This document summarizes guidelines for stability testing according to ICH guidelines. The key points are:
1) ICH guidelines are most commonly accepted and provide information on stability testing in the EU, Japan, and US. Stability testing aims to provide evidence of how quality varies over time under different conditions.
2) The objectives of ICH are more economical use of resources, eliminating delays in global development and availability of medicines, and maintaining safeguards for quality, safety, and efficacy.
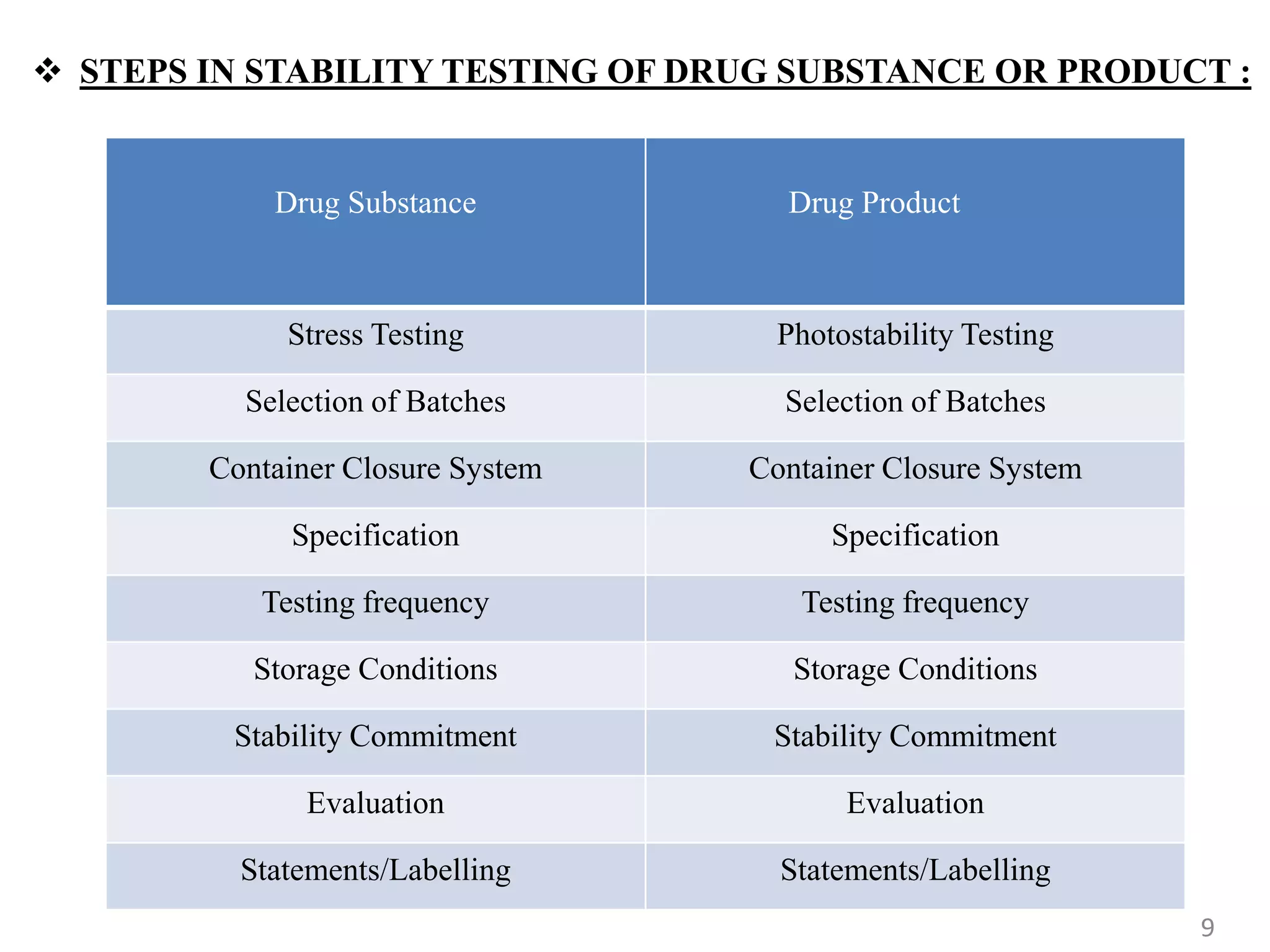
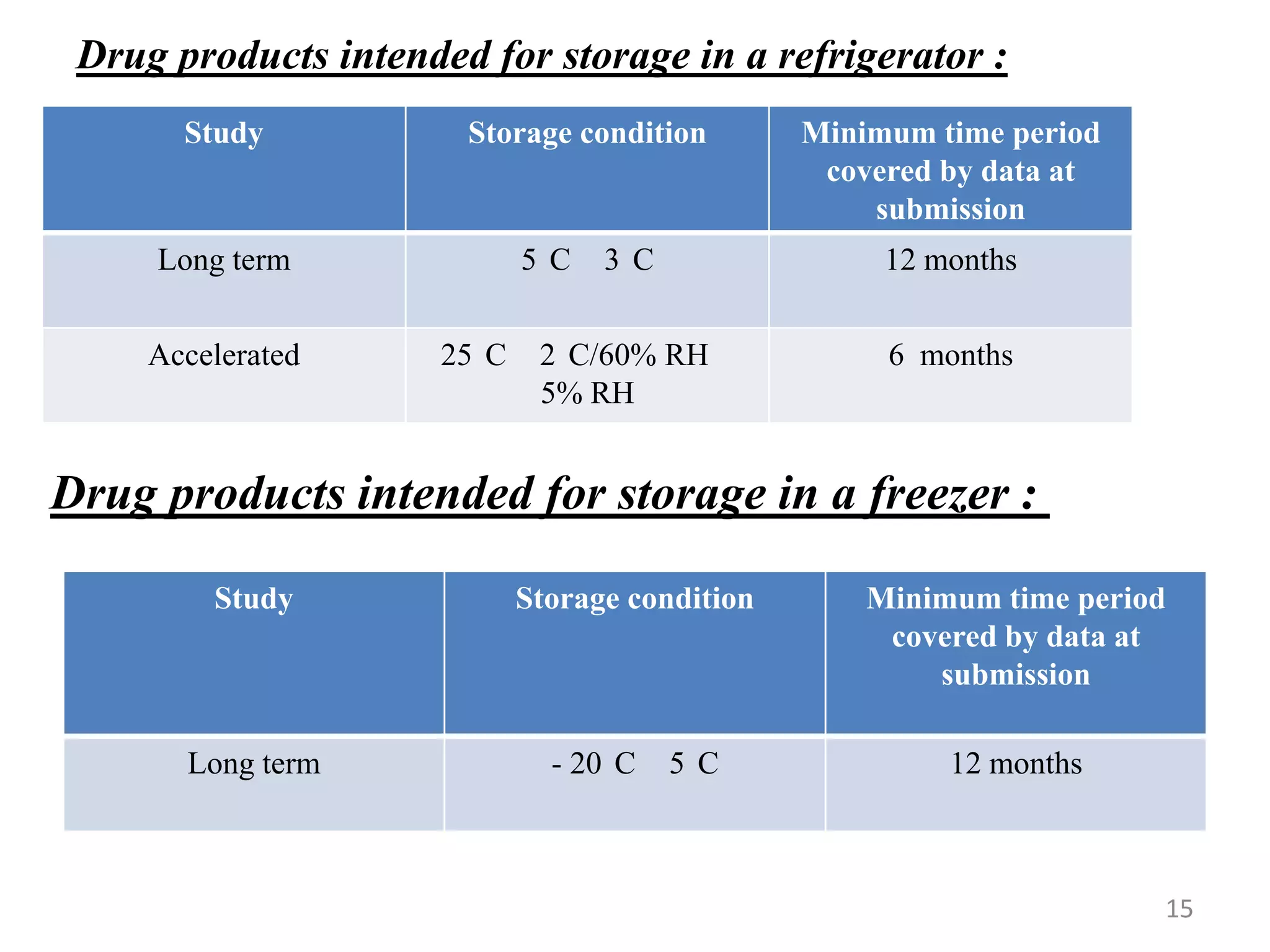
3) Stability topics covered by ICH include testing, validation, impurities, specifications, and manufacturing. This summary focuses on stability testing guidelines for new drug substances and products.