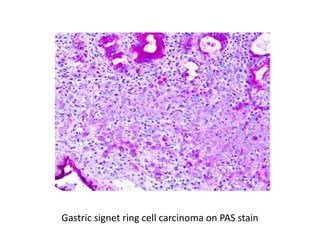
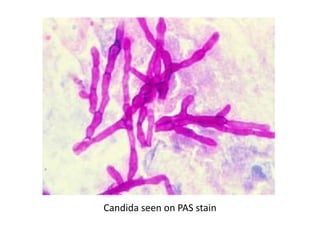
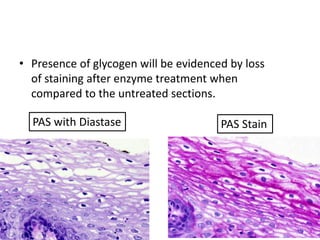
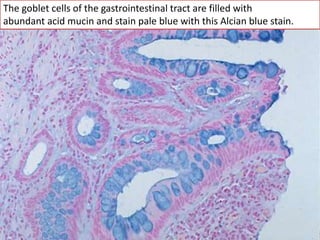
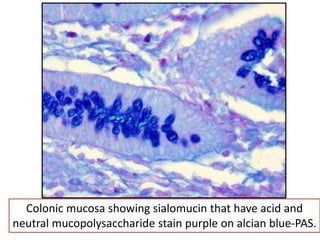
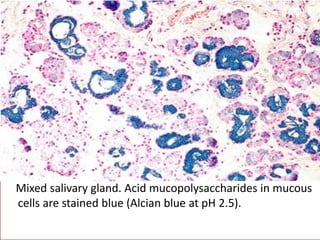
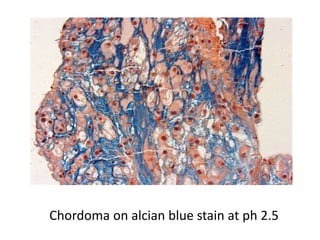
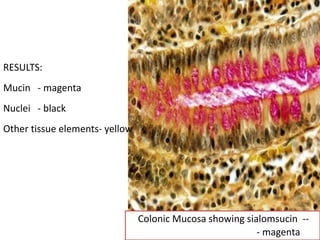
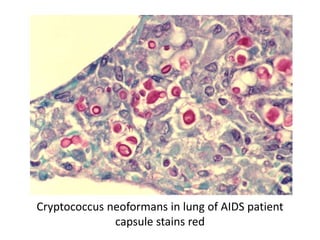
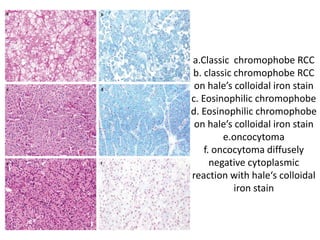
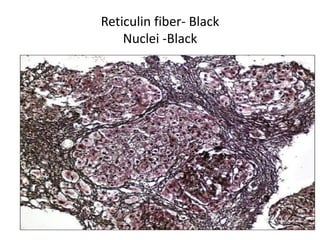
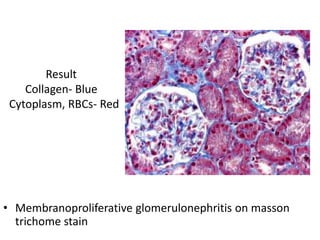
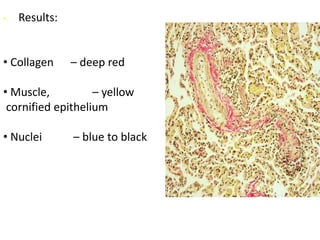
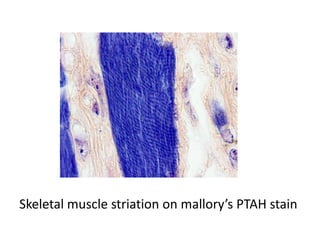
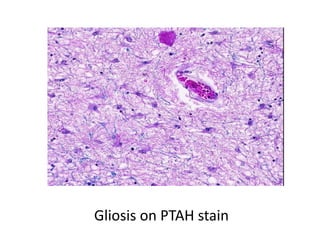
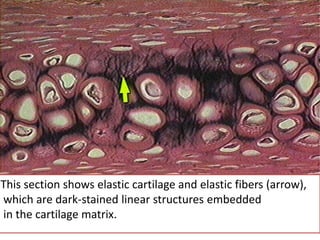
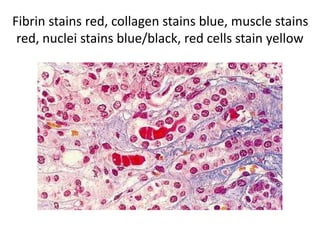
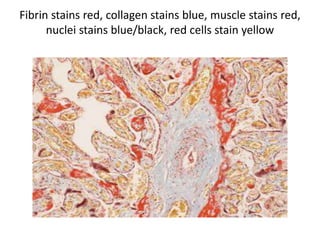
