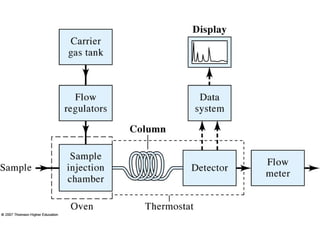
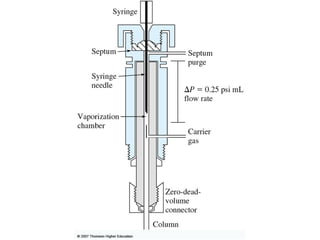
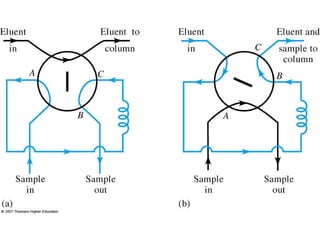
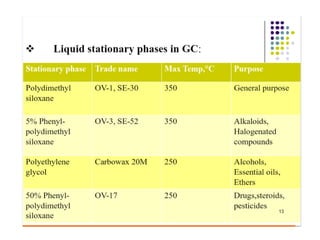
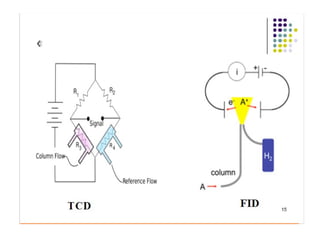
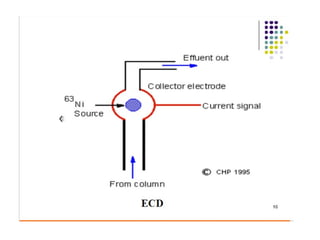
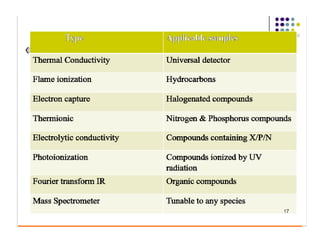
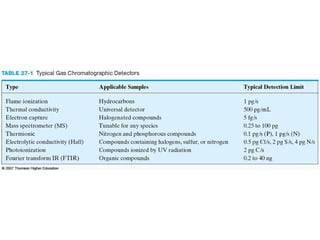
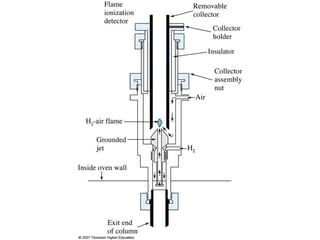
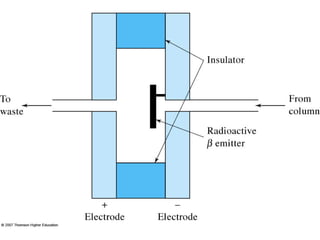
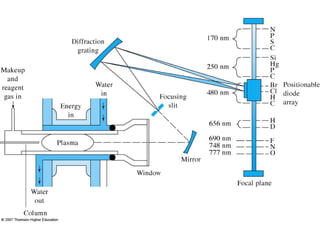
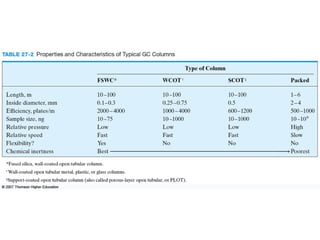
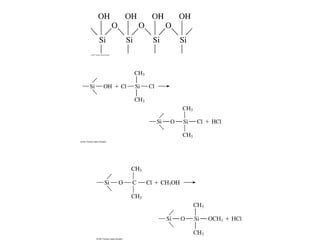
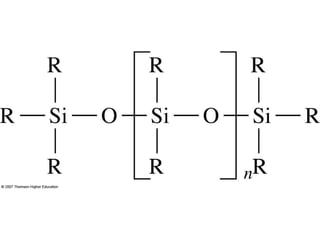
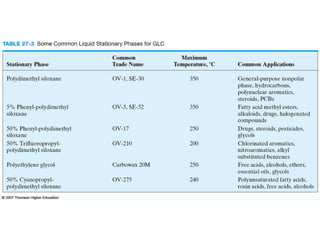
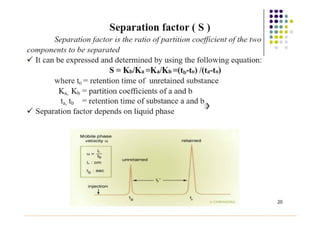
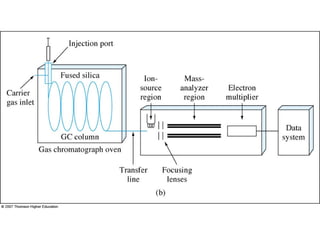
Gas chromatography is a technique used to separate and analyze mixtures that relies on the differential partitioning of analytes between a stationary and mobile phase. Key aspects of GC include vaporizing samples and carrying them through a column with an inert gas, where separation occurs based on interactions with the immobilized stationary phase. Common detectors measure changes in thermal conductivity, ionization, or other properties to identify separated analyte compounds and allow for qualitative and quantitative analysis of complex samples.