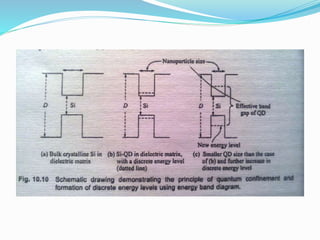
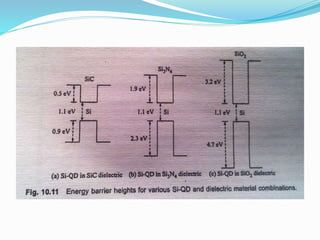
The document discusses quantum dot solar cells (QDSCs). QDSCs use quantum dots as the light-absorbing material instead of bulk semiconductors like silicon. Quantum dots have tunable bandgaps based on their size, allowing different energy levels to be harvested from the solar spectrum. This could enable higher efficiency multi-junction solar cells. The document outlines the history of QDSCs, describes how quantum dots exhibit quantum confinement effects, and discusses methods for fabricating quantum dots with different bandgaps through controlling their size and composition.