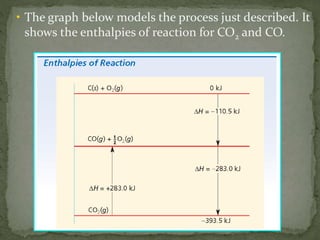
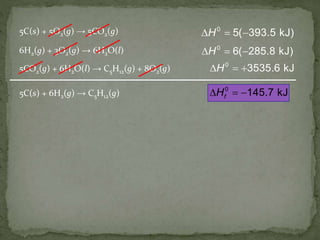
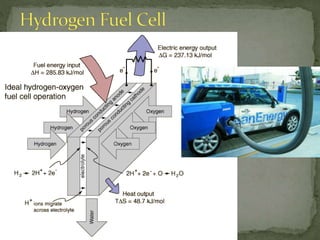
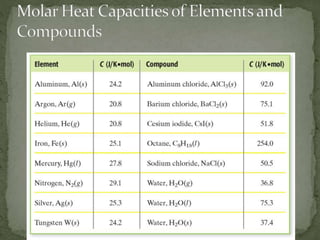
The document discusses thermochemistry and concepts related to heat, temperature, enthalpy, and thermochemical equations. It provides definitions and examples of:
- Specific heat and how to calculate it using the formula q=m*c*ΔT
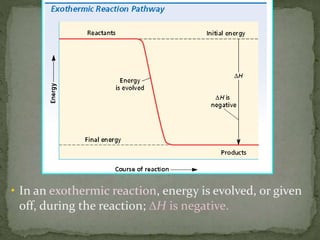
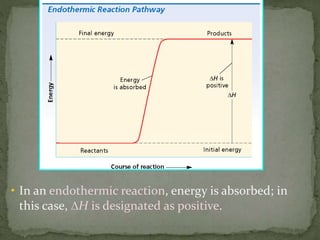
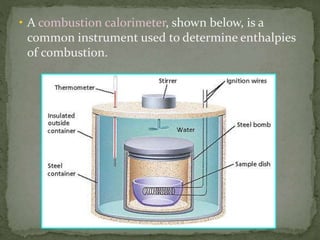
- Enthalpy change (ΔH), enthalpy of reaction, enthalpy of formation (ΔHf), and enthalpy of combustion (ΔHc)
- How to use Hess's law to calculate enthalpy changes from individual reaction steps

























![• Using Hess’s law, any enthalpy of reaction may be
calculated using enthalpies of formation for all the
substances in the reaction of interest, without knowing
anything else about how the reaction occurs.
• Mathematically, the overall equation for enthalpy
change will be in the form of the following equation:
∆H0 = sum of [( of products) × (mol of products)]
– sum of [( of reactants) × (mol of reactants)]
fH0
fH0
](https://image.slidesharecdn.com/chapter17-1-101111141320-phpapp02/85/Chapter-17-1-Thermochemistry-30-320.jpg)