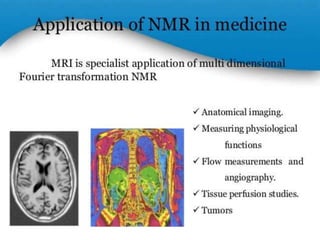
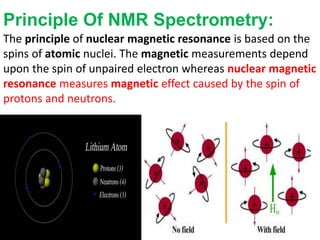
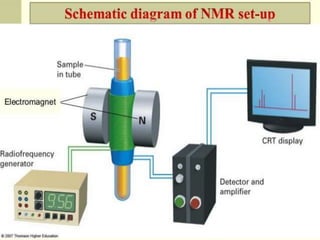
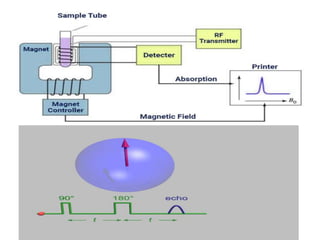
Nuclear magnetic resonance spectrometry (NMR) involves observing local magnetic fields around atomic nuclei. 1H NMR determines the type and number of hydrogen atoms, and 13C NMR determines carbon atoms in a molecule. NMR works based on atomic nuclei spins in a magnetic field, with protons in different environments absorbing at slightly different frequencies. Modern NMR spectrometers use a constant magnetic field and varying radio frequencies to achieve resonance of nuclei such as 1H, 13C, 19F and 31P, which give rise to NMR signals.









![Chemical shift
The relative energy of a particular nucleus resulting from its
local environment is called chemical shift.
NMR spectra show the applied field strength increasing
from left to right.
Left part is downfield, the right is upfield.
Nuclei that absorb on upfield side are strongly shielded
where nuclei that absorb on downfield side is weakly
shielded.
Chart caliberated versus a reference point, set as 0,
tetramethylsilane [TMS]](https://image.slidesharecdn.com/nmrspectrometry-220127094805/85/Nmr-spectrometry-13-320.jpg)