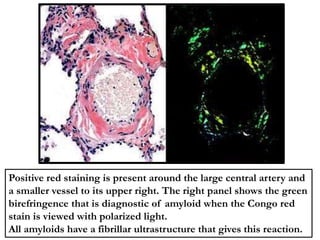
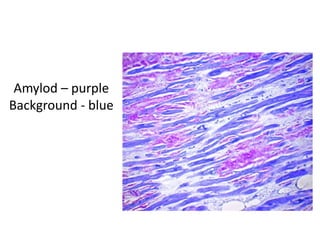
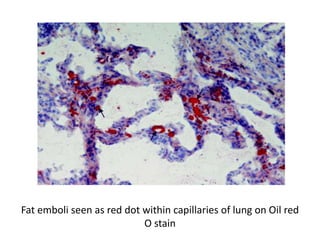
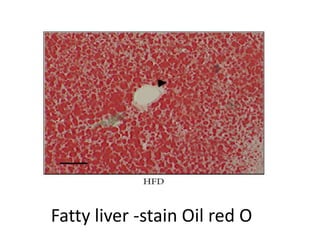
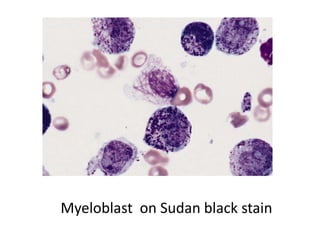
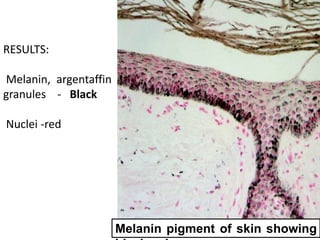
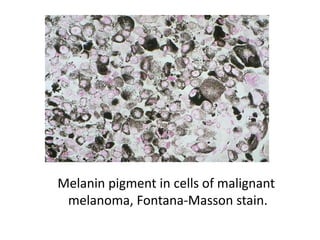
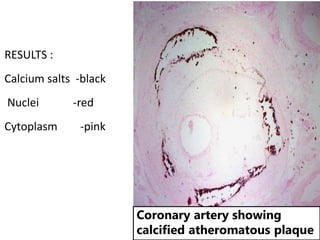
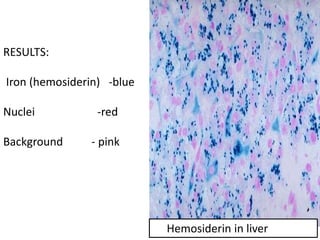
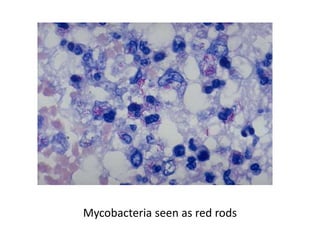
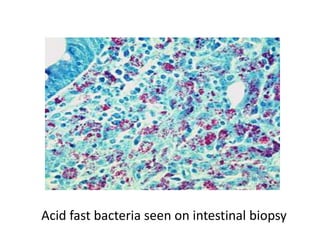
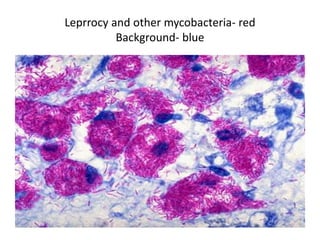
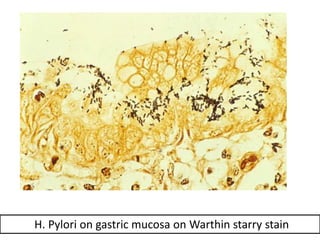
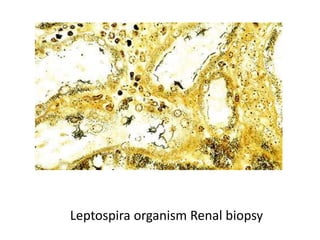
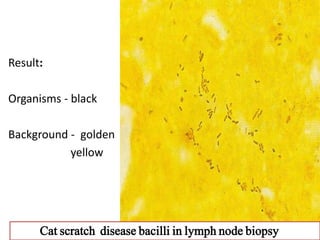
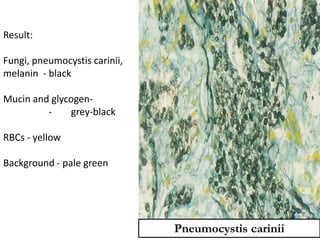
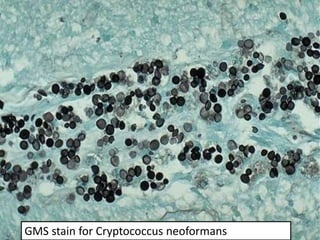
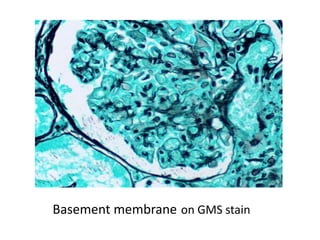
