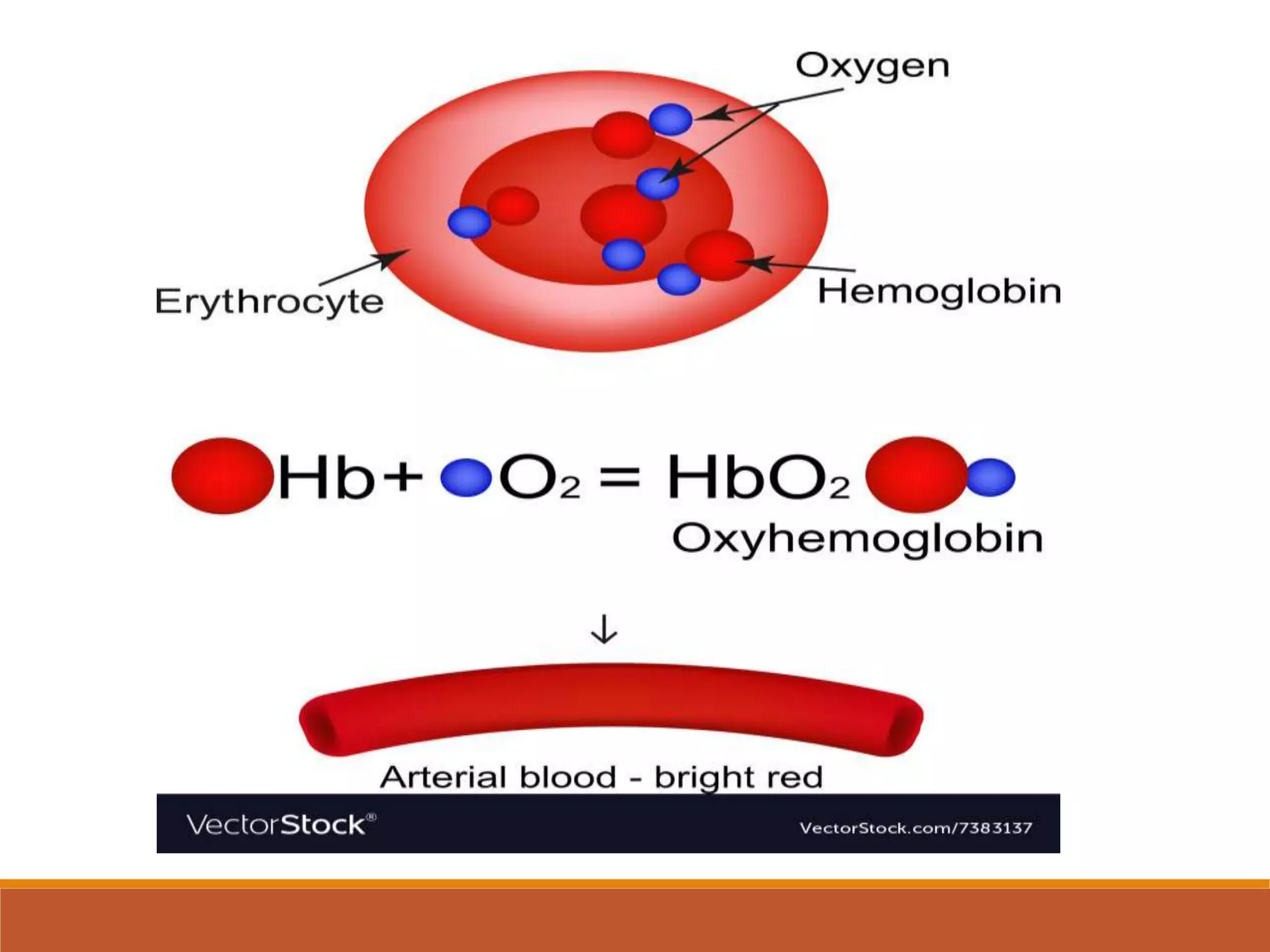
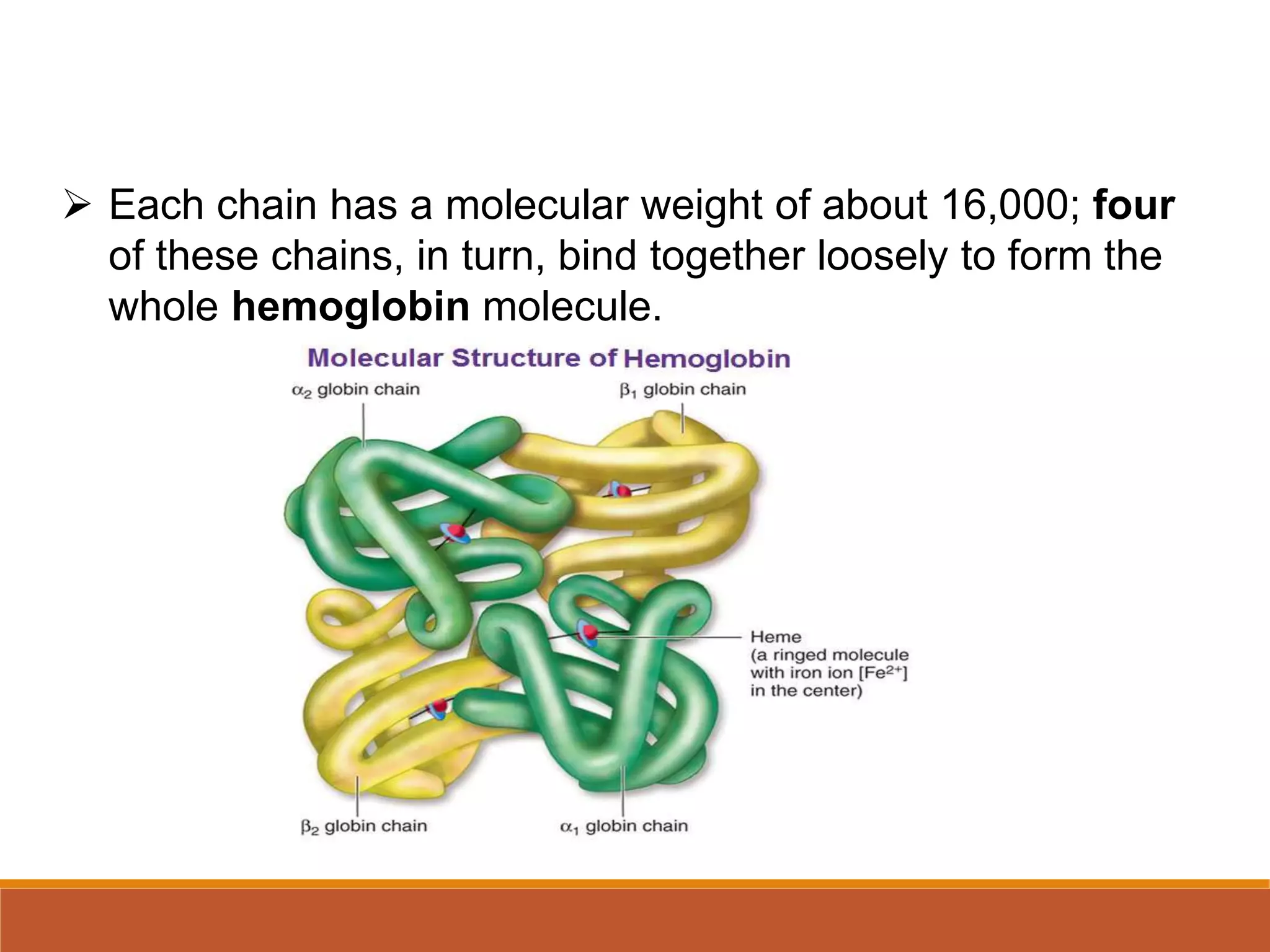
Hemoglobin is the main protein in red blood cells that carries oxygen from the lungs to tissues and returns carbon dioxide back to the lungs. It is composed of four globin chains, including two alpha and two beta chains, that bind to an iron-containing heme group. Hemoglobin synthesis begins in early red blood cells and continues through their maturation. In adults, each red blood cell contains over 600 million hemoglobin molecules to carry oxygen throughout the body before the cells are broken down after around 120 days. Alterations in hemoglobin structure can cause serious diseases like sickle cell anemia.