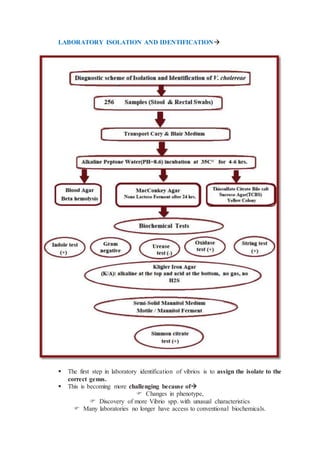
Vibrio species are gram-negative, motile, halophilic rods found predominantly in aquatic environments, with twelve species considered human pathogens, including V. cholerae and V. parahaemolyticus. Their growth is stimulated by sodium chloride, and they can exhibit varying colony morphologies depending on environmental factors and media used. Laboratory identification of vibrios involves various biochemical tests, susceptibility assays, and cultural techniques to differentiate them from other similar organisms.