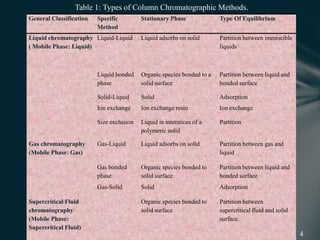
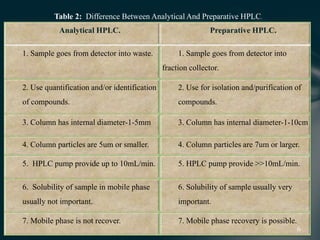
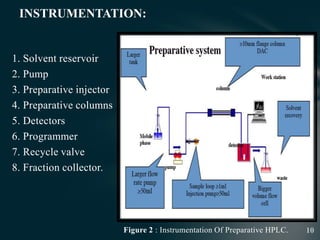
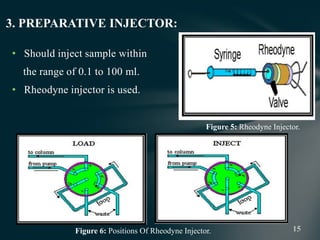
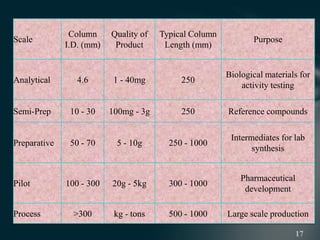
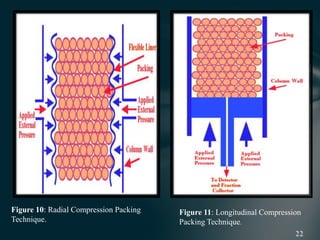
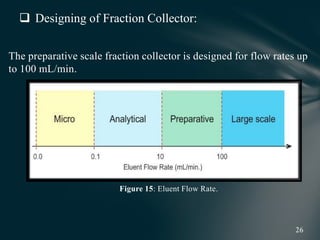
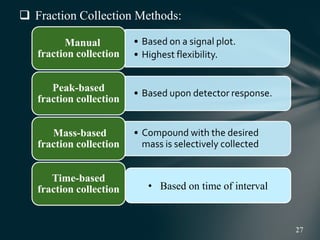
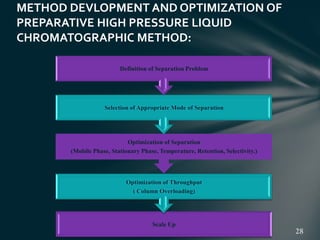
This document presents information on preparative high pressure liquid chromatography. It begins with an introduction to chromatography and classification of column chromatographic methods. It then discusses the differences between analytical and preparative HPLC and the objectives, instrumentation, method development, applications, and commercially available instruments for preparative HPLC. The instrumentation section describes the major components of a preparative HPLC system including the solvent reservoir, pump, injector, columns, detectors, fraction collector and more. Method development and optimization factors like mobile phase selection, temperature, retention, selectivity, and column overloading techniques are also covered.