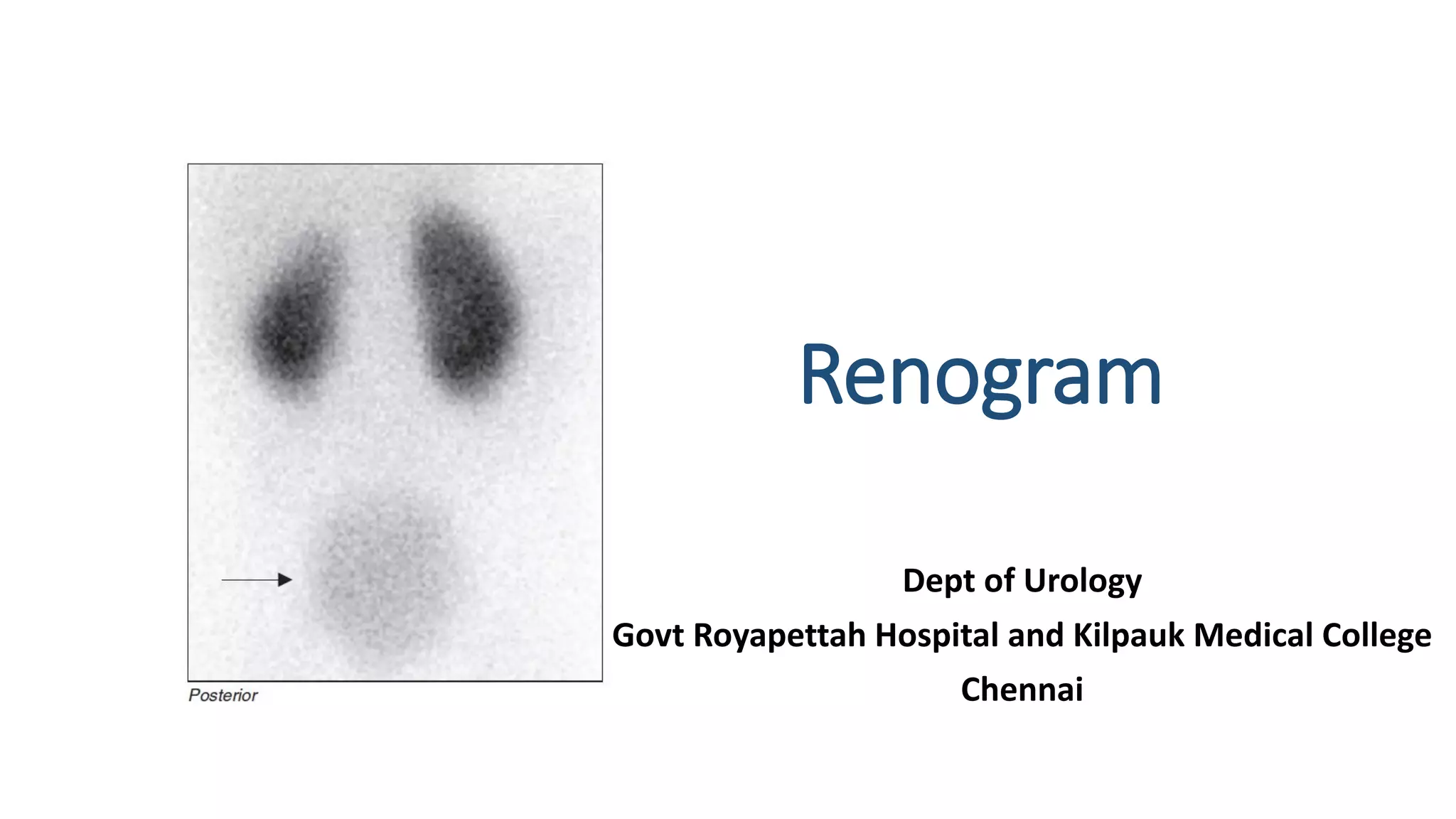
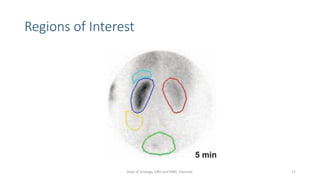
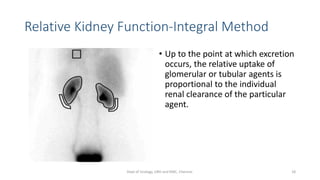
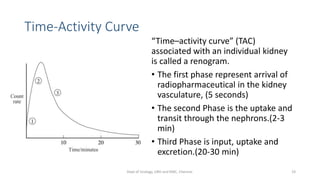
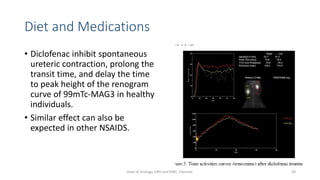
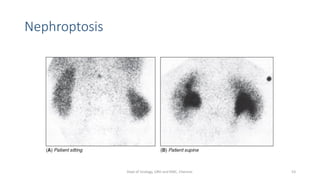
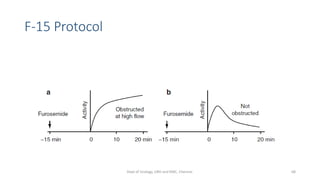
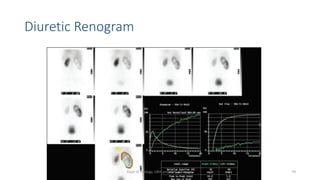
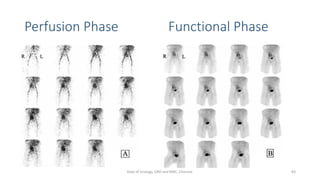
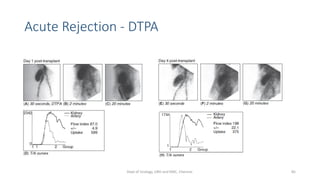
This document describes the renogram procedure. It provides details on:
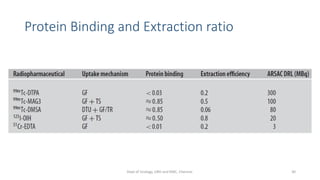
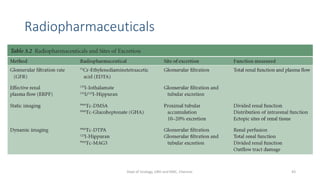
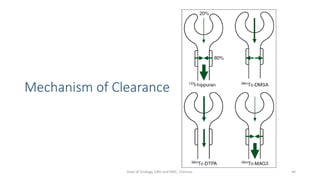
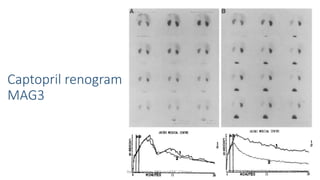
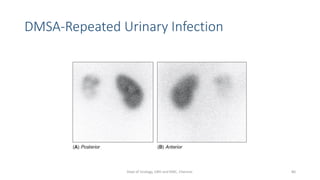
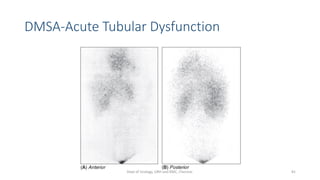
