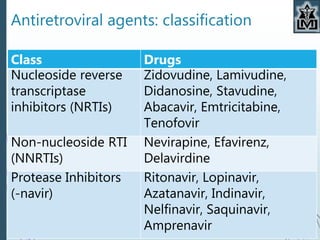
This document provides an overview of antiviral agents for medical students. It discusses the targets of antiviral drugs, including viral enzymes and virus-specific steps. Several classes of antiviral agents are described, including drugs for influenza, hepatitis, HIV, and herpes viruses. Specific drugs like acyclovir, ganciclovir, and famciclovir are examined in depth, outlining their mechanisms of action, pharmacokinetics, uses, and side effects in treating herpes virus infections. The conclusion emphasizes that antiviral drugs achieve selective toxicity by targeting viral processes and that classification is based on activity against different virus families.