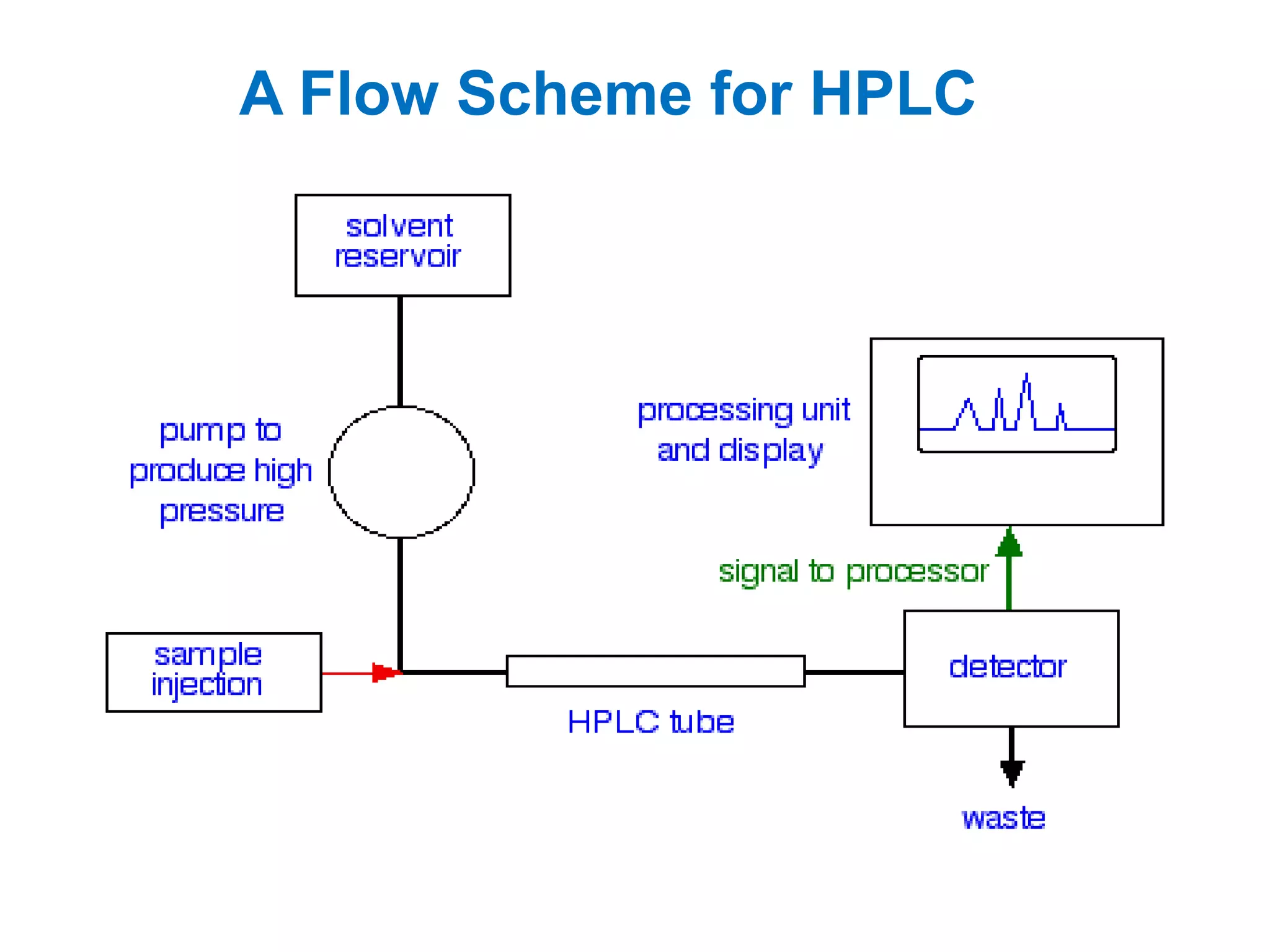
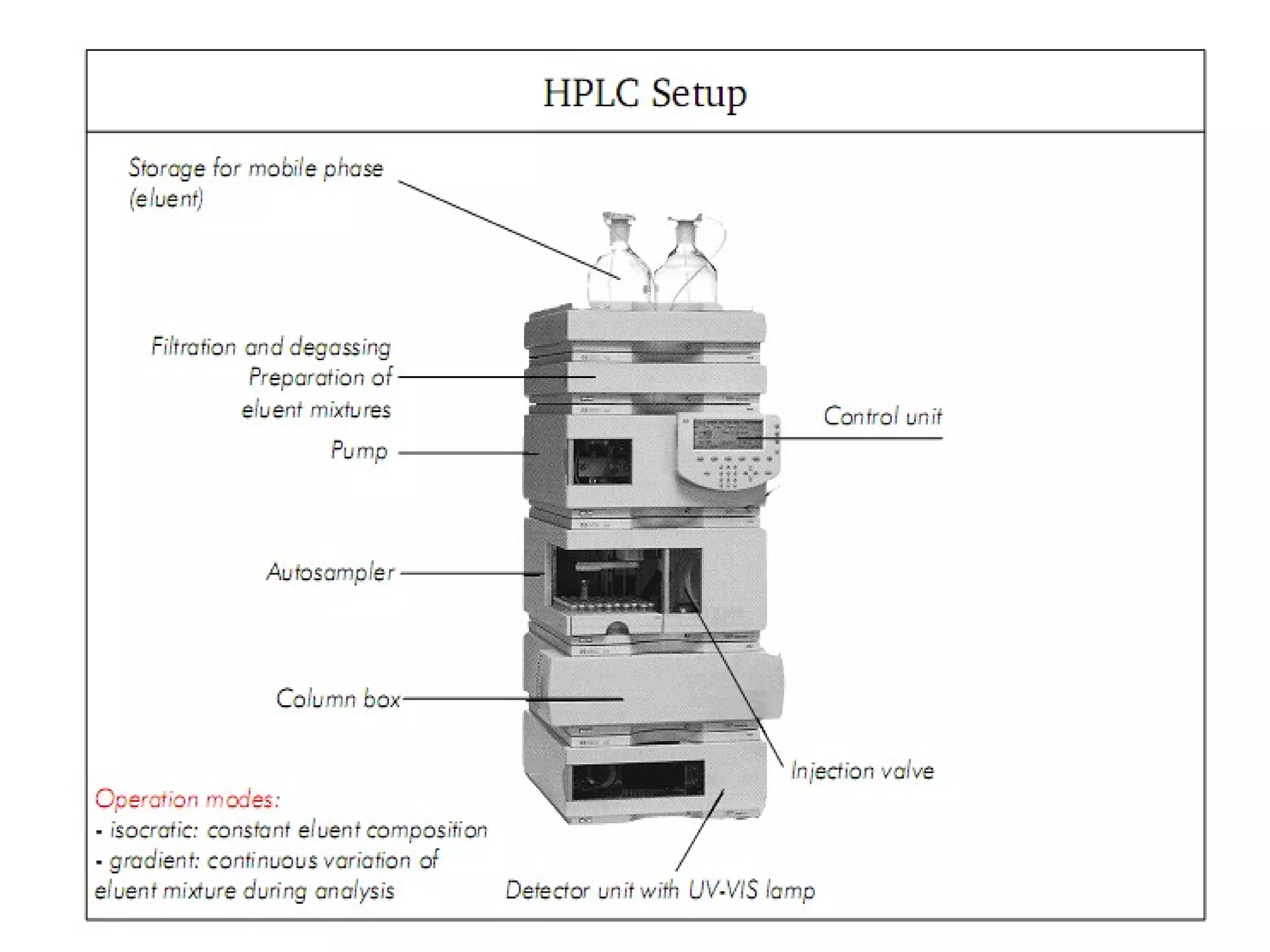
High performance liquid chromatography (HPLC) is a separation technique used to separate mixtures. It uses columns packed with small particle sizes under high pressure, allowing better separation than traditional liquid chromatography. HPLC involves pumping a mobile phase through a column containing a stationary phase, separating components as they flow through at different rates based on interactions with the phases. Components are then detected and quantified as they exit the column. HPLC provides rapid, sensitive, and precise separation of mixtures and is widely used in fields like pharmaceuticals, chemistry, and environmental analysis.