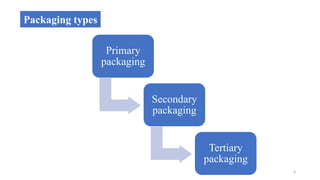
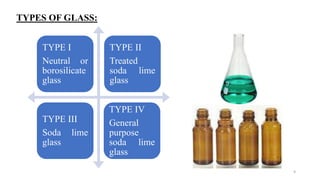
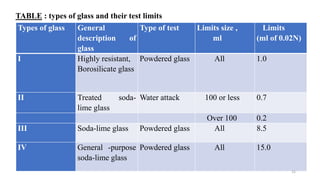
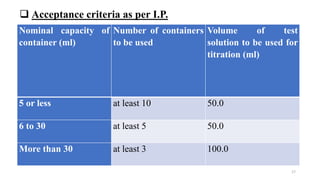
This document discusses quality control tests for containers, closures, and packaging materials. It outlines various tests for glass and plastic containers like powdered glass test, water attack test, and water vapor permeability test. Tests are also described for closures, including fragmentation test, sterility test, and tests for properties like pH and reducing substances of aqueous extracts of closures. Various types of primary, secondary, and tertiary packaging are also mentioned, along with quality control tests for blister packs and strips to check for proper sealing and prevention of moisture ingress.