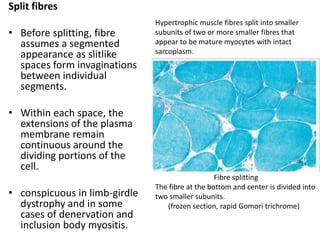
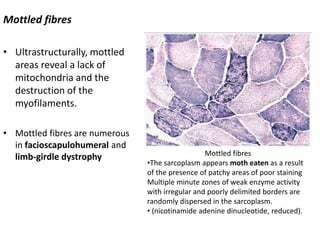
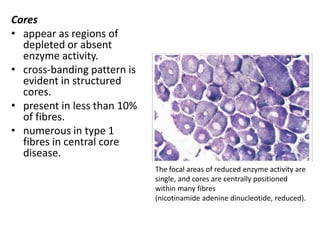
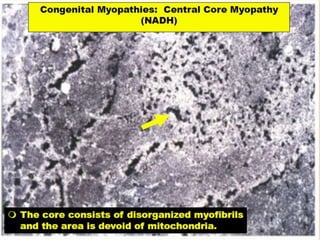
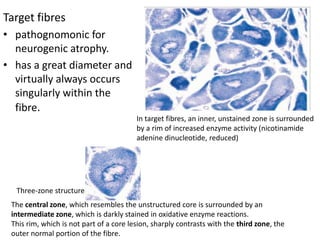
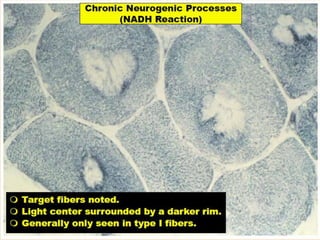
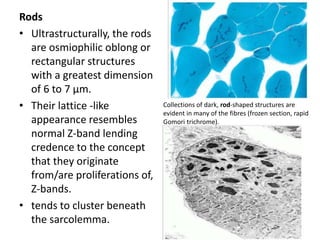
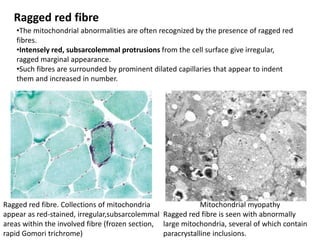
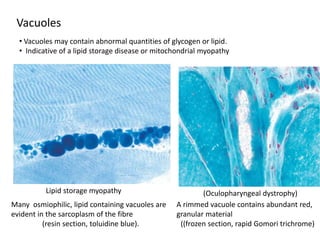
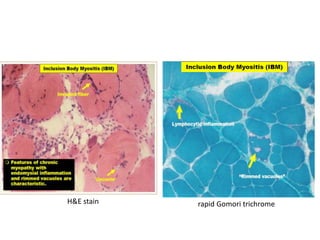
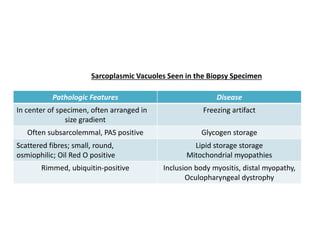
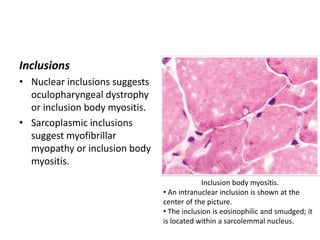
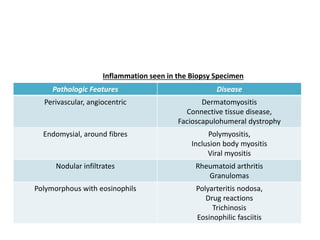
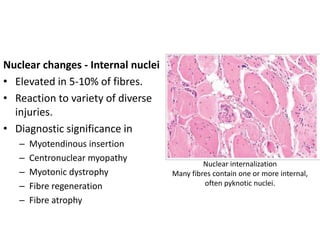
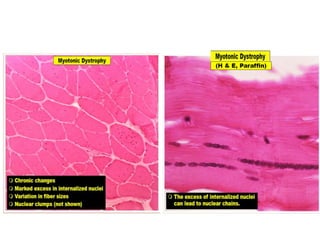
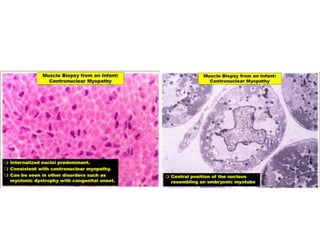
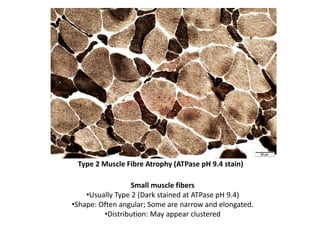
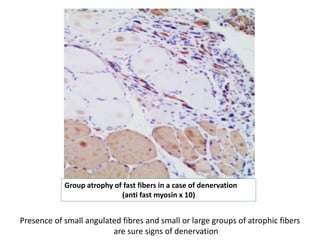
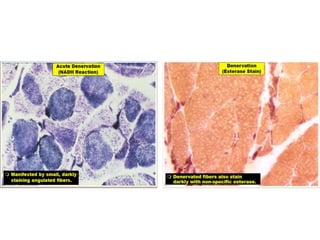
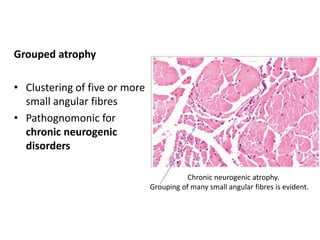
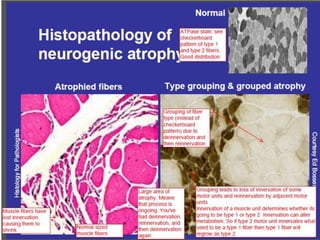
