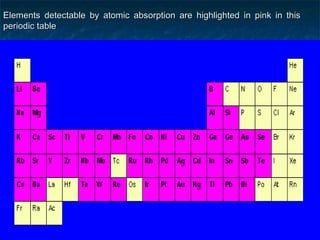
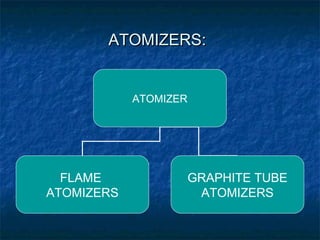
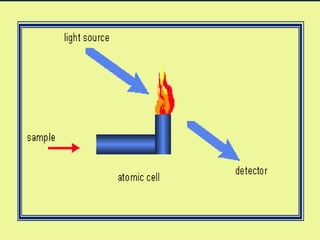
This document provides an overview of atomic absorption spectroscopy. It describes the basic principles, instrumentation, and applications of AAS. The key components of an AAS include a light source, atomizer, monochromator, and detector. Samples are atomized in a flame or graphite furnace and analyzed by measuring the absorption of light from a hollow cathode lamp at specific wavelengths corresponding to the element of interest. Calibration curves are used to determine unknown concentrations in samples. AAS is commonly used to analyze metals in environmental, food, and pharmaceutical applications.