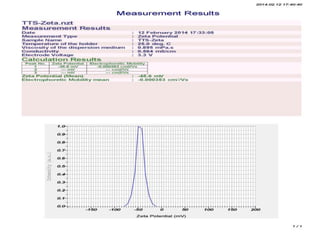
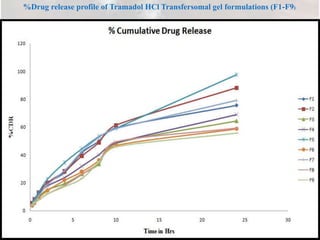
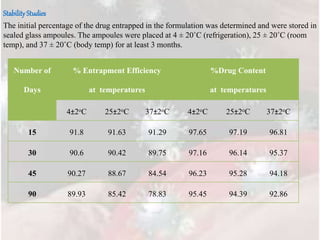
The document provides information on the formulation and evaluation of Tramadol Hydrochloride loaded transferosome gel. Key points include:
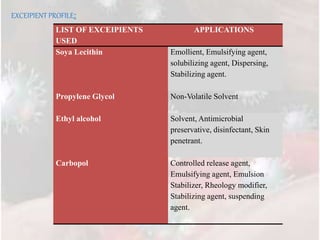
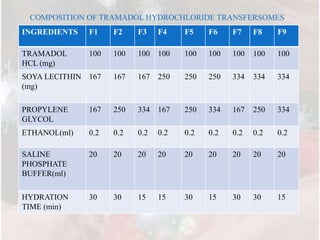
1) Nine different transferosome formulations were prepared using different ratios of soya lecithin, propylene glycol, and other excipients to encapsulate Tramadol HCl.
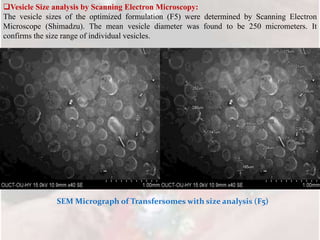
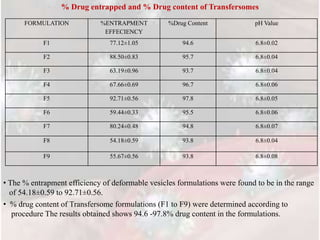
2) Characterization of the formulations found that formulation F5 had the highest entrapment efficiency of 92.71% and drug content of 97.8%.
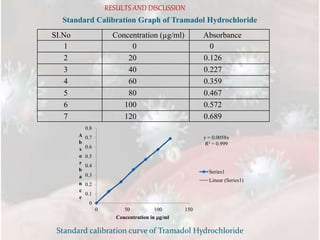
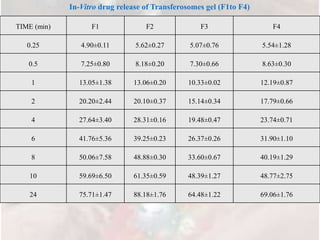
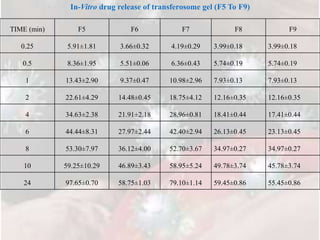
3) In vitro drug release studies through a cellophane membrane showed sustained release of Tramadol HCl from the transferosome gel formulations over 24 hours, with F5 releasing 88.18%









![DRUG PROFILE:
• Name of Drug : Tramadol hydrochloride
• IUPAC name: (1R,2R)-rel-2-[(Dimethylamino)methyl]-1-(3-
methoxyphenyl)cyclohexanol
• Category: Opioid analgesic
• Chemical formula: C16H25NO2.HCl
• Molecular weight: 299.84 g/mol
• Structure:
• Solubility: Freely soluble in water, methanol, ethanol and slightly soluble
in acetone
• Dosage form: Tablets, SR tablets, capsules, Transfersomalgel, injections.](https://image.slidesharecdn.com/transfersomes-160322091258/85/Transfersomes-by-S-Shivani-Shastrulagari-10-320.jpg)