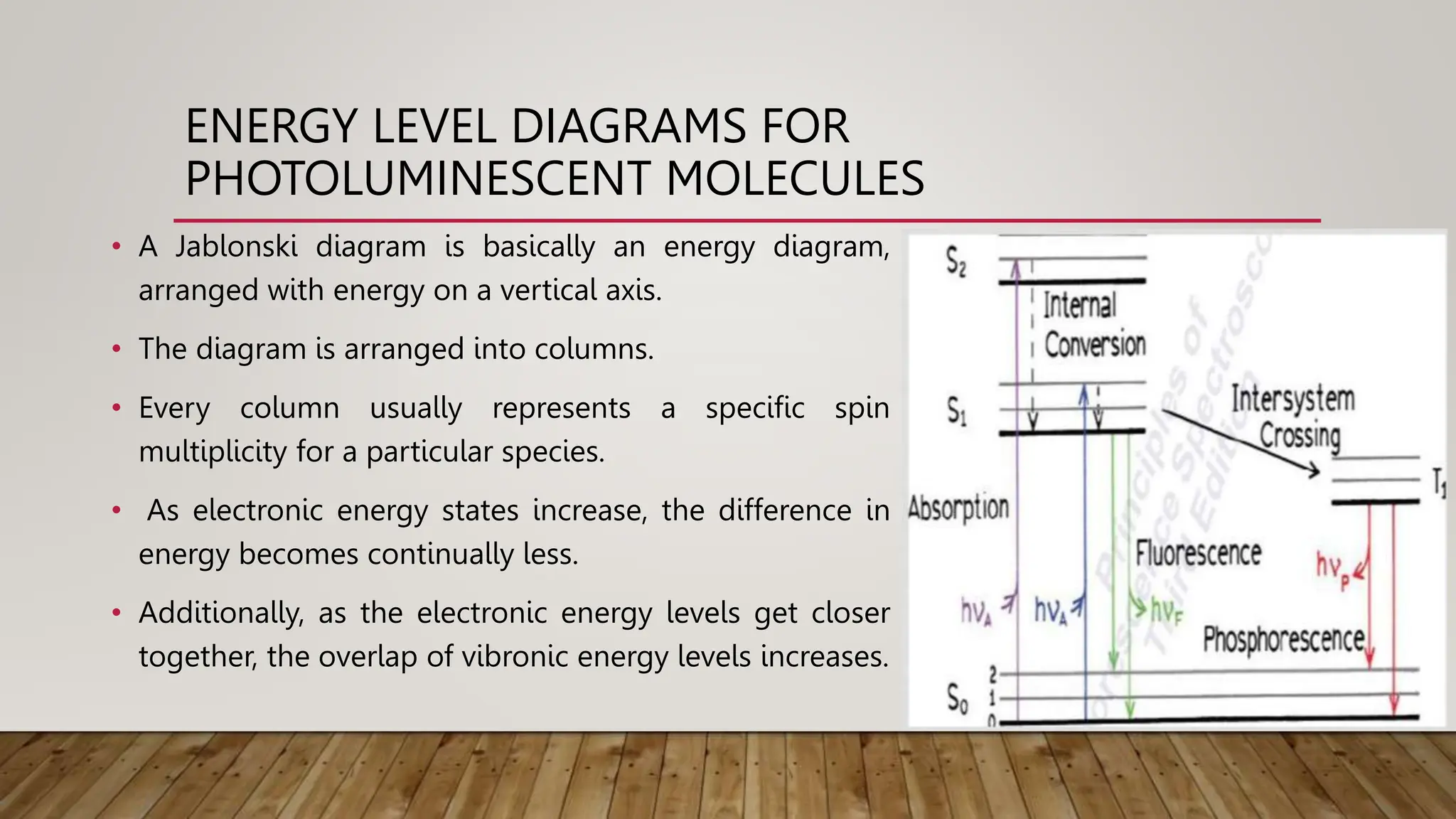
Spectrofluorimetry is a technique that uses fluorescence to measure analytes. It involves exciting a sample with light of a specific wavelength, which causes the sample to emit light of a longer wavelength. The amount of emitted light is proportional to the analyte concentration. Factors like pH, temperature, and solvent can affect fluorescence intensity. The main components of a spectrofluorimeter are a light source, monochromator, sample cell, and light detector. Applications include determining inorganic substances, pharmaceutical analysis, and liquid chromatography.