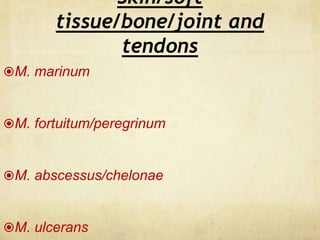
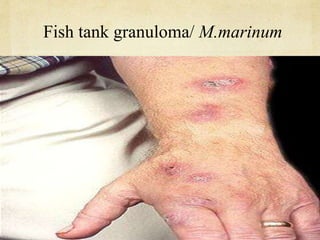
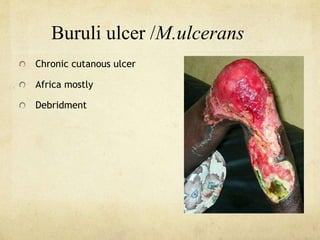
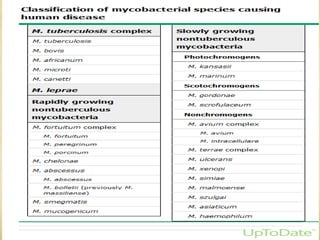
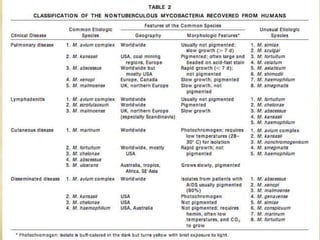
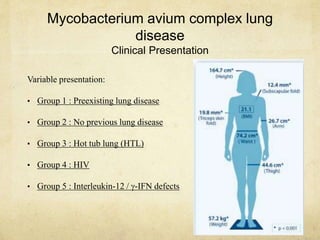
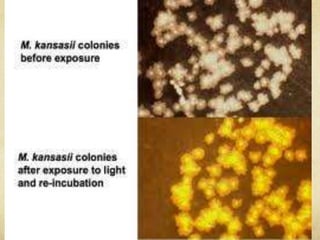
This document discusses atypical mycobacteria (NTM). It describes the Runyon classification system for NTM based on pigment production and growth rate. The most common NTM species are Mycobacterium avium complex (MAC) and Mycobacterium kansasii. MAC is ubiquitous in the environment and a major cause of pulmonary NTM disease. M. kansasii is associated with tap water and causes lung disease resembling tuberculosis, often in older male smokers with COPD. Host immune deficiencies increase risk of NTM infections.









































![NTM Infection Presentation
Nontuberculous Mycobacterial Lung Disease
Common CXR findings:
A. Hazy opacity abutting the
heart border [#1]
1](https://image.slidesharecdn.com/ntm-150309121151-conversion-gate01/85/ATYPICAL-MYCOBACTERIA-47-320.jpg)
![CommonCXR findings:
B. Retrosternal shadowing
overlying the cardiac
silhouette [#2]
NTM Infection Presentation
2](https://image.slidesharecdn.com/ntm-150309121151-conversion-gate01/85/ATYPICAL-MYCOBACTERIA-48-320.jpg)
![NTM Infection Presentation
Nontuberculous Mycobacterial Lung Disease
Common HRCT scan findings:
A.Volume loss and variable
opacities of the right-middle
lobe (RML) and lingular
segment of the left-upper
lobe (LING)
B. Saccular or “honeycomb”
bronchiectasis in both the
RML & LING
C. Diffuse cylindrical and
varicoid bronchiectasis with
scattered nodular opacities
[RML] [LING]](https://image.slidesharecdn.com/ntm-150309121151-conversion-gate01/85/ATYPICAL-MYCOBACTERIA-49-320.jpg)
















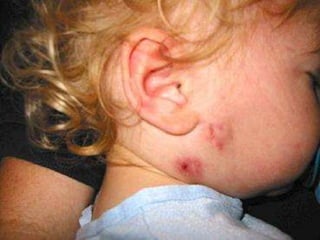
![PPD tuberculin
Intermediate strength (5 tuberculin unit [TU] PPD tuberculin
weakly reactive skin test (5–9 mm) due to cross-reactivity with
NTM
some children may be negative,
one- third will have reactions with 10 mm or more induration
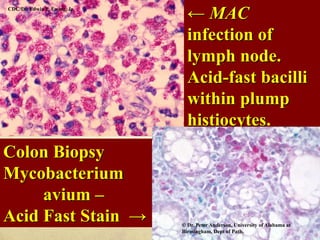
Distinguishing tuberculous from nontuberculous lymphadenitis
is key
Presumptive diagnosis of NTM
lymphadenitishistopathologic appearance of the lymph
node showing caseating granulomata with or without AFB and
a negative tuberculin skin test.
Failure of the node to yield M. tuberculosis provides](https://image.slidesharecdn.com/ntm-150309121151-conversion-gate01/85/ATYPICAL-MYCOBACTERIA-67-320.jpg)