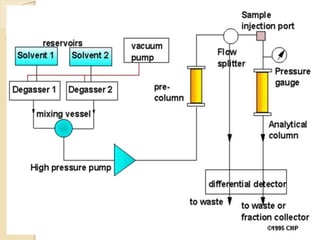
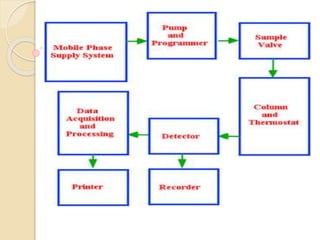
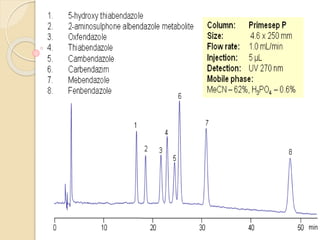
High-performance liquid chromatography (HPLC) is an analytical chemistry technique used to separate, identify, and quantify components in mixtures. It works by forcing a pressurized liquid solvent through a column packed with adsorbent particles under high pressure. This allows for better separation than traditional column chromatography due to smaller particle sizes and detection methods. HPLC has applications in manufacturing, legal, research, and medical fields such as drug analysis, food testing, and pharmaceutical development.