This document provides an overview of chemical thermodynamics, including:
- The first law of thermodynamics which states that change in internal energy equals heat added plus work done.
- The second law of thermodynamics which states that the entropy of the universe increases for spontaneous processes.
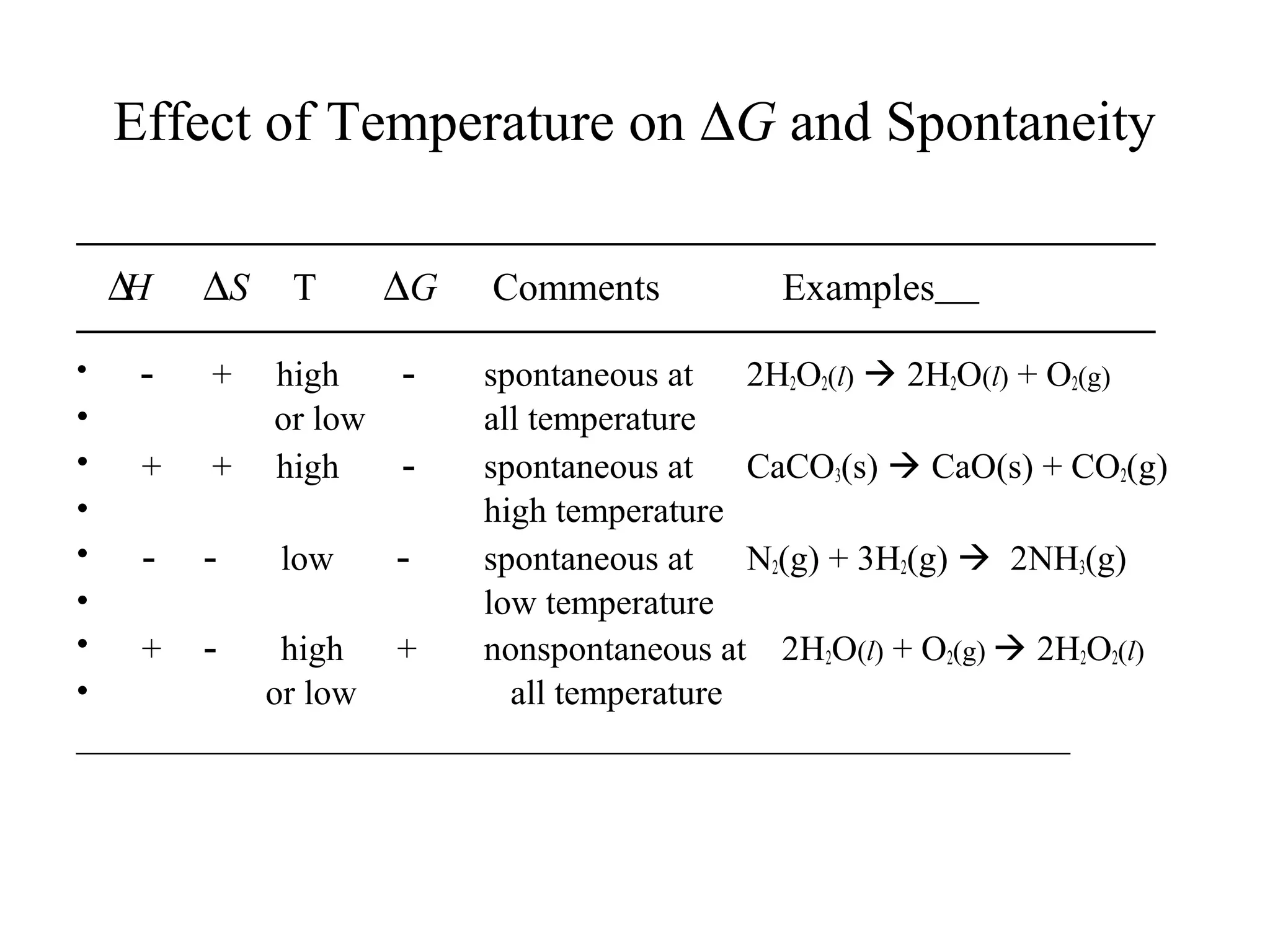
- How changes in entropy and free energy determine whether processes are spontaneous, with spontaneous processes favoring higher entropy and more negative free energy.