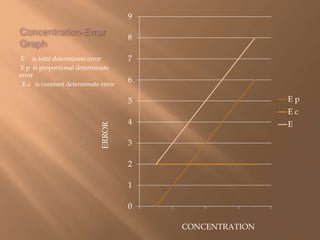
This document discusses various types of errors that can occur in chemical analysis and sampling. It describes determinate errors, which can be identified and corrected, and include constant and proportional determinate errors. It also discusses indeterminate errors, which occur randomly. The document provides definitions and formulas for key statistical concepts used to evaluate precision and accuracy in chemical analysis, including mean, median, standard deviation, variance, control charts, and confidence limits.