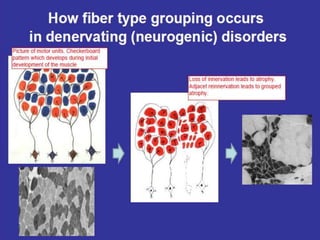
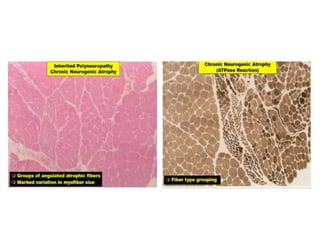
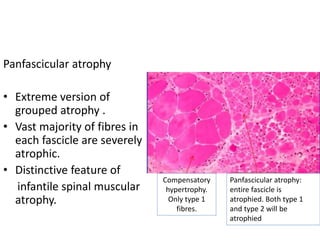
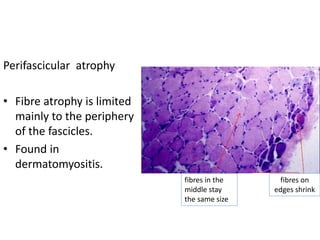
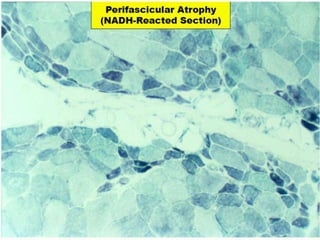
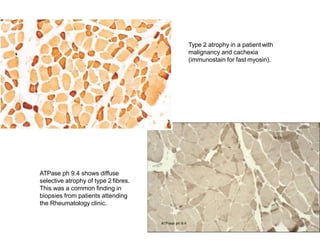
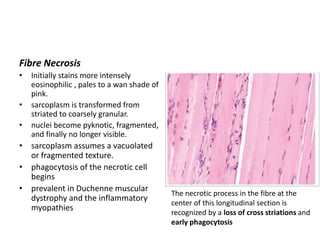
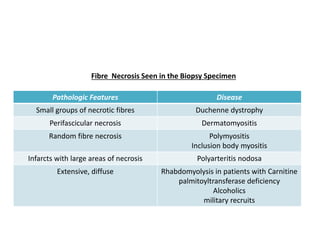
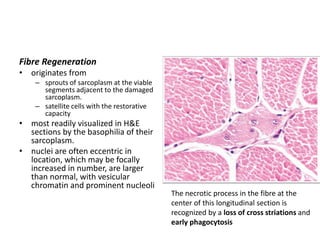
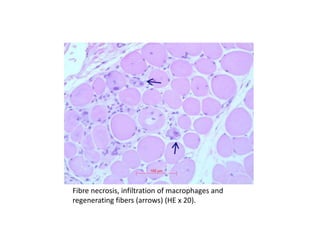
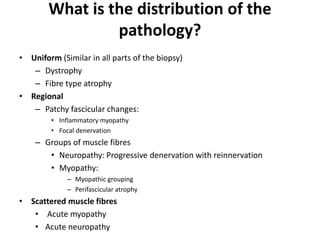
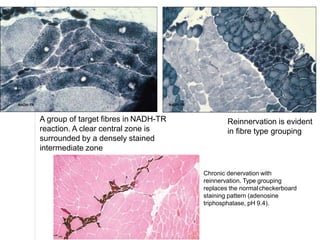
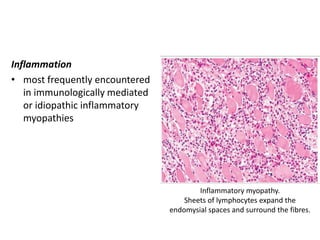
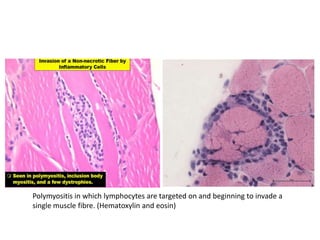
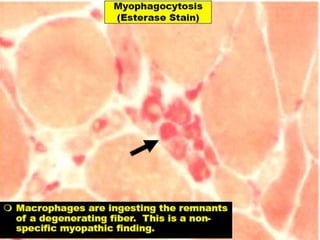
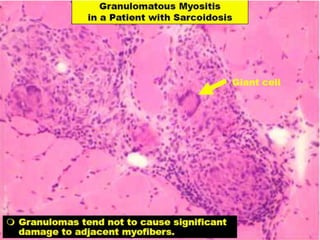
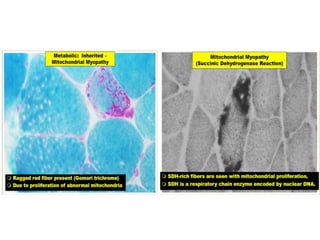
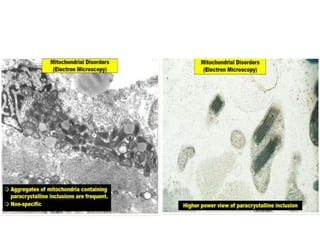
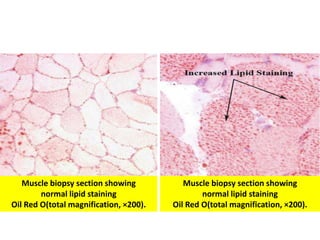
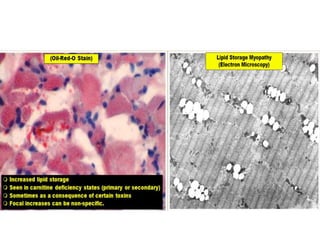
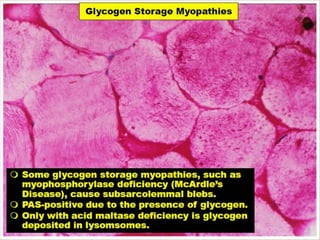
