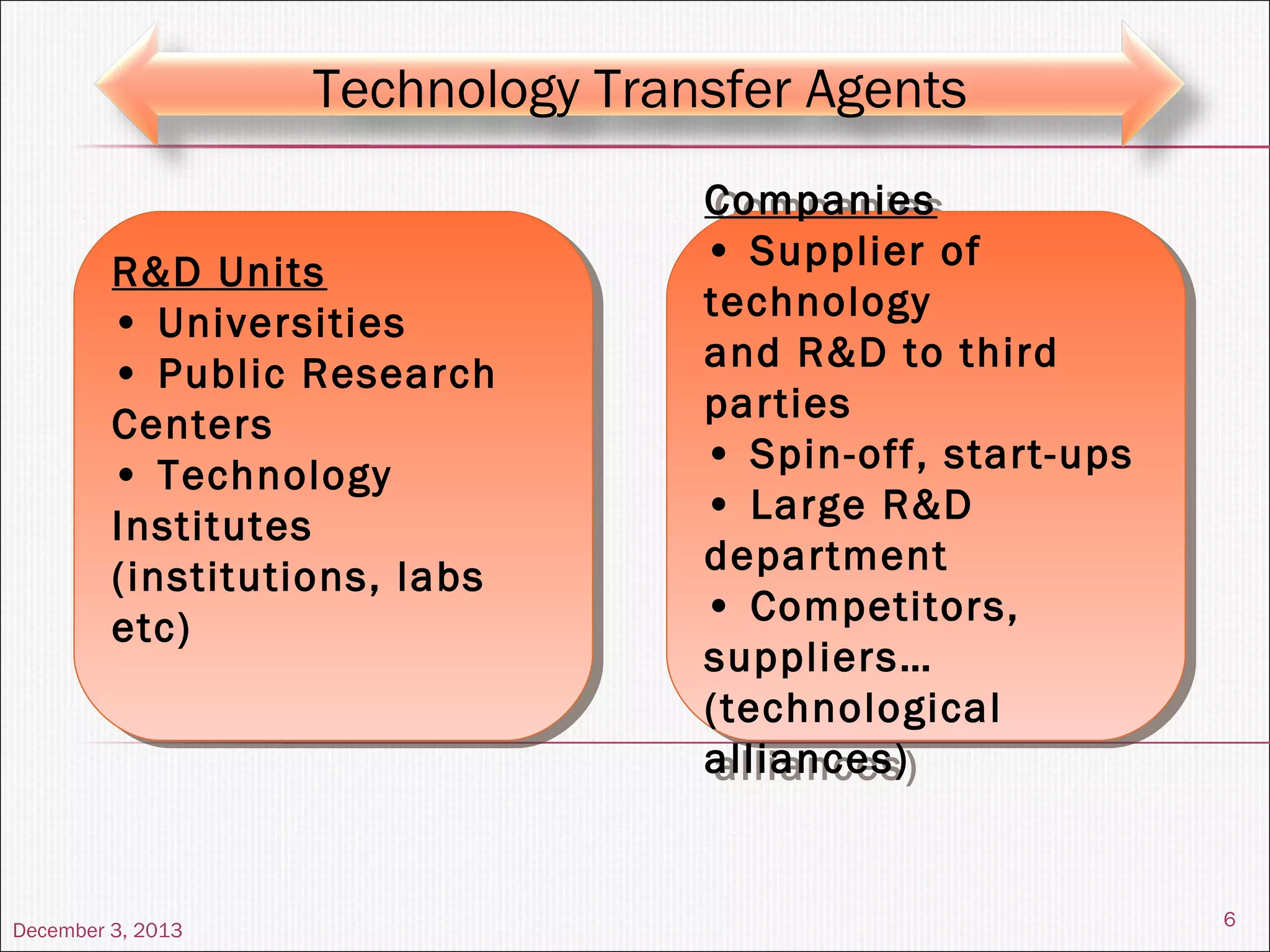
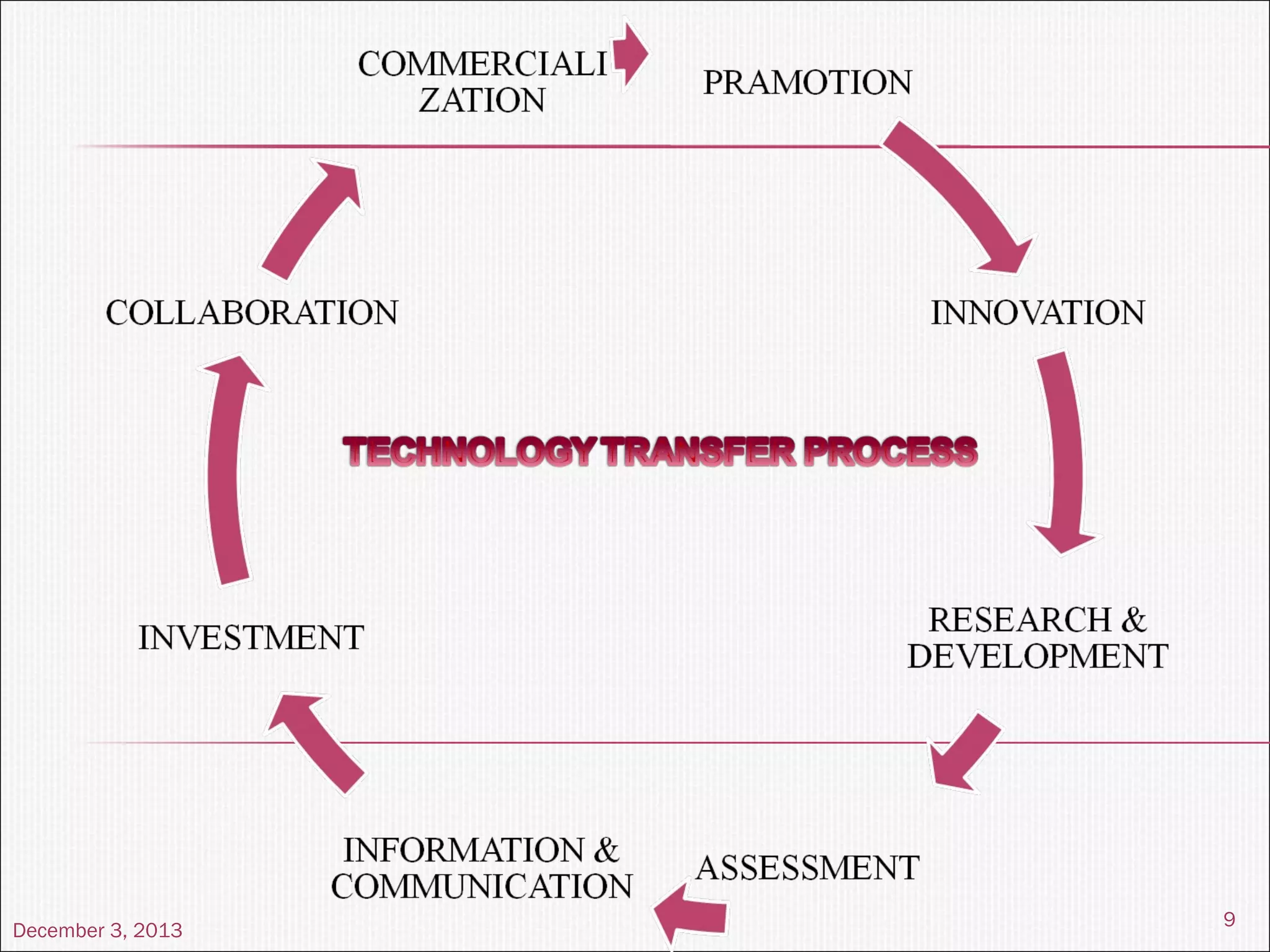
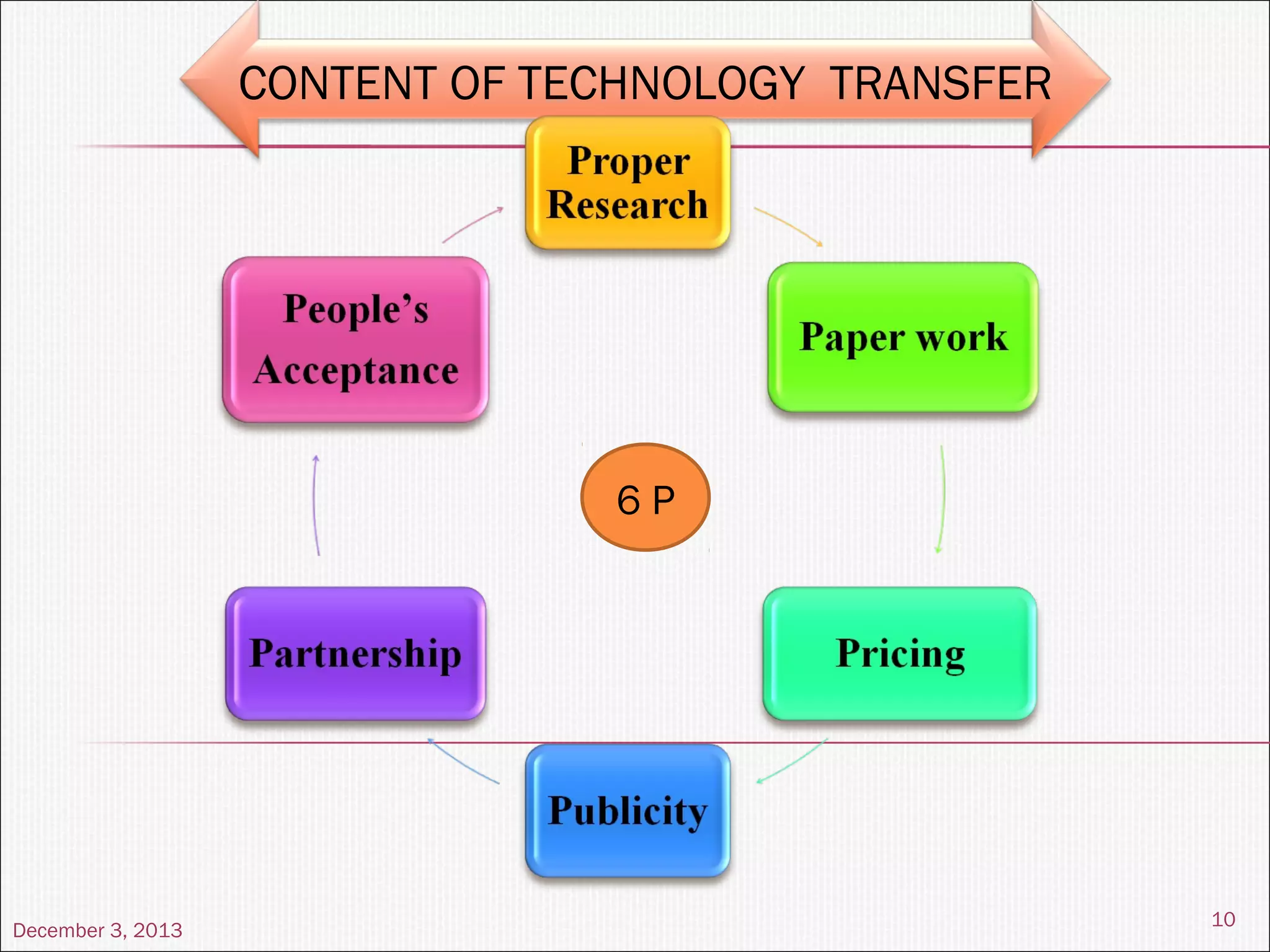
This document summarizes a seminar on technology transfer presented by Chandan Kr. Singh. It discusses what technology transfer is, how it occurs, the agents involved, types of technology, the constituents and content of the technology transfer process, roles and responsibilities of the technology transfer team, factors affecting technology transfer, and examples of technology transfer.