This document provides an overview of the key concepts covered in 5 sections of a chapter on energy and chemical change:
Section 15.1 defines energy and distinguishes between potential and kinetic energy. It relates chemical potential energy to heat released or absorbed in chemical reactions.
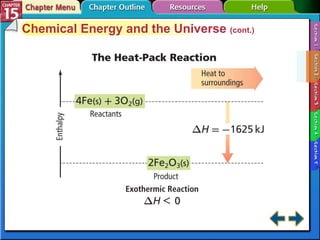
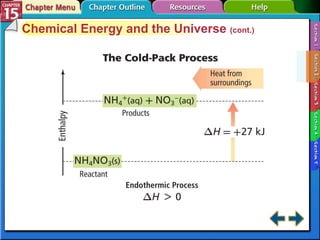
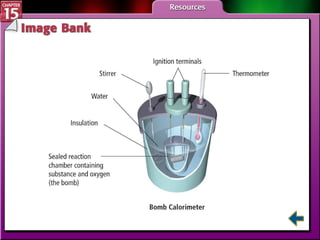
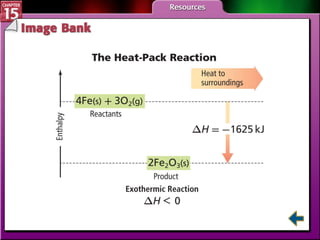
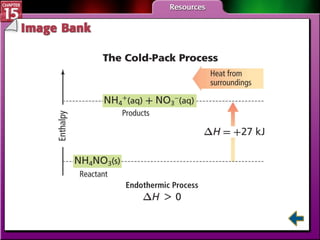
Section 15.2 describes how calorimetry is used to measure energy changes and defines enthalpy and enthalpy changes.
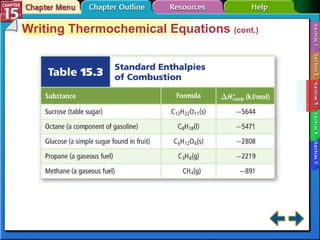
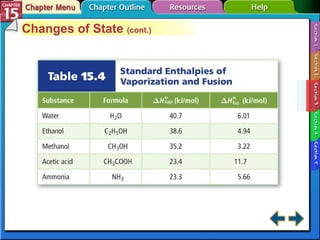
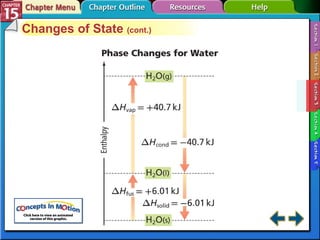
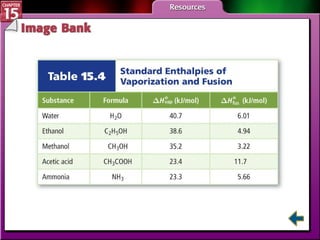
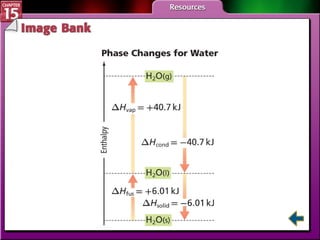
Section 15.3 explains how to write thermochemical equations and describes energy changes during state changes.
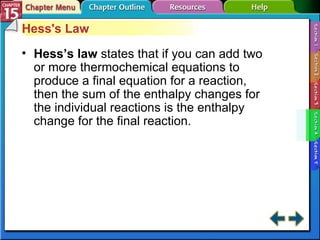
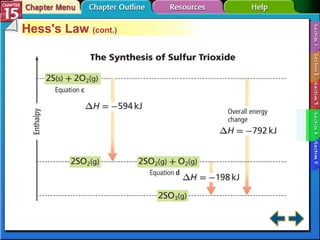
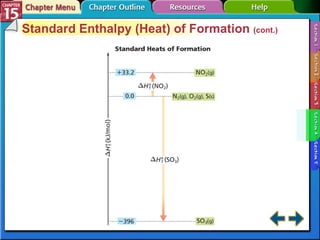
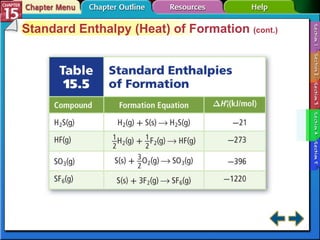
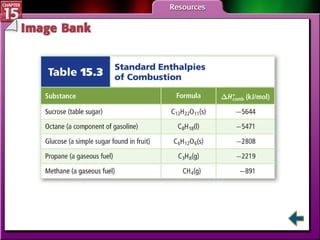
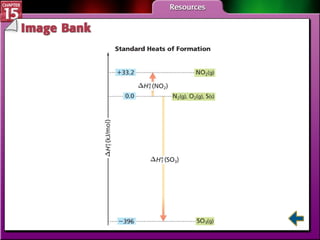
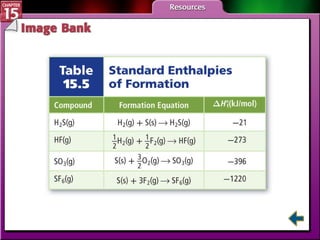
Section 15.4 discusses calculating enthalpy changes using Hess's law and standard enthalpies of formation.
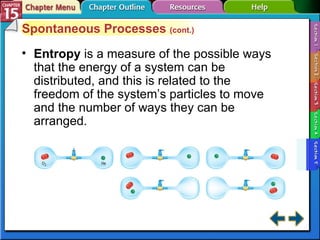
Section 15.5 differentiates between spontaneous and nonspontaneous processes and explains how entropy







































































![Help Click any of the background top tabs to display the respective folder. Within the Chapter Outline, clicking a section tab on the right side of the screen will bring you to the first slide in each respective section. Simple navigation buttons will allow you to progress to the next slide or the previous slide. The “Return” button will allow you to return to the slide that you were viewing when you clicked either the Resources or Help tab. The Chapter Resources Menu will allow you to access chapter specific resources from the Chapter Menu or any Chapter Outline slide. From within any feature, click the Resources tab to return to this slide. To exit the presentation, click the Exit button on the Chapter Menu slide or hit Escape [Esc] on your keyboards while viewing any Chapter Outline slide.](https://image.slidesharecdn.com/cmcchapter15-100613134053-phpapp01/85/Cmc-chapter-15-96-320.jpg)
