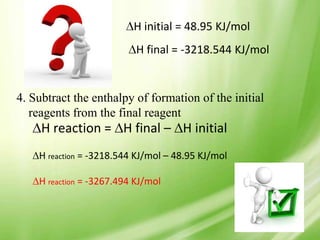
The document provides objectives and information about calculating enthalpies, solving problems in calorimetry, and Hess' Law. The objectives are to know how to calculate enthalpies, solve problems in calorimetry, and apply Hess' Law. It defines important terms like enthalpy, enthalpy of reaction, and discusses how to calculate enthalpies using heat of formation values from tables. Examples are provided to demonstrate calculating enthalpy of reaction. Important enthalpy changes like combustion, formation, atomization, fusion, and vaporization are defined. The document also discusses calorimetry, defining key terms, and outlines Hess' Law which states the heat of a reaction is the same whether it occurs








![Some Important Enthalpy Changes
1. Enthalpy Change of Combustion
- the enthalpy change which
occurs when one mole of the
substance is completely burnt
in oxygen under standard
conditions
Eg. C (graphite) + ½ O2 (g) CO2 (g)
C (graphite) + O2 (g) CO2 (g)
chemist’s shorthand:
H c,m [(graphite)] = -393.5 kJ/mol](https://image.slidesharecdn.com/enthacalorhess2-130320202314-phpapp01/85/Enthalpy-Calorimetry-Hess-s-Law-10-320.jpg)
![Some Important Enthalpy Changes
2. Enthalpy Change of Formation
Eg. The SECF of methane, CH4, refersone mole of
- the enthalpy change when to the change:
the compound is formed from its elements
C (graphite) + 2 H2 (g) ---->CH4 (g) H = -74.8 kJ/mol
under standard conditions
H f,m [CH4(g)] = -74.8 kJ/mol
- may also be called Heats of Formation.
3. Enthalpy Change of Atomisation
Eg. C (graphite C of an element is kJ/mol
The SMECA (g) H = 716.7 the
enthalpy change when one mole of its
atoms in the gaseous 716.7is formed from
H at,m [(graphite)] = state kJ/mol
the element under standard conditions .
* Atomisation is always endothermic.](https://image.slidesharecdn.com/enthacalorhess2-130320202314-phpapp01/85/Enthalpy-Calorimetry-Hess-s-Law-11-320.jpg)
![Some Important Enthalpy Changes
4. Enthalpy Change of Fusion
- The enthalpy change when 1 mole of solid is
converted to one mole of liquid at its melting point at
standard pressure
H fus,m [(H20)] = 716.7 kJ/mol H = 6.01 kJ/mol](https://image.slidesharecdn.com/enthacalorhess2-130320202314-phpapp01/85/Enthalpy-Calorimetry-Hess-s-Law-12-320.jpg)
![Some Important Enthalpy Changes
5. Enthalpy Change of Vaporisation
- The enthalpy change when 1 mole of liquid is
converted to one mole of gas at its boiling point at
standard pressure
H vap,m [(H20)] = 716.7 kJ/mol H = 41.09 kJ/mol](https://image.slidesharecdn.com/enthacalorhess2-130320202314-phpapp01/85/Enthalpy-Calorimetry-Hess-s-Law-13-320.jpg)







