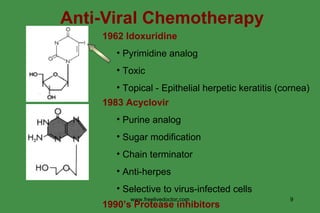
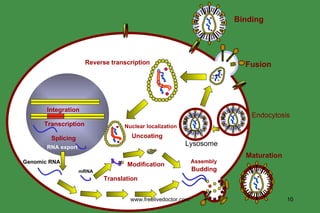
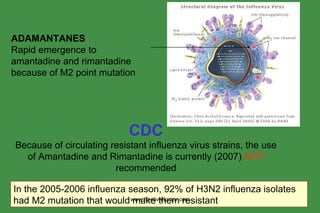
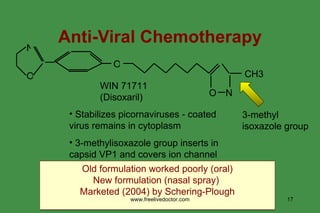
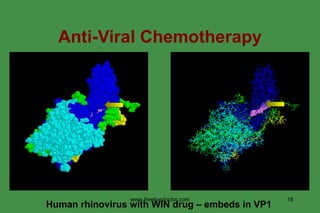
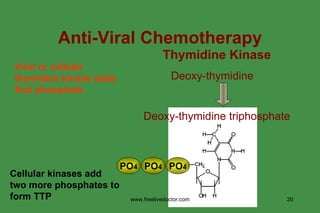
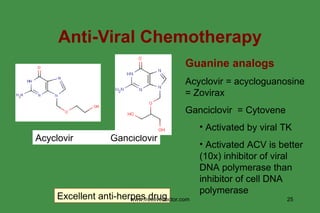
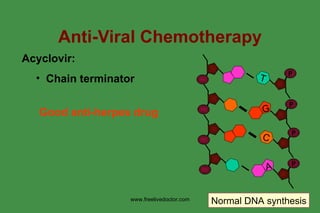
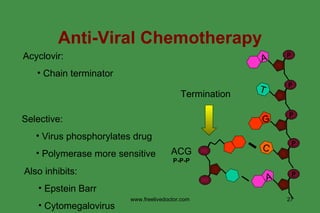
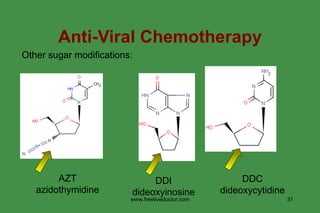
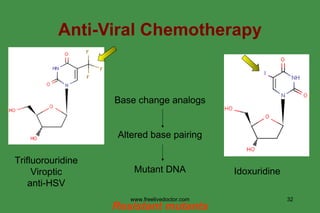
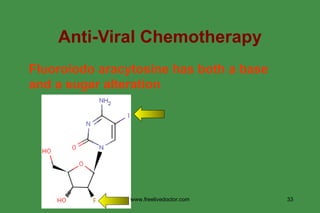
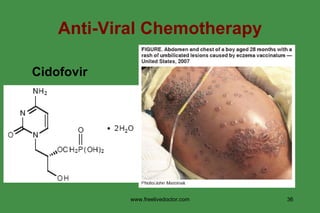
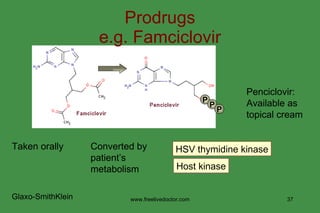
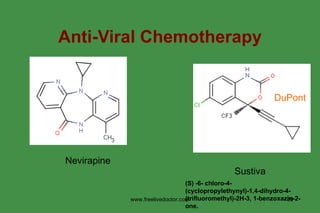
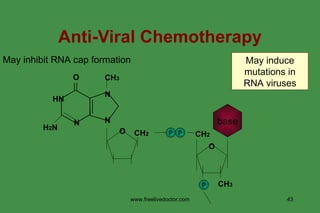
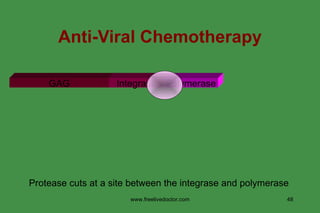
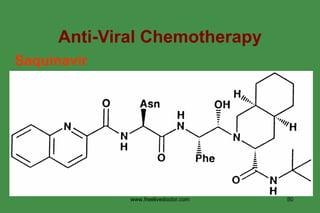
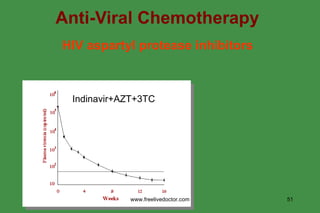
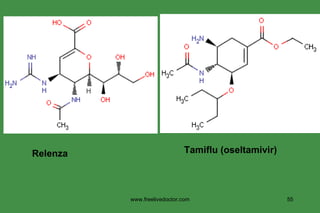
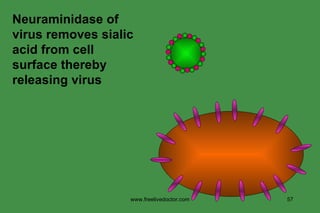
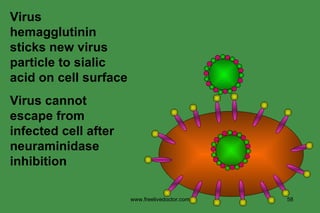
The document discusses anti-viral chemotherapy and considerations for developing effective anti-viral drugs. It notes that ideal drugs are selectively toxic to virus-infected cells, inhibit specific viral enzymes or functions, and have a high therapeutic index. Several classes of anti-viral drugs are described including nucleoside analogs that target viral polymerases, protease inhibitors for HIV, and neuraminidase inhibitors for influenza. Developing drugs that can interfere with viral replication without harming host cells remains an ongoing challenge.