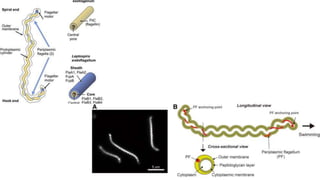
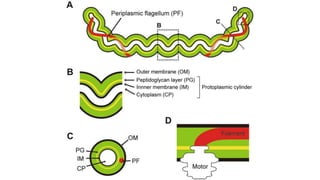
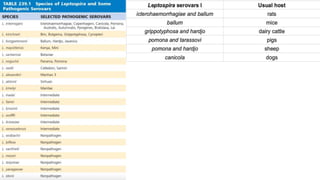
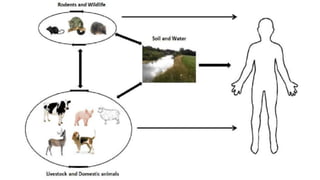
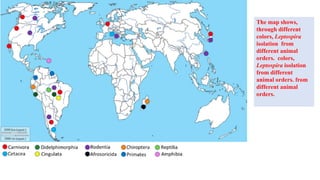
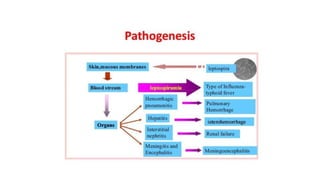
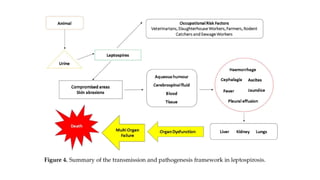
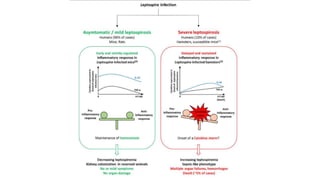
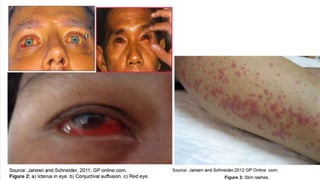
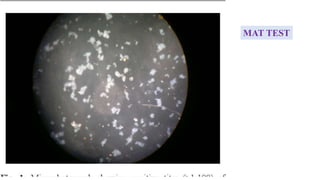
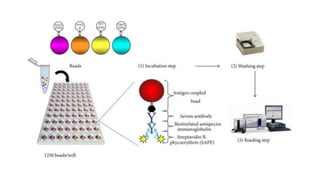
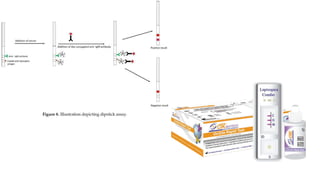
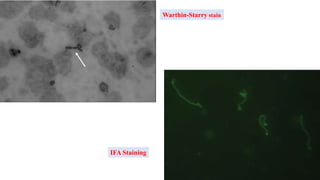
Leptospira are thin, spiral-shaped bacteria that can cause leptospirosis, a zoonotic disease. They have two flagella that allow for motility. Leptospirosis is transmitted through contact with urine from infected animals. It has varied clinical manifestations from mild flu-like symptoms to severe jaundice, kidney damage, and hemorrhaging known as Weil's disease. Diagnosis is made through microscopic agglutination testing of antibodies in serum. Proper treatment requires antibiotics.