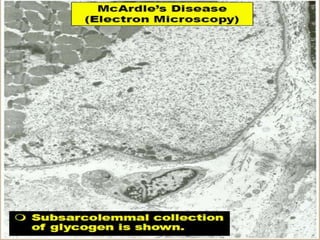
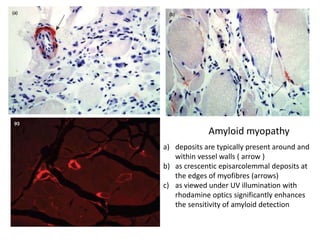
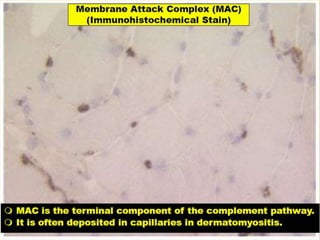
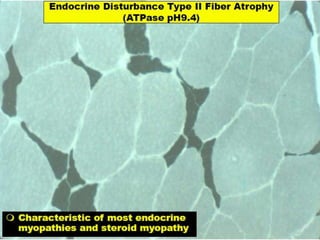
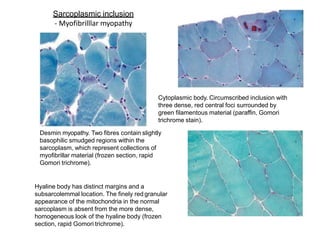
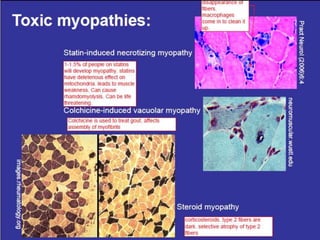
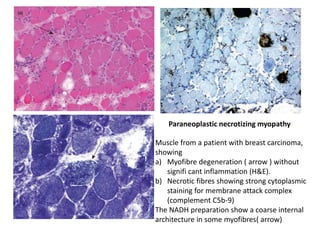
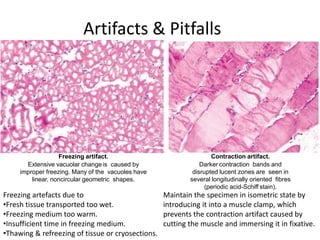
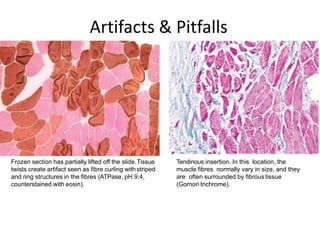
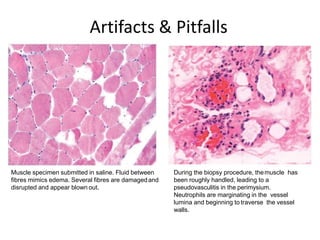
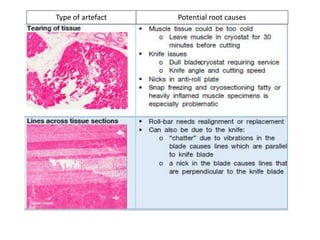
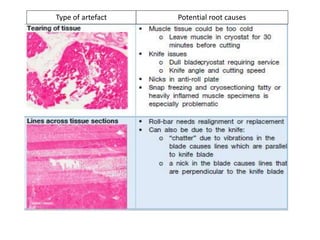
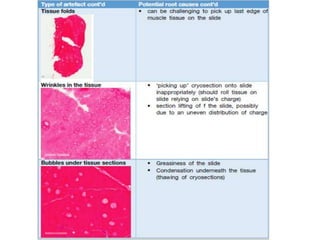
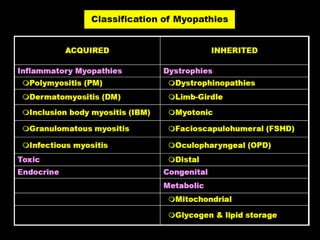
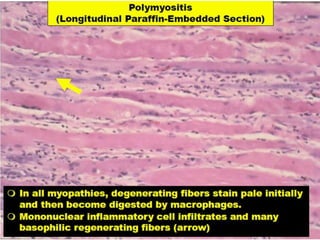
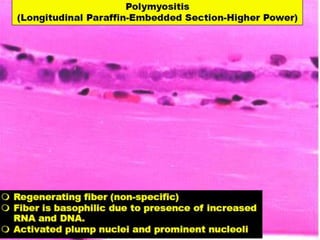
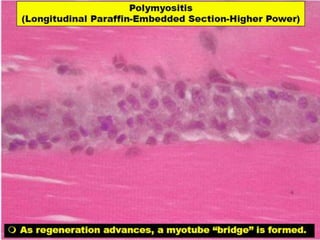
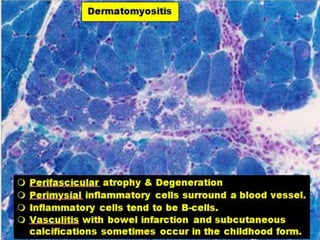
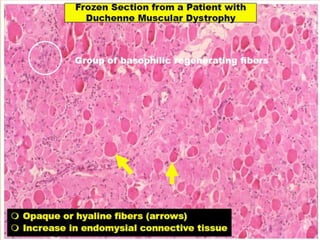
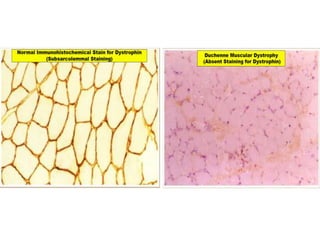