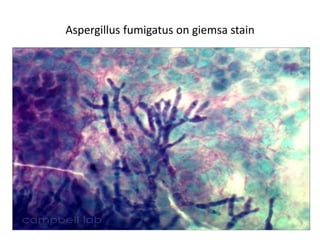
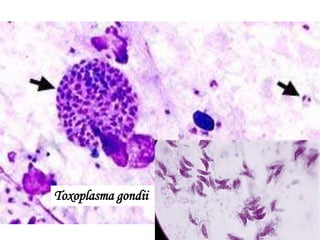
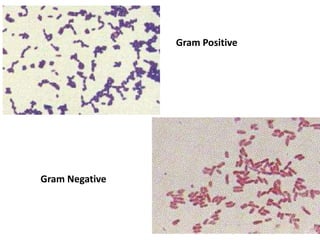
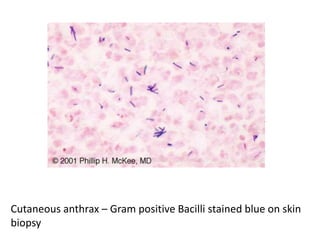
The document discusses various special staining techniques used in histopathology, particularly the Periodic Acid-Schiff (PAS) stain and Alcian Blue stain, to identify specific cellular components and substances that are not visible with routine staining methods. It details the principles, procedures, and diagnostic applications of these stains for various conditions, including different types of carcinomas and fungal infections. Additionally, it covers the use of other stains like mucicarmine and Hale's colloidal iron, outlining their specific functions and results in tissue analysis.