- The radiopharmaceuticals used, including 99mTc-DTPA, 99mTc-MAG3, and 99mTc-DMSA
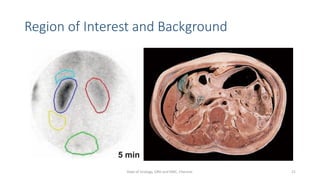
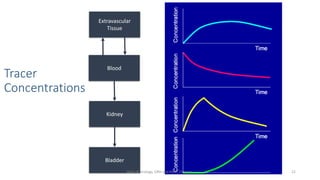
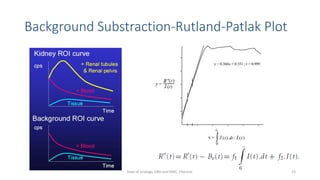
- How the procedure is performed, including patient preparation, image acquisition, and time-activity curve analysis
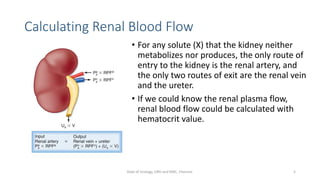
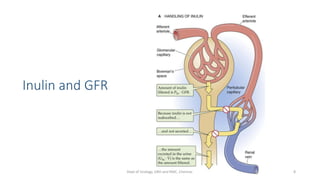
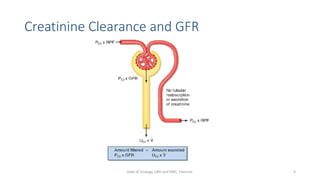
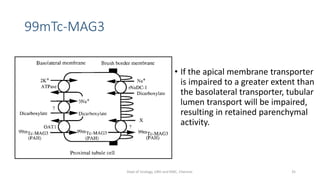
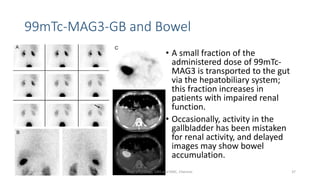
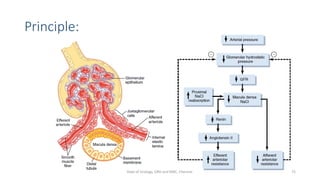
- The roles of the radiopharmaceuticals in evaluating renal blood flow, glomerular filtration rate, and renal handling and excretion
- Factors that can affect the procedure such as hydration, medications, and kidney positioning