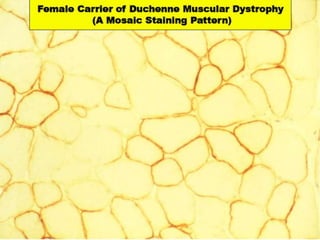
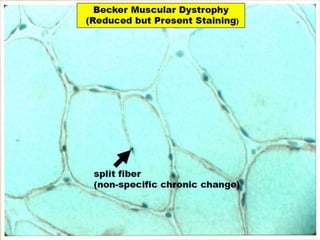
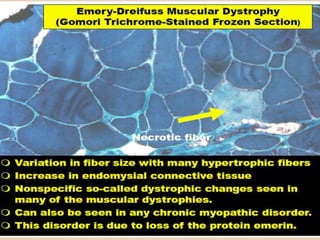
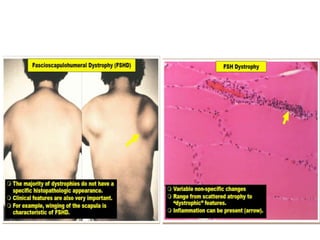
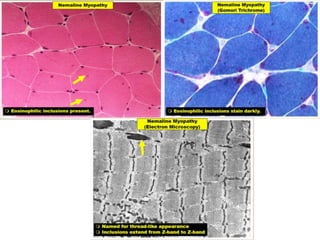
This document provides an overview of muscle biopsy procedures. It discusses the history and development of muscle biopsy techniques from the 1860s onward. It describes the indications and contraindications for muscle biopsy and lists ideal biopsy sites. The key techniques of open biopsy, Bergstrom needle biopsy, and spring-loaded micro-biopsy are outlined. Proper tissue handling, processing, freezing, and staining methods are explained to ensure diagnostic quality. Finally, the document reviews common stains used to interpret muscle biopsy samples under the microscope, including H&E, ATPase, NADH, and SDH, and provides examples of normal muscle histology.