The document discusses the determination of lattice energy of ionic compounds using the Born-Haber cycle. It explains that the lattice energy of sodium chloride can be determined experimentally by considering its formation through two different methods. Method 1 is the direct combination of solid sodium and gaseous chlorine to form solid sodium chloride. Method 2 involves 5 steps including sublimation, dissociation, ionization, and combining to form ions and the ionic solid. Using Hess's law, the lattice energy is calculated by equating the enthalpy change between the two methods. For sodium chloride, the calculated lattice energy is -773.95 kJ/mol.

![Ionic Bonding-
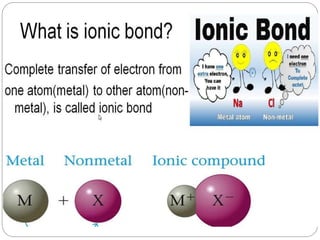
Defn- “The chemical bond which is formed by transfer of one or more
electrons from the valence shell of one atom to the valence shell of another
atom is called as ionic bond.”
Or “The chemical bond which is formed due to electrostatics force of
attraction in between opposite charge ions is called as ionic bond.”
For example – Bond in between Na and Cl
Na + Cl [Na+] [Cl-] or Na+Cl-
(2,8,1) (2,8,7) (2,8) (2,8,8)](https://image.slidesharecdn.com/ionicbonding-210218103058/85/Ionic-bonding-3-320.jpg)











![Ionic Bonding-
Defn- “The chemical bond which is formed by transfer of one or more
electrons from the valence shell of one atom to the valence shell of another
atom is called as ionic bond.”
Or “The chemical bond which is formed due to electrostatics force of
attraction in between opposite charge ions is called as ionic bond.”
For example – Bond in between Na and Cl
Na + Cl [Na+] [Cl-] or Na+Cl-
(2,8,1) (2,8,7) (2,8) (2,8,8)](https://image.slidesharecdn.com/ionicbonding-210218103058/85/Ionic-bonding-15-320.jpg)


























![Once the Na+ and Cl- ions are broken away from the ionic lattice,
following two processes occur same time.
1. Each sodium ion is surrounded by a definite but unknown number of
water molecules, say ‘x’, with their negative ends (oxygen ends)
pointing towards it, as shown in figure.
Na+ + xH2O = [Na(H2O)x]+
This process is called solvation of sodium ion and the energy change
associated with it is called as solvation energy of sodium ion, (Hs)Na+.
The chemical species [Na(H2O)x]+ is called solvated or acquated sodium
ion and may also be represented as [Na(aq)]+ .](https://image.slidesharecdn.com/ionicbonding-210218103058/85/Ionic-bonding-42-320.jpg)
![2) Each chloride ion is surrounded by definite but unknown number of water
molecules, say ‘y’ with their positive ends (hydrogen ends) pointing
towards it, as shown
Cl- + yH2O = [Cl(H2O)y]-
The process is called solvation of chloride ion and the energy change
associated with it is called solvation energy of chloride ion, (Hs)Cl-. The
chemical species [Cl(H2O)y]- is called solvated or aquated chloride ion and
may also be represented as [Cl(aq)]-](https://image.slidesharecdn.com/ionicbonding-210218103058/85/Ionic-bonding-43-320.jpg)
![The total process may be written as:
NaCl(s) + (x+y)H2O [Na (H2O)x]+ + [Cl(H2O)y]-
or NaCl(s) + aq Na+
(aq) + Cl-
aq)
fig- Born-Haber cycle for determination of salvation energy.](https://image.slidesharecdn.com/ionicbonding-210218103058/85/Ionic-bonding-44-320.jpg)






