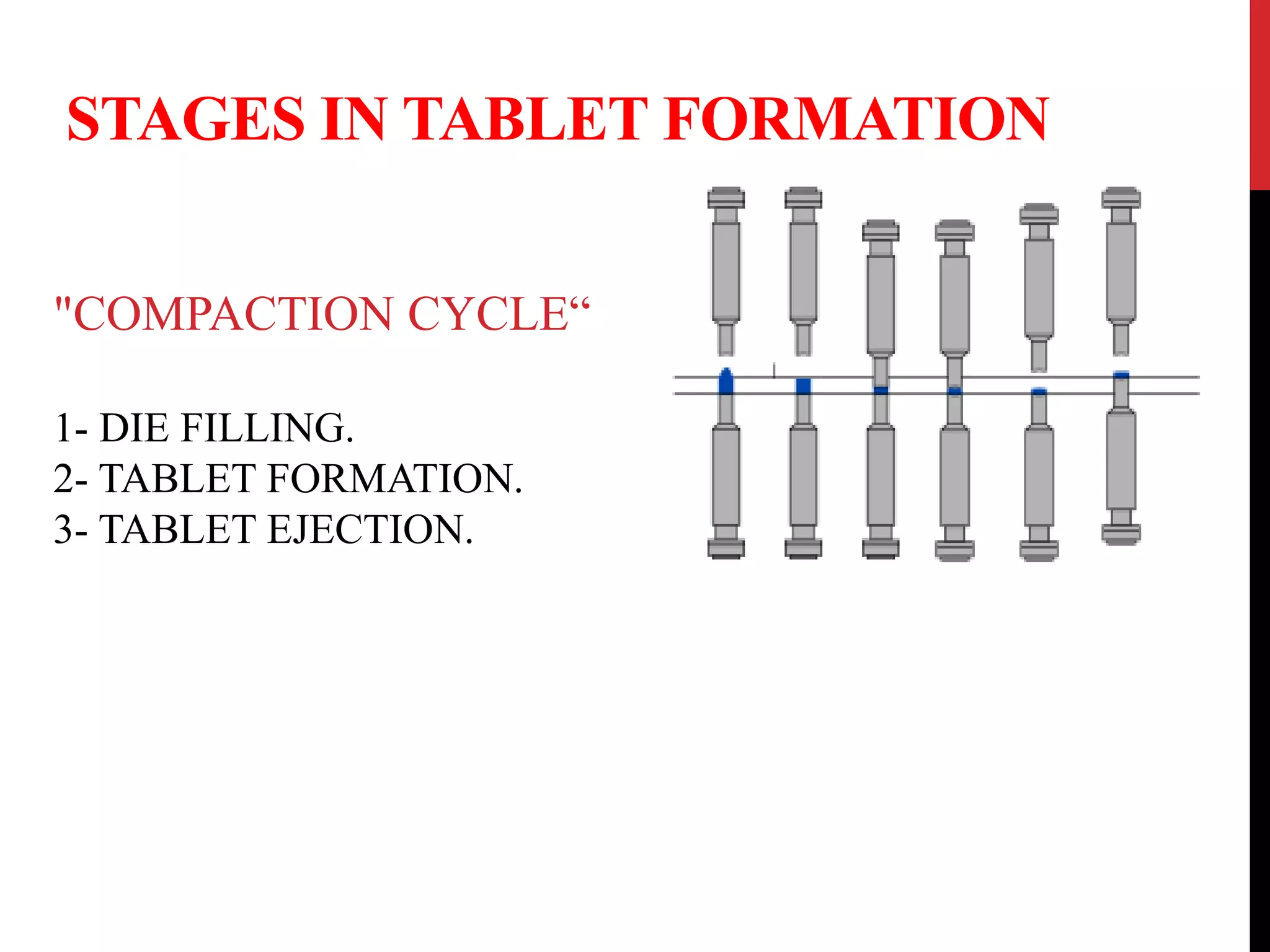
This document discusses different types of tablets and the tablet manufacturing process. It begins by defining what a tablet is and listing some key advantages such as precise dosing, low cost, and stability. It then describes different types of tablets including compressed, coated, chewable, and those for different routes of administration. The document outlines common excipients used in tablets and their functions. It explains the importance of granulation and describes different granulation methods including dry, wet, and direct compression. The wet granulation process is outlined in detail including mixing, granulation, and drying steps.